Materials and Methods
This study is a randomized controlled trial that took place at a single perinatal center from February 2015 to February 2016. The Institutional Review Board of Saint Louis University School of Medicine approved this protocol. This trial is registered with clinicaltrials.gov ( NCT02606643 ).
We included patients with a Bishop score of ≤6 with a singleton, cephalic gestation who had been admitted for induction of labor. Exclusion criteria included any medical conditions that precluded vaginal delivery, chorioamnionitis, vaginal bleeding, intrauterine fetal death, a low-lying placenta, previous cervical surgery, or latex allergy. Rupture of membranes was not an exclusion criterion, although it was up to each provider to decide whether to place a catheter in this situation.
After the patient was admitted, the house staff, under attending physician supervision, selected the initial method of cervical ripening. The most common noncatheter method of cervical ripening was prostaglandin E 1 . It was at the discretion of the clinician when to place the transcervical catheter. A transcervical catheter was not placed if the Bishop score was >6. Once the decision was made to use a transcervical catheter and the patient was not in labor, she was approached for study participation and randomization. Initially, group allocation occurred by a random pull from a pile of opaque, sealed envelopes that contained a group selection. After the recruitment of the first 23 patients, randomization was achieved by a random number generator to assign group allocation. After informed consent was obtained from each participant, an 18F 30-mL Foley bulb was placed digitally by the house staff, with or without a stylette, until it was presumed to be above the internal os. The balloon was filled with 50 mL of saline solution, because this is the standard at our institution. Patients who were assigned randomly to tension had the catheter taped with applied tension to the inner thigh. Tension was replaced approximately every 30 minutes, as needed. Patients who were assigned randomly to no tension did not have any tension applied to their catheter. After randomization, clinicians were not blinded to the allocated group. If the catheter was not expelled after 12 hours, the balloon was deflated, and the catheter was removed. On catheter insertion, an oxytocin infusion was started at 1–2 mU/min and increased by 1–2 mU/min every 20–30 minutes to a maximum of 6 mU/min while the catheter was in place. After catheter removal, the remainder of the patient’s labor course was managed according to provider preference. All patients had continuous fetal heart rate and uterine activity monitoring.
Our primary outcome was the time of catheter placement to the time of delivery. Secondary outcomes included the time of placement to catheter expulsion, vaginal delivery within 24 hours, cesarean delivery rates, pain scores while the catheter was in place, the amount of oxytocin infused while the catheter was in place, and chorioamnionitis. For this study, chorioamnionitis was defined strictly as a combination of maternal temperature at >100.3°F, maternal pulse at >100 beats per minute, and fetal tachycardia (>160 beats per minute), before or during the time of maternal fever, with no other identifiable source for fever or tachycardia. Pain scores were assessed throughout labor; a pain was scored based on a scale of 1–10, 10 being the worst pain the patient ever experienced.
The sample size calculation was based on a mean time to delivery of 24.0±9.6 hours in the no tension group, an hypothesized 20% reduction to 19.2±9.6 hours in the tension group, an alpha level of .05, and a power of 80%. These assumptions required a sample size of 63 patients per group, for a total of 126 patients. Mean time to delivery and standard deviation estimations were based on previously published data.
Chi-square tests and Fisher’s Exact tests were used to assess differences in categoric variables between the 2 groups. The Kolmogorov-Smirnov goodness of fit test was used to assess for normality. Independent Student t tests and the Kolmogorov-Smirnov statistic (for nonparametric data) were used for continuous variables, depending on the normality of the distributions. Survival curves with the use of lifetables were constructed from time of catheter insertion to delivery and from time of catheter insertion to catheter expulsion and were compared with the use of the Wilcoxon (Gehan) Breslow statistic. A probability value of <.05 initially was set to denote statistical significance. Data were analyzed by “intent-to-treat” and “per protocol.” All analyses were performed with SPSS software for Windows (version 21.0; SPSS Inc, Chicago, IL). A planned interim analysis was performed for safety and efficacy at the midpoint of patient recruitment. The primary and secondary outcomes were evaluated at the interim analysis using a probability value of <.005, based on the O’Brien-Fleming group sequential boundaries. To account for the interim analysis, probability values of <.048 were considered statistically significant in the final analysis.
Results
One hundred seventy-two patients were approached to participate in the study. Thirty-two patients were excluded from the analysis, including 28 patients who declined ( Figure 1 ). Randomization occurred after successful catheter placement. Deviations from study protocol occurred in both groups. One patient with a Bishop score of 9 was assigned randomly to the tension group. In the no tension group, 1 patient had a previous loop electrosurgical excision procedure, and 2 patients had a latex allergy that prompted silicone balloon placement (Cook Medical Inc, Bloomington, IN). Sixty-seven patients were analyzed in the tension group, and 73 patients were analyzed in the no tension group ( Figure 1 ). All patient data were analyzed by intent-to-treat. A “per protocol” analysis was also performed that yielded similar results. The planned interim analysis was performed on 66 patients. No patient safety issues were identified. The survival curve for the primary outcome of time from catheter insertion to delivery had a probability value of .35; the survival curve for the secondary outcome of time from catheter insertion to expulsion had a probability value of <.01. Because neither outcome had a probability value less than the critical probability value of .005 for the interim analysis, the decision was made to continue the study to determine whether complete data collection resulted in the finding of significant differences between groups for study outcomes.
Baseline maternal characteristics are described in Table 1 . There were no significant differences between the tension and no tension groups. Although not statistically significant, there were trends toward a greater number of patients with preeclampsia in a previous pregnancy and a lower body mass index in the current pregnancy to be assigned randomly to the tension group, whereas there was a trend toward a greater number of patients with suspected fetal growth restriction who were assigned randomly to the no tension group.
| Characteristic | Tension (n=67) | No tension (n=73) | P value |
|---|---|---|---|
| Maternal age, y a | 26 (23, 30) | 26 (22, 30) | .39 |
| Race, n (%) | |||
| Caucasian | 20 (29.9) | 17 (23.3) | .674 |
| African American | 46 (68.7) | 55 (75.3) | |
| Other | 1 (1.5) | 1 (1.4) | |
| Insurance, n (%) | |||
| Government | 57 (85.1) | 58 (79.5) | .386 |
| Private | 10 (14.9) | 15 (20.5) | |
| Parity, n (%) | |||
| Nulliparity | 38 (56.7) | 35 (47.9) | .299 |
| Multiparity | 29 (43.3) | 38 (52.1) | |
| Gestational age at enrollment, wk a | 39.1 (37.6, 40.4) | 39.0 (37.6, 40.1) | .473 |
| Medical/obstetrics history, n (%) | |||
| Asthma | 9 (13.4) | 13 (17.8) | .477 |
| Chronic hypertension | 9 (13.4) | 12 (16.4) | .619 |
| Diabetes mellitus | |||
| Gestational | 6 (9.0) | 7 (9.6) | .897 |
| Pregestational | 0 | 4 (5.5) | .121 |
| Preeclampsia | 18 (26.9) | 10 (13.7) | .052 |
| Miscarriages | 8 (11.9) | 11 (15.1) | .589 |
| Previous cesarean deliveries, n (%) | |||
| 0 | 55 (82.1) | 58 (79.5) | .444 |
| 1 | 11 (16.4) | 11 (15.1) | |
| 2 | 1 (1.5) | 4 (5.5) | |
| Body mass index at admission, kg/m 2 a | 33.1 (28.9, 41.6) | 37.0 (29.0, 42.2) | .098 |
| Reason for induction, n (%) | |||
| Diabetes mellitus | 2 (3.0) | 5 (6.8) | .392 |
| Elective | 10 (14.9) | 10 (13.7) | |
| Fetal growth restriction | 1 (1.5) | 7 (9.6) | |
| Hypertension | 25 (37.3) | 26 (35.6) | |
| Oligohydramnios | 5 (7.5) | 5 (6.8) | |
| Other | 11 (16.4) | 11 (15.1) | |
| Postdates | 13 (19.4) | 9 (12.3) | |
| Ripening agents given before transcervical catheter insertion, n (%) | 45 (67.1) | 56 (76.7) | .208 |
| Bishop score at time of transcervical catheter placement a | 4 (3, 5) | 3.5 (3, 5) | .753 |
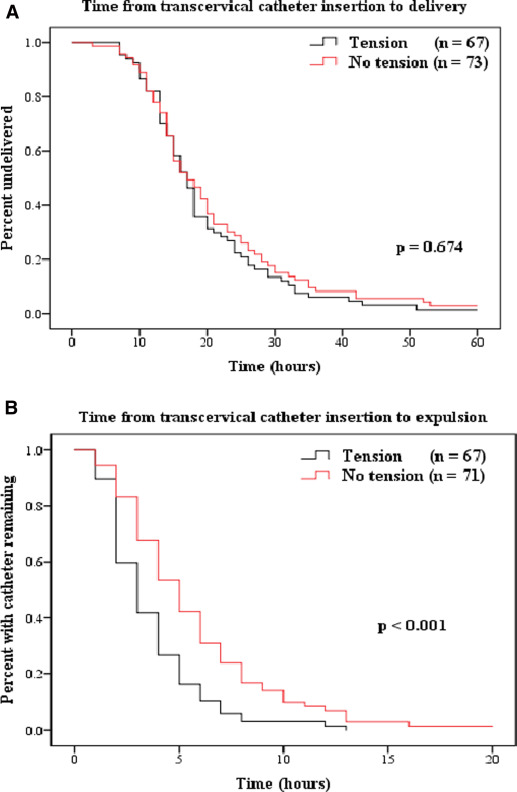
Because this was a pragmatic trial, we included patients with a previous cervical ripening agent. Nonetheless, all patients at randomization were required to have a Bishop score of ≤6 and no labor. The use of a cervical ripening agent before the insertion of a transcervical catheter was similar between groups ( P =.208). Table 2 shows that there were no differences as to the type and amount of agent used. Cervical dilation, effacement, and station were similar between groups in this subset. Table 3 describes the primary and secondary outcomes that were related to the duration of the transcervical catheter and final mode of delivery outcomes. The primary and secondary outcome data were not distributed normally. Therefore, medians and interquartile ranges are reported and compared. Means and standard deviations for the primary outcome were included in Table 3 to facilitate comparisons to other published research. There was no statistically significant difference in the primary outcome between groups (median time, 16.2 vs 16.9 hours; P =.814). However, the median time from catheter insertion to expulsion was significantly shorter in the tension group (2.6 vs 4.6 hours; P <.001). The survival curves are provided in Figure 2 .
| Characteristic | Tension (n=45) | No tension (n=56) | P value |
|---|---|---|---|
| Dilation at time of ripening, cm a | 1 (0, 1) | 0.5 (0, 1) | .876 |
| Effacement at time of ripening % a | 25 (0, 50) | 25 (0, 50) | 1 |
| Station at time of ripening a | –3 (–3, –3) | –3 (–3, –3) | .999 |
| Ripening agents given before transcervical catheter insertion, n (%) | 45 (67.1) | 56 (76.7) | .208 |
| Misoprostol use, n (%) | 37 (55.2) | 40 (54.8) | .959 |
| Misoprostol amount, μg a | 50 (25, 75) | 50 (25, 75) | 1 |
| Dinoprostone use, n (%) | 8 (11.9) | 11 (15.1) | .589 |
| Dinoprostone amount, mg a | 10 (10, 10) | 10 (10, 10) | 1 |
| Oxytocin use, n (%) | 13 (19.4) | 18 (24.7) | .725 |
| Oxytocin amount, mU a | 1133 (515, 5216) | 1511 (943, 2796) | .723 |
| Other (laminaria), n (%) | 0 | 1 (1.4) | 1 |
| Outcomes | Tension (n=67) | No tension (n=73) | P value |
|---|---|---|---|
| Delivery | |||
| Median time to delivery, hr a | 16.2 (12.6, 23.5) | 16.9 (12.4, 25.6) | .814 |
| Mean time to delivery, hr b | 19.1 ± 10.4 | 23.1 ± 26.2 | .814 |
| Median time to vaginal delivery, hr a | 14.8 (12.1, 20.5) | 14.6 (11.3, 20.9) | .99 |
| Delivery within 24 hrs, n (%) | 52 (77.6) | 52 (71.2) | .388 |
| Vaginal delivery within 24 hr, n/N (%) | 41/52 (78.8) | 37/52 (71.2) | .365 |
| Vaginal deliveries after 24 hr, n/N (%) | 9/15 (60.0) | 9/21 (42.9) | .31 |
| Mode of delivery, n (%) | |||
| Vaginal | 50 (74.6) | 46 (63.0) | .139 |
| Cesarean delivery | 17 (25.4) | 27 (37.0) | |
| Primary indication for cesarean delivery | |||
| Arrest of dilation | 10 (58.8) | 12 (44.4) | .309 |
| Nonreassuring fetal heart tracing | 7 (41.2) | 12 (44.4) | |
| Other | 0 | 3 (11.1) | |
| Cervical dilation at cesarean delivery, cm a | 4.8 (4.1, 6.4) | 5.0 (4.3, 7.5) | .995 |
| Transcervical catheter duration | |||
| Time to catheter expulsion, hr a | 2.6 (1.3, 4.3) | 4.6 (2.3, 7.0) | .001 |
| Cervical dilation at time of catheter expulsion, cm a | 3.5 (3, 4) | 3.5 (3, 4) | 1 |
| Highest pain score while catheter was in place a | 7 (5, 10) | 7 (5, 10) | 1 |
| Total oxytocin while catheter in place, mU a | 290 (118, 900) | 782 (313, 1584) | .004 |
a Data are shown as median (interquartile range)
The cesarean delivery rates and indications were similar between groups. In the no tension group, there were 3 “other” indications ( Table 3 ) that included mid-labor fetal presentation change, unsuccessful induction, and patient desire to discontinue induction and proceed with repeat cesarean delivery. Table 4 shows other clinical outcomes that were related to labor. Table 5 shows that there were no differences in neonatal outcomes between groups.
| Characteristic | Tension (n=67) | No tension (n=73) | P value |
|---|---|---|---|
| Rupture of membranes before delivery, n (%) | |||
| Before catheter placement | 3 (4.5) | 1 (1.4) | .692 |
| During catheter placement | 1 (1.5) | 1 (1.4) | |
| After catheter placement before removal | 3 (4.5) | 3 (4.1) | |
| After catheter removal | 58 (86.6) | 63 (86.3) | |
| At delivery | 2 (3.0) | 5 (6.8) | |
| Duration of rupture of membranes, hr a | 7.3 (4.2, 12.6) | 6.0 (3.4, 11.5) | .236 |
| Epidural use, n (%) | 66 (98.5) | 65 (89.0) | .035 |
| Meconium | 7 (10.4) | 8 (11.0) | .922 |
| Group B streptococcus colonized, n (%) | 16 (23.9) | 23 (31.5) | .603 |
| Clinical chorioamnionitis, n (%) | 7 (10.4) | 8 (11.0) | .922 |
| Placental disease, n (%) | |||
| Acute chorioamnionitis | 19 (28.4) | 20 (27.4) | .899 |
| Funisitis | 4 (6.0) | 6 (8.2) | .747 |
| Unremarkable | 29 (43.3) | 36 (49.3) | .475 |
| Missing | 19 (28.4) | 18 (24.7) | .620 |
| Characteristic | Tension (n=67) | No tension (n=73) | P value |
|---|---|---|---|
| Infant birthweight, gm a | 3090 (2805, 3520) | 3095 (2695, 3540) | .485 |
| Low birthweight <2500 g, n (%) | 9 (13.4) | 8 (11.0) | .654 |
| Macrosomia >4000 g, n (%) | 3 (4.5) | 6 (8.2) | .367 |
| Gender: female, n (%) | 37 (55.2) | 37 (50.7) | .591 |
| 1-Minute Apgar score a | 8 (6, 8) | 8 (7, 9) | .999 |
| 5-Minute Apgar score a | 9 (9, 9) | 9 (9, 9) | 1 |
| Umbilical cord gasses a | |||
| Arterial pH | 7.24 (7.20, 7.30) | 7.24 (7.16, 7.29) | .419 |
| Arterial CO 2 , mEq/L | 55.3 (48.8, 65.4) | 55.9 (46.5, 67.6) | .54 |
| Arterial base excess, mEq/L | –5 (–6, –3) | –5 (–7, –3) | .544 |
| Venous pH | 7.31 (7.27, 7.34) | 7.30 (7.25, 7.33) | .702 |
| Venous CO 2 , mEq/L | 44.0 (39.0, 49.0) | 45.0 (40.5, 50.7) | .754 |
| Venous base excess, mEq/L | –5 (–6, –3) | –5 (–7, –3) | .772 |
| Neonatal disposition, n (%) | |||
| Neonatal Intensive Care Unit | 11 (16.4) | 13 (17.8) | .827 |
| Respiratory complications | 9 (13.4) | 12 (16.4) | .619 |
| Intracranial hemorrhage | 1 (1.5) | 0 (0) | .479 |
| Proven sepsis | 1 (1.5) | 1 (1.4) | 1 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


