Background
There is an increasing incidence of pertussis infection in infants too young to be adequately protected via vaccination. Maternal pertussis vaccination during the third trimester of pregnancy is a new strategy to provide protection to newborn infants.
Objective
This study sought to determine the optimal gestational window for vaccination in the third trimester.
Study Design
This prospective study recruited 3 groups of women: an early vaccination group, vaccinated between 28-32 weeks’ gestation; a late vaccination group, vaccinated between 33-36 weeks’ gestation; and an unvaccinated control group. Maternal venous blood was taken prior to pertussis vaccination. At birth, infant cord blood was collected to determine antibody levels to pertussis toxin (PT), pertactin (PRN), and filamentous hemagglutinin (FHA).
Results
In all, 154 women were recruited from April through September 2014. There was no significant difference between maternal PRN and FHA antibody levels among the 3 groups, however, PT was higher in the early compared to late vaccination group ( P = .05). Cord blood antibody levels to PT, PRN, and FHA were significantly higher in those born to vaccinated women compared with unvaccinated controls ( P < .001, P = .001, and P < .001, respectively). Vaccination between 28-32 weeks’ gestation resulted in significantly higher cord blood PT (4.18.0 vs 3.50 IU/mL, P = .009), PRN (5.83 vs 5.31 IU/mL, P = .03), and FHA (5.56 vs 5.03 IU/mL, P = .03) antibody levels than vaccination between 33-36 weeks’ gestation. When adjusted for maternal prevaccination antibody levels, PT levels in early vs late vaccination approached significance ( P = .06). PRN levels were significantly higher in the early vaccination group ( P = .003). There was no significant difference for FHA antibody levels between the 2 groups ( P = .16).
Conclusion
Maternal vaccination during the third trimester is effective in affording higher levels of pertussis antibody protection to the newborn infant. Vaccination early in the third trimester appears more effective than later in pregnancy.
Introduction
Although preventable through vaccination, the incidence of pertussis infection has been on the increase over recent years. The group most vulnerable to significant morbidity (including hospitalization) and mortality, are infants <3 months of age. In recent years, an increasing number of pertussis cases have been diagnosed in this age group despite widespread access to the pertussis vaccine. Several deaths have also been reported in resource-rich settings, triggering a review of vaccination policy.
Newborn infants are most at risk as their immune system is functionally immature and they do not acquire protective levels of antibodies until at least 4 months of age (usually after the second dose of pertussis vaccine). The newborn is therefore reliant on IgG antibodies, acquired through passive transplacental transfer, and IgA antibodies, in breast milk, for protection. Studies have shown low levels of maternal pertussis antibodies available for infant protection in the majority of women of childbearing age. Furthermore, the mother has been identified as the main source of pertussis infection to the newborn.
Maternal vaccination is a strategy that has the potential to afford protection to the neonate via passive transplacental transfer of maternal antibodies. These antibodies protect infants in the first few months of life, prior to the development of active immunity through routine vaccination.
Studies in pregnant women with whole-cell and acellular pertussis vaccine demonstrate safety for mother and fetus. Recent studies confirm that active placental transfer of pertussis-specific antibodies occurs, with peak transfer occurring in the third trimester of pregnancy. The durability of passively acquired pertussis antibodies has also been calculated and data suggest that maternal antibody protection may last up to 4 months of age. Efficacy of maternal vaccination in protecting infants <3 months of age has been reported to be 91%.
These observations make maternal vaccination an attractive intervention to protect newborn infants from pertussis and its complications. In 2011, the United States and the United Kingdom recommended maternal vaccination during pregnancy as a strategy to increase newborn protection. These guidelines recommend vaccination in the third trimester but the optimal gestation window remains unknown. The aim of this study is to examine cord blood pertussis antibody levels comparing early third-trimester vaccination with late third-trimester vaccination.
Materials and Methods
This prospective study evaluated maternal and newborn pertussis-specific antibody levels prior to and following diphtheria, tetanus, and acellular pertussis (dTpa) immunization. Pregnant women at a tertiary obstetric hospital were recruited from April through September 2014. Inclusion criteria were healthy pregnant women with a singleton pregnancy between 28-36+6 weeks who consented to receive dTpa. Women unimmunized for pertussis during their current pregnancy served as controls. Exclusion criteria included women already vaccinated with dTpa in the current pregnancy; immunosuppressed women, such as those with HIV infection; and women considered at high risk for preterm delivery. Women were approached during the third trimester and invited to participate in the study. If they agreed to vaccination then they were allocated to either the early or late group according to their gestation at the time of vaccination. Women vaccinated between 28-32+6 weeks’ gestation comprised the early vaccination group and women vaccinated between 33-36+6 weeks’ gestation comprised the late vaccination group. Women who declined the vaccination but were willing to participate in the study comprised the control group. A prevaccination maternal venous sample was taken from all participants and infant cord blood was collected at delivery.
Prior to the study a sample size calculation based on the effect sizes and associated variances reported by Healy et al was undertaken. We estimated that a minimum sample of 42 patients per group would be required to be able to demonstrate at least a 10% difference in antibody levels between early and late vaccination groups, with 80% power at the 5% significance level. The study of Healy et al, however, did not account for maternal prevaccination levels.
Monash Health Human Research Ethics Committee approved the study and all participants provided informed, signed consent. Demographic data were collected from the women’s medical records and a participant questionnaire upon recruitment and following delivery was completed.
Participants were vaccinated with dTpa containing ≥2 IU diphtheria toxoid, ≥20 IU tetanus toxoid, 8 μg pertussis toxin (PT), 8 μg filamentous hemagglutinin (FHA), and 2.5 μg pertactin (PRN).
All blood samples were processed at the time of collection and stored at –80°C prior to analysis of samples. At birth, infant cord blood was collected to determine antibody levels to PT, PRN, and FHA using a commercially available IgG-specific enzyme-linked immunosorbent assay (Alpha Diagnostic International, San Antonio, TX). Results were reported in IU/mL using the World Health Organization reference 06/142.
All continuous variables were assessed for normality. All maternal and cord antibody body levels were skewed and were therefore log transformed prior to further analysis. Time between vaccination and birth was computed by subtracting the gestational week of the vaccine from the gestational age at birth. The characteristics of the population and cord blood antibody concentrations were tabulated. Data are presented as means and SD or counts and percentages as appropriate. Differences in characteristics and outcomes between the early and late vaccination group were compared using independent t tests or χ 2 tests and across the control, early, and late group with χ 2 tests or analysis of variance. The association between timing of vaccination and the log-transformed cord antibody levels was determined using linear regression. The residuals of the regression model were assessed for normality. Maternal prevaccination antibody levels differed between groups, therefore this was included in the multivariate adjustment. No effect of mode of birth on antibody levels was observed so mode of birth was not included in the final model. Cord antibody levels were plotted against time between vaccination and birth and the correlation was determined using Pearson correlation coefficient. A P value <.05 (2-tailed) was regarded as statistically significant. All analyses were performed using statistical software (Stata, Release 12.1; StataCorp LP, College Station, TX).
Materials and Methods
This prospective study evaluated maternal and newborn pertussis-specific antibody levels prior to and following diphtheria, tetanus, and acellular pertussis (dTpa) immunization. Pregnant women at a tertiary obstetric hospital were recruited from April through September 2014. Inclusion criteria were healthy pregnant women with a singleton pregnancy between 28-36+6 weeks who consented to receive dTpa. Women unimmunized for pertussis during their current pregnancy served as controls. Exclusion criteria included women already vaccinated with dTpa in the current pregnancy; immunosuppressed women, such as those with HIV infection; and women considered at high risk for preterm delivery. Women were approached during the third trimester and invited to participate in the study. If they agreed to vaccination then they were allocated to either the early or late group according to their gestation at the time of vaccination. Women vaccinated between 28-32+6 weeks’ gestation comprised the early vaccination group and women vaccinated between 33-36+6 weeks’ gestation comprised the late vaccination group. Women who declined the vaccination but were willing to participate in the study comprised the control group. A prevaccination maternal venous sample was taken from all participants and infant cord blood was collected at delivery.
Prior to the study a sample size calculation based on the effect sizes and associated variances reported by Healy et al was undertaken. We estimated that a minimum sample of 42 patients per group would be required to be able to demonstrate at least a 10% difference in antibody levels between early and late vaccination groups, with 80% power at the 5% significance level. The study of Healy et al, however, did not account for maternal prevaccination levels.
Monash Health Human Research Ethics Committee approved the study and all participants provided informed, signed consent. Demographic data were collected from the women’s medical records and a participant questionnaire upon recruitment and following delivery was completed.
Participants were vaccinated with dTpa containing ≥2 IU diphtheria toxoid, ≥20 IU tetanus toxoid, 8 μg pertussis toxin (PT), 8 μg filamentous hemagglutinin (FHA), and 2.5 μg pertactin (PRN).
All blood samples were processed at the time of collection and stored at –80°C prior to analysis of samples. At birth, infant cord blood was collected to determine antibody levels to PT, PRN, and FHA using a commercially available IgG-specific enzyme-linked immunosorbent assay (Alpha Diagnostic International, San Antonio, TX). Results were reported in IU/mL using the World Health Organization reference 06/142.
All continuous variables were assessed for normality. All maternal and cord antibody body levels were skewed and were therefore log transformed prior to further analysis. Time between vaccination and birth was computed by subtracting the gestational week of the vaccine from the gestational age at birth. The characteristics of the population and cord blood antibody concentrations were tabulated. Data are presented as means and SD or counts and percentages as appropriate. Differences in characteristics and outcomes between the early and late vaccination group were compared using independent t tests or χ 2 tests and across the control, early, and late group with χ 2 tests or analysis of variance. The association between timing of vaccination and the log-transformed cord antibody levels was determined using linear regression. The residuals of the regression model were assessed for normality. Maternal prevaccination antibody levels differed between groups, therefore this was included in the multivariate adjustment. No effect of mode of birth on antibody levels was observed so mode of birth was not included in the final model. Cord antibody levels were plotted against time between vaccination and birth and the correlation was determined using Pearson correlation coefficient. A P value <.05 (2-tailed) was regarded as statistically significant. All analyses were performed using statistical software (Stata, Release 12.1; StataCorp LP, College Station, TX).
Results
A total of 115 women (53 between 28-32+6 weeks’ and 62 between 33-36+6 weeks’ gestation) were vaccinated with dTpa. There were 39 nonvaccinated control subjects. A total of 82 paired maternal and infant cord blood samples were available from dTpa-immunized mothers, and 27 unimmunized mothers for analysis ( Figure 1 ).
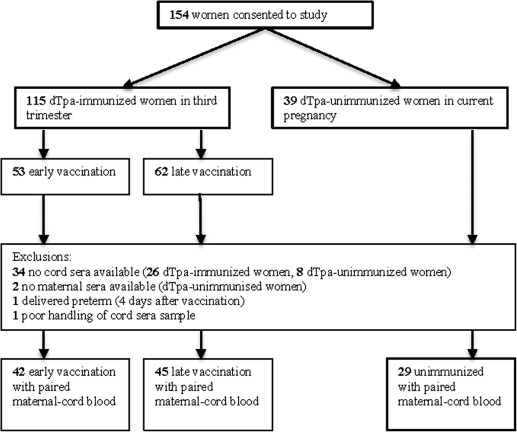
The demographic and clinical characteristics of the study population are presented in Table 1 , along with the pertussis antibodies in maternal serum prior to vaccination.



