Cell/Gene Transplant
Alan W. Flake
Department of Surgery, University of Pennsylvania, Children’s Institute for Surgical Sciences, Children’s Hospital of Philadelphia, Philadelphia, Pennsylvania 19104-4318.
Although not classically considered within the realm of surgical treatment, cellular and genetic transplantation will become increasingly important for surgeons in the future. The scientific revolution that is now underway is providing increasing insight into stem cell biology and gene regulation and function. In the coming decades, translational application of this knowledge will have a profound impact on the fields of tissue engineering, cellular and organ transplantation, tolerance induction, oncology, wound healing, and fetal therapy. In no field of surgery will the importance of these advances be greater than in pediatric surgery. Because of unique aspects of fetal and pediatric development, the potential for cellular and gene therapy early in life is greater than at any other time. In this chapter, current concepts in cellular transplantation and gene transfer technology are reviewed in the context of relevance to surgical treatment and specifically pediatric surgical practice.
NEW PARADIGMS IN STEM CELL BIOLOGY
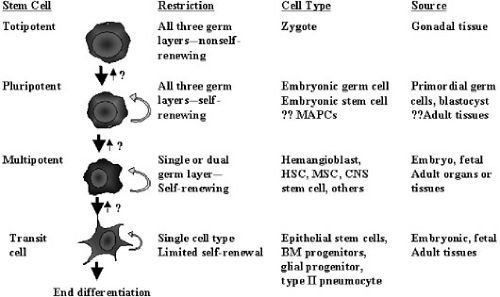
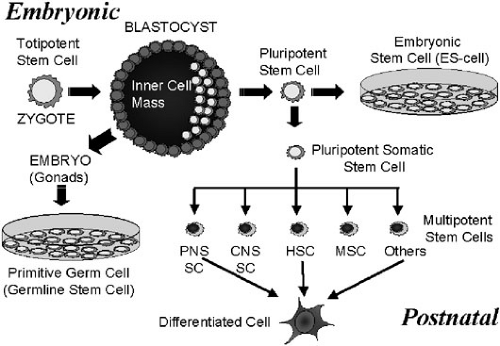
For the purposes of this chapter, a stem cell will be defined as “a cell that can self-replicate and can give rise to more than one type of mature daughter cell.” Thus, the term stem cell incorporates a broad range of cells with different capacities for proliferation and differentiation. Terminology used to classify stem cells includes “totipotent”—stem cells capable of giving rise to an intact organism, including germinal tissues; “pluripotent”—stem cells capable of giving rise to cells derived from all three germ layers, but not capable of independently forming an organism; and “multipotent or organ specific”—stem cells capable of giving rise to the cells comprising a single organ system or tissue (Fig. 51-1). In the classical paradigm of stem cell biology, totipotent and pluripotent stem cells existed only in embryonic life, and were lost with development and aging so only multipotent or organ specific stem cells persisted in adult life (Fig. 51-2). Similarly, lineage specification of stem cells and differentiation were considered unidirectional events (i.e., once they occurred there was no turning back). At a molecular level, this irreversibility was attributed to changes in chromatin binding and methylation events that occurred during differentiation. This progressive restriction of the potential of stem cells occurred relatively early in development so that by postnatal life, all stem cells, with the exception of germ cells, were believed to have, at most, single tissue or organ specificity.
Since the mid-1990s, there have been a number of observations that have shaken the foundations of stem cell biology and resulted in a “new paradigm.” This paradigm shift is of more than theoretical importance because it significantly broadens the realm of possibility for stem cell-based therapeutic applications, including several applications of surgical relevance.
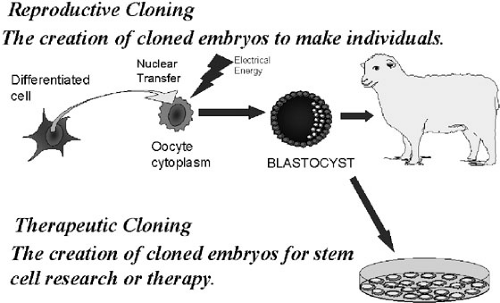
The first observation of importance was the use of nuclear transfer technology to achieve “reproductive cloning” (1). The ability to produce an intact organism under the control of an adult-derived nucleus was a remarkable demonstration of developmental plasticity. When an adult-derived nucleus was placed in oocyte cytoplasm, under specific conditions, the changes in chromatin structure that govern differentiation could be reversed, and the nucleus could be made to control development to term. This observation argued that under appropriate conditions, any nucleus, at any stage of differentiation, could be converted to a totipotent stem cell with the capability of differentiation into stem cells or differentiated cells of any organ or tissue. Thus, not only was reproductive cloning a possibility, but also “therapeutic cloning” (i.e., the cloning of one cell to make other cells or tissues with therapeutic potential) could theoretically also be achieved (Fig. 51-3) (2).
The second observation of fundamental importance was the cultivation of human embryonic stem (ES) cells (3,4). Although ES cells had long been available in the
mouse and had been derived from a number of mammalian species, other species had proven refractory to ES cell cultivation. The ability to culture and expand human embryonic stem cells derived from either blastocysts or primordial germ cells raised new therapeutic possibilities. In addition to possibly providing large numbers of pure populations of cells for transplantation or tissue engineering, ES cells would also lend themselves to several strategies for the prevention of immunologic tissue rejection after transplantation, which is detailed below.
mouse and had been derived from a number of mammalian species, other species had proven refractory to ES cell cultivation. The ability to culture and expand human embryonic stem cells derived from either blastocysts or primordial germ cells raised new therapeutic possibilities. In addition to possibly providing large numbers of pure populations of cells for transplantation or tissue engineering, ES cells would also lend themselves to several strategies for the prevention of immunologic tissue rejection after transplantation, which is detailed below.
The third observation that has fostered the development of the new paradigm in stem cell biology is the observation of adult-derived pluripotent stem cells (5). This challenged the previous dogma of developmental and age-induced restriction of potential in all adult stem cell populations and suggested that there may be adult sources of cells that could be used for the same broad range of therapeutic applications promised by embryonic stem cells.
Finally, in the past few years, there has been an explosion of in vitro and in vivo data supporting the concept of stem cell “plasticity” (6,7,8,9,10,11,12,13,14,15). The concepts that stem cells can undergo a “conversion” event and turn into another type of stem cell (transdifferentiation) or that more differentiated cells can become less differentiated cells (de-differentiation) remain highly controversial (16,17,18). One of the reasons for skepticism is that essentially all the observations made supporting adult-derived pluripotent stem cells or events described as “transdifferentiation” or “dedifferentiation” have been confined to highly contrived in vitro circumstances, or in vivo models of injury or extreme selective pressure. Many of the early studies observing stem cell conversion events could be criticized
for the methodology employed (lack of demonstration of clonality) and, more recently, the argument of fusion as an explanation for transdifferentiation has gained support (19,20,21). However, even if fusion is the explanation for all such observations (which is doubtful), it remains a mechanism of genetic transfer that may be clinically exploited. In summary, it seems likely that pluripotent stem cells do persist beyond the embryonic state and that, at least under specific circumstances, adult stem cells can display some degree of “plasticity.”
for the methodology employed (lack of demonstration of clonality) and, more recently, the argument of fusion as an explanation for transdifferentiation has gained support (19,20,21). However, even if fusion is the explanation for all such observations (which is doubtful), it remains a mechanism of genetic transfer that may be clinically exploited. In summary, it seems likely that pluripotent stem cells do persist beyond the embryonic state and that, at least under specific circumstances, adult stem cells can display some degree of “plasticity.”
The consideration of new paradigms is usually accompanied by an acceleration of progress and expansion of knowledge. The current state of stem cell biology can be described as a field in rapid evolution in which new and significant information is being gathered at an unprecedented rate. Successful clinical translation of stem cell biology will reside in our understanding of stem cell regulation and the ability to manipulate stem cell populations to perform desired tasks. Thus, genetic manipulation and regulation of stem cell activity may play a critical role in the successful application of stem cell therapy in the future. In addition, cellular transfer can be considered the simplest approach to regulated gene transfer. Thus, the subjects of stem cell transplantation and gene transfer often overlap, and strategies combining the two technologies may ultimately result in powerful therapeutic approaches.
GENE TRANSFER—PROGRESS AND PROBLEMS
Gene therapy continues to be embroiled in controversy, its seemingly unlimited potential obscured by repeated disappointments and, more recently, adverse events. The year 2000 brought the first clinical gene therapy success [i.e., treatment of X-linked severe combined immune deficiency (X-SCID)] (22), only to have this dramatic achievement undermined by the occurrence of leukemia in two patients. This and other adverse events threaten to overshadow the substantial progress made in gene transfer technology in recent years. Slowly, but surely, methods for gene transfer are being developed that will have greater safety, specificity, and efficacy than ever before. Although complex issues remain to be solved, it is likely that successful gene therapy strategies will be developed and proven within the next few years. The technology of gene transfer can be divided into viral vector-based gene transfer and nonviral gene transfer. Because of the limited scope of this chapter and the limited efficiency of nonviral-based gene transfer thus far, only the current state of viral-based gene transfer is reviewed here.
The Use of Viral Vectors for Gene Transfer
Viruses are highly evolved biological machines that efficiently penetrate hostile host cells and exploit the hosts’ cellular machinery to facilitate their replication. Ideally, viral vectors harness the viral infection pathway, but avoid the subsequent replicative expression of viral genes that causes toxicity. This is traditionally achieved by deleting some, or all, of the coding regions from the viral genome, but leaving intact those sequences that are needed for the vector function, such as elements required for the packaging of viral DNA into virus capsid, or the integration of vector DNA into host chromatin. The chosen expression cassette is then cloned into the viral backbone in place of those sequences that were deleted. The deleted genes encoding proteins involved in replication or capsid/envelope proteins are included in a separate packaging construct. The vector genome and packaging construct are then
cotransfected into packaging cells to produce recombinant vector particles.
cotransfected into packaging cells to produce recombinant vector particles.
TABLE 51-1 The Five Main Viral Vector Groups. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Given the diversity of therapeutic strategies and disease targets involving gene transfer, it is not surprising that a large number of vector systems have been devised. Although there is no single vector suitable for all applications, the characteristics that are desirable for all vectors to be potentially clinically useful are (1) the ability to be reproducibly and stably propagated, (2) the ability to be purified to high titers, (3) the ability to mediate targeted delivery (i.e., to avoid widespread vector dissemination), and (4) the ability to achieve gene delivery and expression without harmful side effects. There are, at the present time, five main classes of vectors that at least under specific circumstances, satisfy these requirements: oncoretroviruses, lentiviruses, adeno-associated viruses (AAVs), adenoviruses, and herpes viruses. Table 51-1 compares the general characteristics of these vectors.
Oncoretroviruses and lentiviruses are “integrating”; that is, they insert their genomes into the host cellular chromatin. Thus, they share the advantage of persistent gene expression. Nonintegrating viruses can achieve persistent gene expression in nondividing cells, but integrating vectors are the tools of choice if stable genetic alteration needs to be maintained in dividing cells. It is important to note, however, that stable transcription is not guaranteed by integration and that transgene expression from integrated viral genomes can be silenced over time (23). Oncoretroviruses and lentiviruses differ in their ability to penetrate an intact nuclear membrane. Whereas retroviruses can only transduce dividing cells, lentiviruses can naturally penetrate nuclear membranes and can transduce nondividing cells, making them particularly useful for stem cell targeting applications (24,25). Because of this difference, lentiviral vectors are superceding retroviral vectors for most applications. Both types of vector, because of their ability to integrate, share the potential hazard of alteration of the host cell genome. This could lead to the undesirable complications of human germ-line alteration or insertional mutagenesis, particularly important considerations for pediatric or fetal gene therapy (26). Nevertheless, these vectors have proven most efficient for long-term gene transfer into cells in rapidly proliferative tissues and for stem cell-directed gene transfer.
Nonintegrating vectors include adenoviral, AAV, and herpes viral vectors. Adenoviral vectors have the advantages of broad tropism, moderate packaging capacity, and high efficiency, but carry the usually undesirable properties of high immunogenicity and consequent short duration of gene expression. Modifications of adenoviral vectors to reduce immunogenicity and further increase the transgene capacity have primarily consisted of deletion of “early” (E1–E4) viral genes that encode immunogenic viral proteins responsible for the cytotoxic immune response (27,28). The most important advance, however, has been the development of helper-dependent adenoviruses (HD-Ads) that are deleted of all viral genes, thus eliminating the immune response to adenoviral-associated proteins (29).
AAV is a helper-dependent parvovirus that in the presence of adenoviral infection or herpes virus infection undergoes a productive replication cycle. AAV vectors are single-strand DNA vectors and represent one of the most promising vector systems for safe long-term gene transfer and expression in nonproliferating tissues. AAV is the
only vector system for which the wild-type virus has no known human pathogenicity adding to its safety profile. In addition, the small size and simplicity of the vector particle make systemic administration of high doses of vector possible without eliciting an acute inflammatory response or other toxicity. Although the majority of AAV’s vector genome after transduction remains episomal, approximately a 10% rate of integration has been observed (30). There are two primary limitations of AAV vectors. The first is the need to convert single-stranded DNA genome into a double strand, limiting the efficiency of transduction. Recently, this obstacle has been overcome by development of double-stranded vectors that exploit a hairpin intermediate of the AAV replication cycle (31). Although these vectors can mediate a 10- to 100-fold increase in transgene expression in vitro and in vivo, they can only package 2.4 kb of double-stranded DNA, limiting their therapeutic usefulness. This relates to the second primary limitation of AAV vectors, which is limited packaging capacity (4.8 kb of single-stranded DNA). One approach to address this limitation is to split the expression cassette across two vectors, exploiting the in vivo concatemerization of rAAV genomes. This results in reconstitution of a functional cassette after concatemerization in the cell nucleus (32,33). Finally, an approach that has become commonplace for enhancing or redirecting the tissue tropism of AAV vectors is to pseudotype the vectors with capsid proteins from alternate serotypes of AAV (34). Although most rAAV vectors have been derived from AAV2, eight distinct AAV serotypes have been identified thus far, all of which differ in efficiency for transduction of specific cell types. AAV vectors have proven particularly useful for muscle, liver, and CNS-directed gene transfer.
only vector system for which the wild-type virus has no known human pathogenicity adding to its safety profile. In addition, the small size and simplicity of the vector particle make systemic administration of high doses of vector possible without eliciting an acute inflammatory response or other toxicity. Although the majority of AAV’s vector genome after transduction remains episomal, approximately a 10% rate of integration has been observed (30). There are two primary limitations of AAV vectors. The first is the need to convert single-stranded DNA genome into a double strand, limiting the efficiency of transduction. Recently, this obstacle has been overcome by development of double-stranded vectors that exploit a hairpin intermediate of the AAV replication cycle (31). Although these vectors can mediate a 10- to 100-fold increase in transgene expression in vitro and in vivo, they can only package 2.4 kb of double-stranded DNA, limiting their therapeutic usefulness. This relates to the second primary limitation of AAV vectors, which is limited packaging capacity (4.8 kb of single-stranded DNA). One approach to address this limitation is to split the expression cassette across two vectors, exploiting the in vivo concatemerization of rAAV genomes. This results in reconstitution of a functional cassette after concatemerization in the cell nucleus (32,33). Finally, an approach that has become commonplace for enhancing or redirecting the tissue tropism of AAV vectors is to pseudotype the vectors with capsid proteins from alternate serotypes of AAV (34). Although most rAAV vectors have been derived from AAV2, eight distinct AAV serotypes have been identified thus far, all of which differ in efficiency for transduction of specific cell types. AAV vectors have proven particularly useful for muscle, liver, and CNS-directed gene transfer.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree