The History of Oral Contraception
It wasn’t until the early 1900s that inhibition of ovulation was observed to be linked to pregnancy and the corpus luteum. Beginning in 1920, Ludwig Haberlandt, professor of physiology at the University of Innsbruck, Austria, demonstrated that ovarian extracts given orally could prevent fertility in mice. Haberlandt is acknowledged as the first to perform experiments with the aim of producing a method of hormonal contraception; he called it “hormonal sterilization.”
1 In the 1920s, a Viennese gynecologist, Otfried Otto Fellner, conducting experiments in his spare time, and administering ovarian and placental extracts to a variety of animals, also reported hormonal sterilization.
2 By 1931, Haberlandt proposed the administration of hormones for birth control. An extract named Infecundin was produced in collaboration with the Hungarian pharmaceutical company Gideon Richter, but Haberlandt’s early death of a heart attack in 1932, at age forty-seven, brought an end to this effort. Fellner disappeared after the annexation of Austria to Hitler’s Germany.
The concept was annunciated by Haberlandt, but steroid chemistry wasn’t ready. The extraction and isolation of a few milligrams of the sex steroids required starting points measured in gallons of urine or thousands of pounds of organs. Edward Doisy processed 80,000 sow ovaries to produce 12 mg of estradiol.
Russell Marker
The supply problem was solved by a cantankerous iconoclast, Russell E. Marker, who completed his thesis, but not the course work, for his Ph.D. The following story is derived from Marker’s own words, in an autobiographical article and from a two-hour interview for the oral history archives of the Chemical Heritage Foundation in Philadelphia.
3,4Marker, born in 1902 in a one-room log cabin on a farm near Hagerstown, Maryland, received his bachelor’s degree in organic chemistry and his master’s degree in colloidal chemistry from the University of Maryland. Although he had completed his work for a Ph.D., his supervisor, Morris S. Kharasch, announced that Marker still lacked some required chemistry courses. Considering the courses a waste of time, Marker said, “The hell with it,” and abruptly left.
After leaving the University of Maryland, Marker worked first in the laboratory of the Naval Powder Factory, then with the Ethyl Gasoline Corporation, where in 1926 he developed the system of octane rating of gasoline. Frank Whitmore, dean of Pennsylvania State College, now Pennsylvania State University, visited Marker at Ethyl. Impressed with his work, Whitmore said, “If you’re ever looking for a job, let me know.”
From 1927 to 1934, Marker worked at the Rockefeller Institute, publishing a total of 32 papers on configuration and optical rotation as a method of identifying compounds. He became interested in steroid chemistry, but he was told to continue with his work in optical technology. Instead, Marker called Dean Whitmore at Penn State.
In September 1935, Marker moved to Penn State at a reduced salary, from $4,400 per year at Rockefeller to $1,800, but with the freedom to pursue any field of research. His work was supported mainly by research grants from the Parke-Davis pharmaceutical company. At that time, it required the ovaries from 2,500 pregnant pigs to produce 1 mg of progesterone. Marker decided to pursue the goal of an abundant and inexpensive supply of progesterone, and for several years he concentrated on urine from pregnant animals. Then in 1939, Marker devised the method, called the Marker degradation, to convert a sapogenin molecule into a progestin.
Marker was convinced that the solution to the problem of obtaining large quantities of steroid hormones was to find plants in the family that includes the lily, the agave, and the yam that contained sufficient amounts of diosgenin, a plant steroid, a sapogenin, that could be used as a starting point for steroid hormone production. He discovered that a species of Trillium, known locally as Beth’s root, was collected in North Carolina for the preparation of Lydia Pinkham’s Compound, popular at the time to relieve menstrual discomfort. A principal ingredient in Beth’s root was diosgenin, but the rhizome was too small to provide sufficient amounts for commercial production.
Marker’s search for an appropriate plant took him to California, Arizona, and Texas. Spending his summer vacations in the Southwest and Mexico collecting sapogenin-containing plants, Marker’s laboratory analyzed more than a hundred thousand pounds of over four hundred different species of plants. Marker discovered that the roots of the Dioscorea plant (a wild yam) were the richest source of sapogenins.
On a visit to Texas A & M University, Marker found a picture of a large Dioscorea (Dioscorea mexicana) in a book that he just happened to pick up and browse through while spending the night at the home of a retired botanist who was helping him collect diosgenincontaining plants. After returning to Pennsylvania, he traveled by train for 3 days to search for this Dioscorea in Mexico.
Marker first went to Mexico City in November 1941, but his effort was blocked by the lack of a plant-collecting permit from the Mexican government. He returned in January 1942, and the American Embassy arranged for a Mexican botanist who had a collecting permit to accompany Marker to Veracruz. Marker rented a truck with a driver, and when the botanist arrived at Marker’s hotel, he was accompanied by his girlfriend and her mother, who served as the girl’s chaperone. Marker was forced to take the entire group. They covered 80 miles the first day, staying overnight in Puebla. The next day, the drive to Tehuacan was a shorter trip, but the botanist insisted on a 2-day stay devoted to his own collection of specimens. Then next morning, the botanist refused to go any further, claiming that the natives had discovered Marker was American and wanted nothing to do with him. They turned around, managed to overcome a breakdown of the truck near Puebla, and made it back to Mexico City 5 days after starting, with nothing to show for the trip.
The next day, a Monday morning, Marker reported to the American Embassy and was advised to leave Mexico. It was just after Pearl Harbor and Mexico was being courted by Germany. The Embassy was concerned for the safety of Americans traveling in Mexico. Instead of returning home, Marker took an overnight bus to Puebla, arriving after midnight, and boarded a second bus that already held pigs and chickens in addition to a few passengers. He arrived in Orizaba the next morning, and fortunately there was a small hotel next to the bus terminal. Marker remembered that the botany book in which he first read a description of D. mexicana indicated that the plant, a wild yam vine that grows up trees in the mountains of southern Mexico, could be found along a stream that crossed the road between Orizaba and Cordoba. He climbed aboard the local bus to Cordoba, which he stopped and disembarked when the bus drove through a large stream crossing the road about ten miles after leaving Orizaba. He found a small country store next to the road, owned by an Indian named Alberto Moreno.
Moreno did not speak English; Marker did not speak Spanish. But somehow, Marker conveyed his desire to obtain the Dioscorea that was known locally as “cabeza de negro,” black tubers. Moreno in turn somehow made Marker understand that he should return the next morning. And there in the store, the next morning, were two plants, each in a bag that Moreno placed on the roof of the next bus back to Orizaba. Each tuber was nine to twelve inches long and consisted of white material like a turnip; it was used by local Mexicans as soap and as a poison to catch fish. When Marker got off the bus in Orizaba, both bags were missing. A policeman was there, but it became apparent he was there to collect a fee for the return of the bags. Marker gave him what he had, a ten-dollar bill, but that only retrieved one bag, which he managed to smuggle back to Pennsylvania.
Marker used only a portion of the plant to isolate diosgenin. In February 1942, he took the remainder to the Parke-Davis chemists in Detroit. Demonstrating his process for obtaining diosgenin, Marker convinced the director of research, Oliver Kamm, that he was on to something, a source for raw material that could provide for the commercial production of hormones. Unfortunately, they could not convince the president of Parke-Davis, nor could Marker convince anyone at several other companies.
Unable to obtain support from the pharmaceutical industry, Marker, drew on half of his life savings and returned to Mexico in October 1942. He arranged with Albert Moreno to collect the roots of the Mexican yam. Marker paid Mexican medical students to collect the yams. The students were arrested when farmers reported that their yams were being stolen, but not before Marker had enough to prepare a syrup.
Back in the U.S. with his syrup, Marker arranged to work in the New York laboratory of a friend, Norman Applezweig, an organic chemist involved in steroid research, in return for one-third of whatever progesterone his syrup could yield.
5 He isolated diosgenin and synthesized 3 kg of progesterone, the largest lot of progesterone ever produced. United States
pharmaceutical companies still refused to back Marker, and even his university refused, despite Marker’s urging, to patent the process.
Before Marker left Mexico, he looked through the yellow pages in a Mexico City telephone directory and found something he recognized, a company called “Laboratorios Hormona,” owned by a lawyer who was a Hungarian immigrant, Emeric Somlo, and a German immigrant who had both a medical degree and a Ph.D. in chemistry, Frederick A. Lehman.
… when the phone rang. A distant voice asked in barely comprehensible Spanish if
he {Frederick Lehman} spoke English.
“Yes, of course.”
“I found your company’s name in the telephone book, since I recognized two words,
‘Laboratories’ and ‘Hormones.’ I have something you may be interested in: a cheap
source for progesterone.”
“Who are you?”
“I am Marker, a steroid chemist.”
6
Visiting the company, Marker met Lehman, the minority owner of Laboratorios Hormona, who had the good sense to see where this was going. From his reading of the literature, he knew who Marker was; he knew the value of steroids; and he was a businessman. Lehman called his partner who was visiting New York and convinced him to return as soon as possible. The three men agreed to form a Mexican company for the production of hormones, and Marker returned to the U.S., leaving behind a list of equipment and chemicals to be ordered.
Marker returned to Mexico in spring 1943 to collect plants and to check on progress at Laboratorios Hormona. He just happened to mention to Lehmann that he had 2 kg of progesterone. As soon as Marker returned to Pennsylvania, he received a phone call from Somlo who said that if Marker still had those 2 kg of progesterone he sure would like to see it; could he meet him in New York? Over dinner at the Waldorf-Astoria, Somlo offered Marker 40% of their new company in exchange for the progesterone, with a share in future profits. Marker arranged for a friend to deliver the progesterone to Somlo in New York. Somlo had a small company in New York called Chemical Specialties, and the progesterone used in the first studies leading to oral contraception were obtained from this Syntex subsidiary.
In December 1943, Marker resigned from Pennsylvania State College and went to Mexico where he collected the roots of D. mexicana—ten tons worth! Marker chopped them up with a machete, and left the pieces to dry in the sun across from Moreno’s store in a small structure for drying coffee. It took 2 months of work in an old pottery shed in Mexico City to prepare several pounds of progesterone, worth $160,000, with the help of several young women who had little education and spoke no English.
Somlo suggested calling their new company Synthesis, but Marker insisted on some link to Mexico, and the three partners formed Syntex (from synthesis and Mexico), incorporated in March 1944. Marker moved into a new four-room laboratory, and over the next year, produced over 30 kg of progesterone and 10 kg of dehydroepiandrosterone. The price of progesterone fell from $200 to $50 a g.
During this time, Marker received expenses, but he was not given his share of the profits or the 40% share of stock due to him. In March 1945, Somlo claimed there were no profits, but then admitted that the profits had been paid to the two partners in Mexico as salaries. Failing to reach a settlement, Marker left Syntex in May 1945, took some of his young female workers with him, and started a new company in Texcoco, called Botanica-Mex. He changed to Dioscorea barbasco, which gave a greater yield of diosgenin, and the price of progesterone dropped to $10 a g, and later to $5.
After I broke up with Lehmann and Somlo, I chose a place east of Mexico City (Texcoco), where labor and water were plentiful. I there repeated my simple procedure of converting diosgenin into progesterone. My workers were happy but one day they came to me and said, “We all live on this dry-lake bed, and we come from very far away. If you want us to go on working for you, we need bicycles.” “Sure,” said Marker, “I’ll buy them for you, and you will pay them back from your salary.” The workers, happy with this offer, and the image of a white man with promise, celebrated drunkenly one evening. Late at night they went to a nearby quarry where a great effigy of the Aztec rain god was still attached by its back to the bedrock (It wasn’t moved to the museum until 1964). They then began chiseling my name over Tláloc’s right eyebrow, but were interrupted by angry villagers and had to run away after having carved only the first two letters.
3The volcanic stone monolith of Tláloc the rain god was carved in a horizontal position sometime in the period of 400 B.C. to 200 A.D. On April 16, 1964, the unfinished statue was detached and transported on a day’s journey to Mexico City, and placed in a vertical position at the road entrance to the Museo Nacional de Antropologia, an imposing 168 tons, 23 feet high. The initials “MA” can be easily discerned at the right edge of the headdress; Marker’s workers obviously intended to place his full name across the entire width. The evening arrival of the rain god was greeted by a crowd of 25,000 people. Despite the fact that it was the dry season, a record rainfall fell on the day the statue arrived!
7Marker’s new company was allegedly harassed, legally and physically, by Syntex, and in 1946 it was sold to Gideon Richter, which moved it to Mexico City and renamed it Hormosynth. Eventually it came under the ownership of Organon of Holland, which still uses it under the name of Quimica Esteroides. By the 1960s, several pharmaceutical companies were benefiting from the root-gathering operations in Mexico, closely regulated by the Mexican government that imposed annual quotas, about 43 thousand tons, to balance harvesting with the new annual growth. Mexican yams provided the starting material for the manufacture of oral contraceptives for about 15 years, giving way to other sources, such as soya beans, methods for total synthesis, or microbial fermentation.
8In 1949, Marker retired to Pennsylvania to devote the rest of his life to traveling, and in 1959 he began an association with a French silversmith who had emigrated to Mexico City, and then with his son, Pedro Leites. After 1970, Marker turned to collecting paintings by Mexican artists. The artwork and the replicas of antique works in silver were successful businesses that allowed him, in the 1980s, to endow scientific lectureships at both Pennsylvania State University and the University of Maryland. In 1970, the Mexican government honored Marker and awarded him the Order of the Aztec Eagle; staying true to his irascible nature, he declined. In 1984, Pennsylvania State University established the annual Marker Lectures in Science and, in 1987, the Russell and Mildred Marker Professorship of Natural Product Chemistry. In 1987, Marker was granted an honorary doctorate in science from the University of Maryland, the degree he failed to receive in 1926.
In 1990, Marker was planning on a quiet visit to Mexico to present a plaque made in his honor by Pennsylvania State University to Adolfina Moreno, the daughter of Alberto, the owner of the small country store whom Marker met in 1942. Mexican scientists and pharmaceutical people learned of the visit, and that summer a chartered busload of fifty people retraced Marker’s trip from Mexico City to Orizaba.
6 Marker rode in a car with Frederico Lehman’s son, Pedro, who had become a distinguished chemist. Meeting in an auditorium at the University of Veracruz, Marker was honored by speeches and an engraved silver tray. After lunch at a local brewery, nearly 100 people made their way to the bridge over the Mezcala River. Marker entered the living quarters behind the store now owned by Adolfina. She tearfully thanked him and pointed to a nearby photo, her marriage picture from fifty years ago, with Marker in the wedding group. At the age of 92, Russell Earl Marker died in Wernersville, Pennsylvania, in 1995, from complications after a broken hip.
The Race for Cortisone
When Marker left Syntex, he took his know-how with him. Fortunately for Syntex, there still was no patent on his discoveries. George Rosenkranz left his native Hungary to study chemistry in Switzerland under the renowned steroid chemist Leopold Ruzicka, who was awarded the 1939 Nobel Prize in Chemistry.
9 On the day Pearl Harbor was attacked, Rosenkranz was in Havana waiting for a ship to Ecuador where the chair in organic chemistry awaited him at the University of Quito. The ship never showed. Rebuffed by the national university in Cuba, Rosenkranz took a job with a local pharmaceutical firm for $25 per week. Because of his success in developing new products, he was soon earning $1,000 per month and directing a research program with Ph.D. candidates from the university. He was also learning how to be a business man; for example, he organized the shark-fishing business in Cuba in order to produce vitamin A from shark liver oil.
10The Rosenkranz laboratory was following Marker’s published techniques and making small amounts of progesterone and testosterone from sarsaparilla roots imported from Mexico. The news of this activity led to an invitation from Syntex to take over for Marker, with an option of buying 15% of Syntex stock, although the company was currently practically bankrupt.
Rosenkranz’s task was complicated by Marker’s secretiveness. He found reagents labeled with code words; Marker’s workers identified solvents by their weight and smell. Rosenkranz gave up on reconstructing Marker’s process, and worked out his own commercial manufacture of progesterone and testosterone from Mexican yams, and soon Syntex was making large profits providing the sex hormones as raw material to other pharmaceutical companies. Rozenkranz now had a large active laboratory that attracted a young chemist, Carl Djerassi. These men knew each other, meeting and interacting with each other at the Laurentian Hormone Conference, the annual meeting organized and directed by Gregory Pincus.
The Djerassi family lived in Bulgaria for hundreds of years after escaping Spain during the Inquisition.
11 Carl Djerassi, the son of a Bulgarian physician, was born in Vienna, as was his physician mother. Djerassi, age 16, and his mother fled the Nazi Anschluss and emigrated to the U.S. in 1939. A Jewish refugee aid organization placed Djerassi with a family in Newark, New Jersey. With a scholarship to Tarkio College in Tarkio, Missouri, he was exposed to Middle America, where he earned his way giving talks to church groups about Bulgaria and Europe. His education was further supported by another scholarship from Kenyon College in Ohio, where he pursued chemistry. After a year working for CIBA, Djerassi received his graduate degree from the University of Wisconsin. Returning to CIBA and being somewhat unhappy, he responded to an invitation to visit Syntex. Rosenkranz proposed that Djerassi head a research group to concentrate on the synthesis of cortisone. Djerassi’s initial reaction was that “the location of Syntex in the chemical desert of Mexico made the offer seem ludicrous.”
12 But the 26-year-old Djerassi, impressed by Rosenkranz and excited by the challenge to develop a method to synthesize cortisone, accepted the position and moved to Mexico City in the fall of 1949.
Earlier in 1949, Philip S. Hench, a Mayo Clinic rheumatologist, showed a movie at a medical meeting documenting crippled arthritic patients before treatment and the same patients active, even dancing, after daily injections with cortisone. Cortisone can be converted to the more active Cortisol (also called hydrocortisone), the major product of the adrenal cortex. Cortisone is produced by hydroxylation, which converts the oxygen attached at the 11 position to a hydroxyl group by adding a hydrogen.
Hench had obtained the very expensive cortisone through a biochemist at the Mayo Foundation, Edward C. Kendall, the discoverer of the thyroid hormone, thyroxine, who had been working with Lewis H. Sarett at Merck & Company to determine the structures of
compounds isolated from extracts of the adrenal cortex and from cattle bile; cortisone was known as Kendall’s Compound E. Hench reported good results in 14 patients; his movie received a standing ovation,
13 and in 1950, Hench and Kendall were awarded the Nobel Prize in Physiology or Medicine. It was recognized that continuing regular treatment would be necessary, and the race was on to develop an easy and cheap method to synthesize cortisone and related drugs.
In Mexico City, Carl Djerassi was using the plant steroid diosgenin from the Mexican yam as the starting point. In 2 years time, Syntex achieved the partial synthesis of cortisone, reported in 1951.
14 The Syntex method never reached commercialization, however, because a more efficient process was developed by the Upjohn Company. Djerassi’s productivity at Syntex, 60 publications, attracted a job offer from Wayne State University.
15 Wanting all along to be in the academic world, Djerassi moved to Detroit in January 1951. Five years later, he took a leave of absence to return to Syntex, now American-owned and a public company. Syntex’s topical corticoid anti-inflammatory products, Synalar and Neosynalar, came from Djerassi’s laboratory. Djerassi maintained his laboratory at Wayne State, and in 1959, when W.S. Johnson at Wisconsin moved to head the chemistry department at Stanford University, Djerassi joined him—a professorial position he held for the next 25 years.
The Upjohn Company and G.D. Searle & Company joined the competition to synthesize cortisone, with Upjohn, the bigger company, devoting over 150 scientists and technicians to the task. Upjohn leadership assigned a symbol to represent the project, a blow torch, making it clear that this was a heated race they wished to win.
16 G.D. Searle was a smaller company, but its participation in this race would cement a long-term relationship with Gregory Pincus.
G.D. Searle was founded in 1888 by Gideon Daniel Searle, a pharmacist in Indiana, to provide elixirs, syrups, and drugs directly to clinicians. Searle’s son, Claude, graduated from Rush Medical College in 1898 and developed a large, successful practice in Sabula, Iowa. In 1909, when his father suffered a stroke, the son returned to Chicago to manage the company, setting up a research department that developed new products. His son, Jack Searle, graduated from the University of Michigan with a degree in pharmacy, and succeeded his father as president of the company in 1936. He recruited Albert L. Raymond from the Rockefeller Institute to serve as director of research, working in new laboratories in Skokie, Illinois. Dramamine, to prevent motion sickness, and Banthine, to treat peptic ulcers, came from these laboratories.
By 1949, Raymond and the G.D. Searle company were supporting steroid research at the Worcester Foundation for Experimental Biology in Massachusetts, and Gregory Pincus, the cofounder of the Worcester Foundation, was a Searle consultant.
17 Pincus and Oscar Hechter had developed a perfusion method, pumping blood, serum, or a serum-like solution through fresh endocrine glands (adrenal glands, testicles, or ovaries) held in a glass apparatus and collecting the perfused fluid. Using the enzymes in the glands, precursors in the perfusing fluid were converted to the final products, hydrocortisone or the sex steroids. This was a method that could be used to produce commercial amounts of cortisone products.
The round-faced, balding, acerbic Oscar Hechter came to the Worcester Foundation in 1944 on a fellowship funded by G.D. Searle. Pincus assigned him the task of perfusing adrenal glands, with the aim of identifying the products of adrenal secretion and the hope of creating a system for commercial production. Five years later, Hechter presented the first positive results at a conference in Detroit in 1949.
18 At that same meeting, Hechter saw Hench’s movie and listened to his results. Hechter returned to the Foundation and urged that his project be given top priority. Pincus’s enduring relationship with Searle that yielded research support and new steroid compounds for almost never-ending testing began in earnest with the race for cortisone and his development of the perfusion system to use animal glands for the synthesis of steroid drugs. The perfusion system was complicated. It required the development of methods to maintain the animal organs, a web of glassware to
infuse and collect appropriate perfusing solutions, and the separation and identification of the steroid products. At the moment of its coveted value in 1946, Pincus chose to sell his rights to Searle for only one dollar, allowing Searle to patent the process.
19 In return, Pincus obtained and tested steroids that could yield products for clinical use.
Responding to Pincus and Hechter’s success, the Searle company constructed rows of perfusion systems in their Skokie plant. Each contained a periodically replaced fresh beef adrenal gland, producing every few hours a large volume of perfused solution. The long-term plan was to engineer a more economical and profitable system. But in the meantime, Searle was able to provide substantial amounts of cortisone to clinical researchers throughout the U.S.
At the same time, Merck ramped up Sarett’s 36-step synthesizing process from bile acids, and by the end of 1950, they were selling cortisone acetate to clinicians for a price that had been reduced from $200 per gram to $35. In Kalamazoo, Michigan, Upjohn chemists were pursuing a method based on the process used to make penicillin, conversion of precursors by microbes to the desired product. The work was headed by Durey H. Peterson, the son of Swedish immigrants. Peterson supported his education by playing semi-professional baseball.
16 Early in his career, he developed nylon surgical suturing material as well as “Toni,” a product for home permanents to create curly hair. Peterson joined Upjohn in 1946 to work on antibiotics, but he almost immediately became part of the race to synthesize cortisone. Peterson believed that lower microorganisms might possess the same enzymes used by adrenal glands to make cortisone, especially the difficult step of introducing an oxygen molecule to the structure. When told this could not be done, Peterson said, “The microorganisms do not know this.”
16Using paper chromatography methods developed by Alejandro Zaffaroni, Peterson and H.C. Murray attacked the problem, beginning in 1949. First they needed a microorganism. This they acquired, a fungus of the
Rhizopus species, by leaving an agar plate on the window sill of the “oldest and dirtiest laboratory at the Upjohn Company.”
16 In one year’s time, the two chemists proved the value of microorganisms in chemical synthesis. Their method used
Rhizopus nigricans to covert progesterone to 11-hydroxyprogesterone, that could in turn be processed into hydrocortisone, also called Cortisol, the major corticosteroid secreted by the adrenal cortex.
By 1955, Upjohn had become the market leader, and Searle shut down its perfusion cells and quit the race. Upjohn’s commercialization of the methods developed by Peterson and Murray led to popular and successful products. But the Searle people had gained valuable experience that would eventually pay off with other synthesized hormones and products.
The Upjohn method used progesterone as the starting point, available in the early 1950s only from Syntex. George Rosenkranz’s laboratory at Syntex was also pursuing the industrial synthesis of cortisone, and in July 1951, Syntex was about to sign a contract with a large chemical firm to begin production. This never happened because of a phone call. Rosenkranz told the story: “I received a phone call from Upjohn asking me whether we would be able to accept an order for ten tons of progesterone at forty-eight cents a gram.”
10 The quantity was unheard of, and Upjohn’s order remained a puzzle until the microfermentation method was published. Rosenkranz accepted the order, and Syntex found itself as the key supplier of progesterone to other companies.
The Synthetic Progestational Drugs, Norethindrone and Norethynodrel
Djerassi and other Syntex chemists turned their attention to the sex steroids. They discovered that the removal of the 19-carbon from yam-derived progesterone increased the
progestational activity of the molecule. The clue for this work came from Maximilian Ehrenstein at the University of Pennsylvania, who reported in 1944 that a potent progestational compound he had produced appeared to be progesterone without its carbon at the 19 position; henceforth the 19-nor family of compounds indicated steroid chemical structures without the carbon atom at the 19 position.
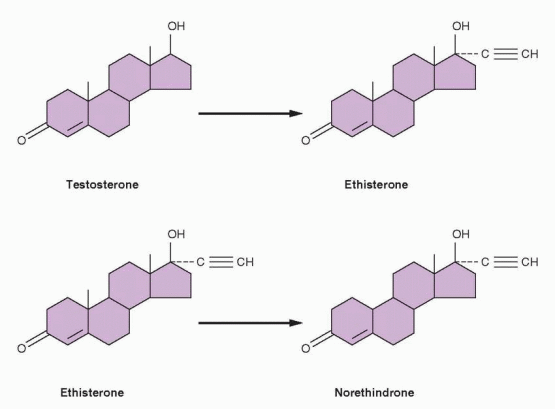
20 Chemists at Schering A.G. in Berlin had produced orally active versions of estradiol and testosterone in 1938, by substituting an acetylene group in the 17-position of the parent compounds. The resulting ethinyl estradiol later became the estrogen component in oral contraceptives. The ethinyl testosterone product was known as ethisterone, marketed in 1941, and the Syntex chemists reasoned that removal of the 19-carbon would increase the progestational potency of this orally active compound.
On October 15, 1951, norethindrone was synthesized at Syntex; the final steps were performed by Luis Miramontes, working on his undergraduate thesis in chemistry under Djerassi’s supervision.
12 The patent application was filed 6 weeks later on November 22, 1951, and the work was presented in April 1952 at the annual meeting of the American Chemical Society and published in 1954.
21 The greater potency of norethindrone, achieved by removing the 19-carbon of ethinyl testosterone, compared with progesterone was demonstrated in monkeys and then 4 women at the National Institutes of Health, reported in 1953, 1956, and 1957.
22,23 and
24 Syntex supplied norethindrone to many investigators, including Gregory Pincus. Edward T. Tyler first reported its clinical use in 1955 for the treatment of menstrual disorders.
25Frank Colton, a chemist at G.D. Searle & Company, filed a patent for norethynodrel, a compound closely related to norethindrone, differing only in the position of the double bond, on August 31, 1953. The Polish-born Colton received his Ph.D. in chemistry from the University of Chicago. From 1949 to 1951, he was a research fellow working with Edward Kendall at the Mayo Foundation on the synthesis of cortisone. Colton joined Searle in 1951, along with Byron Riegel, to develop steroid drugs, succeeding with Nilevar, the first commercial anabolic agent marketed in 1956 and Aldactone, the anti-aldosterone antihypertensive agent introduced in 1959.
Norethynodrel was the result of a deliberate and planned program to create orally active agents with progestational activity. Later, Colton pointed out that although the Syntex and Searle chemists followed a similar path, they were independently pursuing the trail blazed by previous scientists.
17 Along the way, hundreds of compounds were sent to Pincus at the Worcester Foundation to test for ovulation inhibition in rabbits. Their best drug, norethynodrel, assigned the number SC-4642, was synthesized at Searle in a process that was considered to be significantly different from the Syntex method.
17Djerassi urged legal proceedings for patent infringement, claiming that norethynodrel was converted to Syntex’s compound, norethindrone, by gastric acid, but Parke-Davis, the American company licensing norethindrone, did not want to make waves presumably because Parke-Davis was supplying the antihistamine component of Searle’s best-selling product for motion sickness, Dramamine.
12 Pincus would ultimately choose the Searle compound, norethynodrel for clinical testing as an oral contraceptive, and Syntex, not having marketing capability, licensed norethindrone to other pharmaceutical companies. Norethindrone was tested as a contraceptive by Edward Tyler in Los Angeles and Joseph Goldzieher in San Antonio, Texas, but Parke-Davis chose not to pursue government approval, probably fearing religious reactions. Subsequently, Syntex turned to the Ortho division of Johnson & Johnson. By 1964, Ortho, Parke-Davis, and Syntex (now in California) were marketing oral contraceptives containing norethindrone or its acetate.
The creation of norethindrone and norethynodrel by the chemists was essential in the development of oral contraception because the natural hormone progesterone is relatively impotent given orally, requiring very large doses that even then do not achieve a uniform response. The synthetic progestational agents are very active when administered orally, producing reliable effects with small doses.
A Wall Street entrepreneur, Charles Allen, acquired Syntex in 1956 for $2 million cash and a loan of $2 million to be paid from future profits.
9 Rosenkranz became president and CEO, Alejandro Zaffaroni, an Italian who emigrated from Montevideo, Uruguay, executive vice president. Zaffaroni obtained his Ph.D. in 1949 in biochemistry from the University of Rochester, developing a paper chromatography system that soon became a principal method of studying steroid hormones.
26 Rosenkranz met Zaffaroni at the Laurentian Hormone Conference in 1951. Their aim was to develop a pharmaceutical company on a foundation of research. Carl Djerassi, who had left for an academic position at Wayne State University, was recruited back to the company. Rosenkranz said, “We were the brilliant amateurs with a ‘can do anything’ attitude. We were like stem cells (though then none of us really knew the concept). We could differentiate into anything we desired. Production, finance, sales, marketing—all held no fear for us.”
9In 1961, the company moved to Palo Alto, California, influenced by Djerassi who was teaching at Stanford University. The growth of the company was meteoric, with blockbuster hits like Synalar, a topical corticoidsteroid for the treatment of psoriasis, and Naproxen, a nonsteroid, anti-inflammatory drug. Much of this success was to an innovative philosophy in the pharmaceutical business, “patent and publish.”
9 The Syntex scientists were encouraged to promptly publish their results, gaining the peer recognition that is such a motivating force for basic scientists. In 1994, Roche Holdings acquired Syntex for $5.3 billion.
Djerassi eventually left Syntex to become a full-time professor at Stanford University, and is now a playwright and novelist living in San Francisco. Zaffaroni started his own company in 1968, ALZA (after his own name), dedicated to new methods of drug delivery, such as a skin patch. ALZA was acquired by Johnson & Johnson in 2000.
Gregory Pincus
Gregory Goodwin (Goody) Pincus was born in 1903 in New Jersey, the son of Russian Jewish immigrants who lived on a farm colony founded by a Jewish philanthropic organization.
19 Pincus was the oldest of six children and grew up in a home of intellectual curiosity and energy, but even his family regarded him as a genius.
Pincus graduated from Cornell and went to Harvard to study genetics, joining Hudson Hoagland and B.F. Skinner as graduate students of W.J. Crozier in physiology, receiving degrees in 1927. Crozier’s hero was Jacques Loeb who discovered artificial parthenogenesis working with sea urchin eggs. Most importantly, Loeb was a strong believer in applying science to improve human life. Thus, Crozier, influenced by Loeb, taught Pincus, Hoagland, and Skinner (respectively, in reproductive biology, neurophysiology, and psychology) to apply science to human problems. This was to be the cornerstone of Pincus’s own philosophy.
Hoagland, after a short stay at Harvard, spent a year in Cambridge, England, and then moved to Clark University in Worcester, Massachusetts, to be the chair of biology at the age of 31. Pincus went to England and Germany, and returned to Harvard as an assistant professor of physiology.
Pincus performed pioneering studies of meiotic maturation in mammalian oocytes, in both rabbit and human oocytes. In 1934, Pincus reported the achievement of in vitro fertilization of rabbit eggs, earning him a headline in the New York Times that alluded to Haldane and Huxley. An article in Colliers depicted him as an evil scientist. By 1936, Harvard had cited Pincus’s work as one of the university’s outstanding scientific achievements of all time, but Harvard denied him reappointment in 1937.
At Clark University, Hudson Hoagland was in constant conflict with the president of the university, Wallace W. Atwood, the senior author of a widely used textbook on geography. In 1931, the Department of Biology consisted of one faculty member and his graduate student, and their chair, Hudson Hoagland. Hoagland, upset and angry over Harvard’s refusal to grant reappointment to his friend (suspecting that this was because of anti-Semitism), invited Pincus to join him. Hoagland secured funds for Pincus from philanthropists in New York City, enough for a laboratory and an assistant. This success impressed the two men, especially Hoagland, planting the idea that it would be possible to support research with private money.
Min-Chueh Chang was born in Tai Yuan, China, on October 10, 1908. In 1933, he earned a bachelor’s degree in animal psychology from the Tsing Hua University in Peking, and stayed at the university as a teacher. Chang won a national competition in 1938 that funded study abroad. He chose to study agricultural science at Edinburgh University. After one year, he was pleased to receive an invitation from Arthur Walton to study the physiology of sheep sperm at The University of Cambridge, and he promptly accepted.
Chang received his Ph.D. in animal breeding under the direction of Walton and Sir John Hammond at the University of Cambridge in 1941. It was virtually impossible to leave England during the early years of World War II, and Chang continued to work at the University. In 1944, Chang planned to return to China, but first he wanted to spend a year in the U.S. He wrote three letters to American scientists, and only Pincus answered, offering a fellowship at Clark University. Chang mistakenly assumed that a fellowship in the U.S. was the same as at the University of Cambridge where a Fellow was assured of a lifetime income. The successful recruitment of M-C Chang by Pincus was to pay great dividends. Years later, Chang would direct the testing of new progestins to effectively inhibit ovulation in animals.
Soon Hoagland had put together a group of outstanding scientists, but because of his ongoing antagonism with President Atwood, the group was denied faculty status. Working in a converted barn, they were totally supported by private funds. By 1943, 12 of Clark’s 60 faculty were in the Department of Biology.
Frustrated by the politics of academia, Hoagland and Pincus (who both enjoyed stepping outside of convention) had a vision of a private research center devoted to their philosophy of applied science. Indeed, the establishment of the Worcester Foundation for Experimental Biology, in 1944, can be attributed directly to Hoagland and Pincus, their friendship for each other, and their confidence, enthusiasm, ambition, and drive. It was their spirit that turned many members of Worcester society into financial supporters of biologic science. Hoagland and Pincus accomplished what they set out to do. They created and sustained a vibrant, productive scientific institution in which it was a pleasure to work.
Although named the Worcester Foundation for Experimental Biology, the Foundation was located in the summer of 1945 across Lake Quinsigamond in a house on an estate in Shrewsbury. From 1945 to the death of Pincus in 1967, the staff grew from 12 to 350 (scientists and support people), 36 of whom were independently funded and 45 were postdoctoral fellows. The annual budget grew from $100,000 to $4.5 million. One hundred acres of adjoining land were acquired, and the campus grew to 11 buildings. In its first 25 years, approximately 3,000 scientific papers were published.
But in those early years, Pincus was the animal keeper, Mrs. Hoagland the bookkeeper, M-C. Chang was the night watchman, and Hoagland mowed the lawn. During the years of World War II, Pincus and Hoagland combined their interests in hormones and neurophysiology to focus on stress and fatigue in industry and the military.
Katharine Dexter McCormick (1875-1967) was a trained biologist, an early suffragist, and rich, inheriting millions from her mother and a McCormick fortune from her husband.
She was the second woman to graduate from the Massachusetts Institute of Technology, socially conscious, and a generous contributor to family planning efforts. Her intervention with money, energy, incisive thinking, and persistent dedication was instrumental in the development of oral contraception. In 1904, she married Stanley McCormick, the son of Cyrus McCormick, the founder of International Harvester. Katharine’s husband suffered from schizophrenia, and she established the Neuroendocrine Research Foundation at Harvard to study schizophrenia. This brought her together with Hoagland, who told her of the work being done by Chang and Pincus who were seeking orally active progestins to inhibit ovulation.
Pincus attributed his interest in contraception to his growing appreciation for the world’s population problem, and to a 1951 visit in New York with Margaret Sanger, at that time president of the Planned Parenthood Federation of America. Sanger promised a small amount of money and expressed hope that a method of contraception could be derived from the laboratory work being done by Pincus and Chang. During this meeting, Pincus formulated his thoughts derived from his mammalian research. He envisioned a progestational agent in pill form as a contraceptive, acting like progesterone in pregnancy.
Margaret Sanger brought Pincus and Katharine McCormick together. On June 7, 1953, when 78-year-old Katharine met with 50-year-old Pincus at the Worcester Foundation and wrote him a check for $20,000; she promised him another $20,000. A week later, Pincus and Hoagland met with Katharine and her lawyer. They signed a contract outlining the goals, the decision-making process, and the timetable. Pincus received a second check for $20,000, and Katharine agreed to fund laboratory improvements, which ended up as the completion of a new building in 1955.
Katharine’s contract with the Worcester Foundation stipulated that Pincus would provide written reports every 2 weeks. In addition, Pincus and John Rock, the Boston gynecologist performing the initial oral contraceptive studies in his patients, made many visits to Katharine’s home office on Beacon Street across the street from the Harvard Club. Katharine had Sara De Laney, her secretary, take careful notes in shorthand, and at the next visit De Laney read the transcribed notes to her boss so that she would be prepared. Periodically the principals met at the Worcester Foundation. Katharine peppered Pincus, Chang, and Rock with questions and urged them to stop wasting time. She found Pincus “imaginative and inspirational; Rock was informative and very realistic about medical work.” By now everyone was familiar with Katharine’s methods. She had earned their respect, and detailed reports on laboratory results, clinical planning, and budgets were immediately forthcoming. Time and time again, Katharine proved that she handled delays poorly, but she approached each meeting with an eagerness that slowly but surely was rewarded with success after 7 years and an expenditure of about $2 million of Katharine’s money.
In her last years, Katharine continued to support the work of Pincus and Chang. When testing the hundreds of compounds that yielded the progestational agents in birth control pills, Chang observed that some of them prevented implantation of fertilized eggs in rabbits.-From 1962 to 1966, Chang and Pincus were pursuing a drug that could prevent pregnancy with one administration, a day or two after sexual intercourse. With Pincus’s death, this project was abandoned. It is not certain whether Chang and Pincus coined the phrase the “morning after” pill, but it is accurate to state that the concept came from Chang.
When Pincus and Chang began their studies, the focus was on inhibition of ovulation, first by progesterone, and then by synthetic progestins. Chang’s contribution was easy to overlook. Chang worked away in his laboratory, and it was Pincus who was highly visible, raising the money and providing direction. Chang started by repeating the experiments reported by Makepeace in 1927, documenting that progesterone could inhibit ovulation. The first experiment was on April 25, 1951, and Chang quickly moved to testing the newly synthesized progestins from Searle and Syntex.
By December 1953, three synthetic progestins were selected as the most potent and effective in inhibiting ovulation: norethindrone from Syntex, and Searle’s norethynodrel and norethandrolone. The animal and human results were published in
Science in 1956.
27,28 In 1957, these three compounds were approved for the treatment of menstrual disorders with the trade names of Norlutin, Enovid, and Nilevar, respectively.
It was Pincus who made the decision to involve a physician because he knew human experiments would be necessary. John Rock, chief of gynecology and obstetrics at Harvard, met Pincus at a scientific conference and discovered their mutual interest in reproductive physiology. Rock and his colleagues pursued Pincus’s work. Using oocytes from oophorectomies, they reported in vitro fertilization in 1944, the first demonstration of fertilization of human oocytes in vitro. Rock was interested in the work with progestational agents, not for contraception, however, but because he hoped the female sex steroids could be used to overcome infertility.
In their first collaborative study, Pincus and Rock administered oral progesterone, 300 mg/day. Pincus suggested a 20-day regimen beginning on day 5 of the menstrual cycle.
29 He had two reasons for choosing this regimen: (1) it covered the time period during which nearly all, if not all, ovulations occurred, and (2) the withdrawal menstrual bleed at the conclusion of the treatment period would mimic the timing of a normal menstrual cycle and reassure the women that they were not pregnant. The first study involved 33 volunteers who ovulated regularly but had been infertile for 2 years. The women were treated for 1 to 3 cycles after a baseline control month. About 85% of the treated women did not ovulate during the treatment cycles. Not one became pregnant during treatment, pleasing Pincus who all along was aiming for contraception, and four became pregnant after treatment, pleasing Rock who initially was motivated by his pursuit of the “rebound” phenomenon for the treatment of infertility.
Sanger and McCormick needed some convincing that Rock’s Catholicism would not be a handicap, but they were eventually won over because of his stature. Rock was a physician who literally transformed his personal values in response to his recognition of the problems secondary to uncontrolled reproduction. With the help of Luigi Mastroianni, the first administration of synthetic progestins to women was to Rock’s patients in 1954. Of the first 50 patients to receive 10-40 mg of synthetic progestin (a dose extrapolated from the animal data) for 20 days each month, all failed to ovulate during treatment (causing Pincus to begin referring to the medication as “the pill”), and 7 of the 50 became pregnant after discontinuing the medication, again pleasing Rock, who all along was motivated to treat his infertile patients.
In 1956, with Celso-Ramon Garcia and Edris Rice-Wray, working in Puerto Rico, the first human trial was performed. The initial progestin products were contaminated with about 1% mestranol. In the amounts being used, this added up to 50-500 μg of mestranol, a sufficient amount of estrogen to inhibit ovulation by itself. When efforts to provide a more pure progestin lowered the estrogen content and yielded breakthrough bleeding, it was decided to retain the estrogen for cycle control, thus establishing the principle of the combined estrogen-progestin oral contraceptive. Early clinical trials were also conducted by J.W. Goldzieher in San Antonio and E.T. Tyler in Los Angeles.
Pincus, a longtime consultant to Searle, picked the Searle compound for extended use, and with great effort, convinced Searle that the commercial potential of an oral contraceptive warranted the risk of possible negative public reaction. Pincus also convinced Rock, and together they pushed the U.S. Food and Drug Administration for acceptance of oral contraception. In 1957, Enovid was approved for the treatment of miscarriages and menstrual disorders, and on June 23, 1960, for contraception. Neither Pincus nor the Worcester Foundation got rich on the pill; alas, there was no royalty agreement.
The pill did bring Pincus fame and travel. There is no doubt that he was very much aware of the accomplishment and its implications. As he traveled and lectured in 1957, he said:
“How a few precious facts obscurely come to in the laboratory may resonate into the lives of men everywhere, bring order to disorder, hope to the hopeless, life to the dying. That this is the magic and mystery of our time is sometimes grasped and often missed, but to expound it is inevitable.”
30Pincus was the perfect person to bring oral contraception into the public world, at a time when contraception was a private, suppressed subject. Difficult projects require people like Pincus. A scientific entrepreneur, he could plow through distractions. He could be hard and aggressive with his staff. He could remain focused. He hated to lose, even in meaningless games with his children. Yet he combined a gracious, warm, charming manner with his competitive hardness. He was filled with the kind of self-confidence that permits an individual to forge ahead, to translate vision into reality. Pincus died in 1967 (as did Katharine McCormick at the age of 92), of myeloid metaplasia. Rock died in 1984, at the age of 94. Chang died in 1991 at the age of 82, and was buried in Shrewsbury, near his laboratory and close to the grave of Pincus.
Pincus wrote his book,
The Control of Fertility, in 1964-1965, because “a break came in the apparent dam to publication on reproductive physiology and particularly its subdivisions concerned with reproductive behavior, conception, and contraception.”
30
“We have conferred and lectured in many countries of the world, seen at first hand the research needs and possibilities in almost every European, Asiatic, Central, and South American country. We have faced the hard fact of overpopulation in country after country, learned of the bleak demographic future, assessed the prospects for the practice of efficient fertility control. This has been a saddening and a heartening experience; saddening because of the sight of continuing poverty and misery, heartening because of the dedicated colleagues and workers seeking to overcome the handicap of excess fertility and to promote healthy reproductive function. Among these we have made many friends, found devoted students.”
30
Syntex, a wholesale drug supplier, was without marketing experience or organization. By the time Syntex had secured arrangements with Ortho for a sales outlet, Searle marketed Enovid in 1960 (150 μg mestranol and 9.85 mg norethynodrel). Ortho-Novum, using norethindrone from Syntex, appeared in 1962. Wyeth Laboratories introduced norgestrel in 1968, the same year in which the first reliable prospective studies were initiated. It was not until the late 1970s that a dose-response relationship between problems and the amount of steroids in the pill was appreciated. Health care providers and patients, over the years, have been confronted by a bewildering array of different products and formulations. The solution to this clinical dilemma is relatively straightforward, the theme of this chapter: use the lowest doses that provide effective contraception.
Pharmacology of Steroid Contraception
The Estrogen Component of Combination Oral Contraceptives
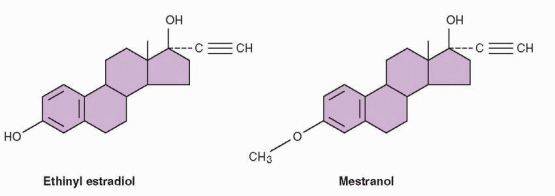
Estradiol is the most potent natural estrogen and is the major estrogen secreted by the ovaries. The major obstacle to the use of sex steroids for contraception was reduced activity of the compounds when given orally. A major breakthrough occurred in 1938 when
it was discovered that the addition of an ethinyl group at the 17 position increased oral activity. Ethinyl estradiol is a very potent oral estrogen and is the form of estrogen in most oral contraceptives. Another estrogen, present in older products, was the 3-methyl ether of ethinyl estradiol, mestranol.
Mestranol and ethinyl estradiol are different from natural estradiol and must be regarded as pharmacologic drugs. Animal studies suggested that mestranol is weaker than ethinyl estradiol, because mestranol must first be converted to ethinyl estradiol in the body. Indeed, mestranol will not bind to the cellular estrogen receptor. Therefore, unconjugated ethinyl estradiol is the active estrogen in the blood for both mestranol and ethinyl estradiol. In the human body, differences in potency between ethinyl estradiol and mestranol do not appear to be significant, certainly not as great as indicated by assays in rodents. This is now a minor point because, with the exception of a contraceptives with estradiol valerate or estradiol, all of the low-dose oral contraceptives contain ethinyl estradiol.
 |
The metabolism of ethinyl estradiol (particularly as reflected in blood levels) varies significantly from individual to individual, and from one population to another.
31,32 There is even a range of variability at different sampling times within the same individual. Therefore, it is not surprising that the same dose can cause side effects in one individual and not in another.
Estradiol valerate is an esterified form of estradiol, allowing oral administration with significant potency. The ester is rapidly hydrolyzed to estradiol. Combinations of several progestins with estradiol valerate have demonstrated good contraceptive efficacy.
The estrogen content (dosage) of the pill is of major clinical importance. Thrombosis is one of the most serious side effects of the pill, playing a key role in the increased risk of death (in the past with high doses) from a variety of circulatory problems. This side effect is related to estrogen, and it is dose related. Therefore, the dose of estrogen is a critical issue in selecting an oral contraceptive.
The Progestin Component of Combination Oral Contraceptives
The discovery of ethinyl substitution and oral potency led (at the end of the 1930s) to the preparation of ethisterone, an orally active derivative of testosterone. In 1951, it was demonstrated that removal of the 19-carbon from ethisterone to form norethindrone did not destroy the oral activity, and most importantly, it changed the major hormonal effect from that of an androgen to that of a progestational agent. Accordingly, the progestational derivatives of testosterone were designated as 19-nortestosterones (denoting the
missing 19-carbon). The androgenic properties of these compounds, however, were not totally eliminated, and minimal anabolic and androgenic potential remains within the structure.
 |
The “impurity” of 19-nortestosterone, i.e., androgenic as well as progestational effects, was further complicated in the past by a belief that they were metabolized within the body to estrogenic compounds. This question was restudied, and it was argued that the previous evidence for metabolism to estrogenic compounds was due to an artifact in the laboratory analysis. More recent studies indicate that norethindrone can be converted to ethinyl estradiol; however, the rate of this conversion is so low that insignificant amounts of ethinyl estradiol can be found in the circulation or urine following the administration of the commonly used doses of norethindrone.
33 Any estrogenic activity, therefore, would have to be due to a direct effect. In animal and human studies, however, only norethindrone, norethynodrel, and ethynodiol diacetate have estrogen activity, and it is very slight due to weak binding to the estrogen receptor.
34 Clinically, androgenic and estrogenic activities of the progestin component, therefore, are insignificant due to the low dosage in the current oral contraceptives. As with the estrogen component, serious side effects have been related to the high doses of progestins used in old formulations, and routine use of oral contraceptives should now be limited to the low-dose products.
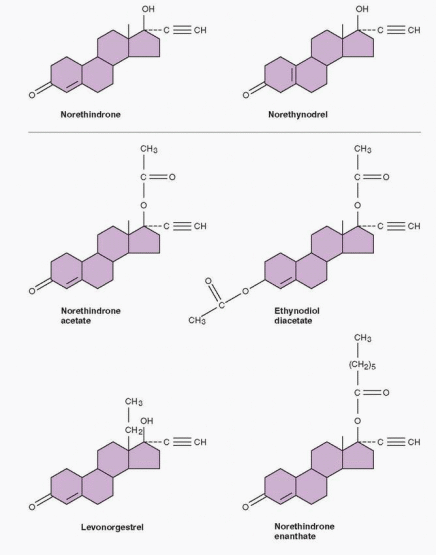
The norethindrone family contains the following 19-nortestosterone progestins: norethindrone, norethynodrel, norethindrone acetate, ethynodiol diacetate, lynestrenol, norgestrel, norgestimate, desogestrel, and gestodene.
Most of the progestins closely related to norethindrone are converted to the parent compound. Thus the activity of norethynodrel, norethindrone acetate, ethynodiol diacetate, and lynestrenol is due to rapid conversion to norethindrone.
Norgestrel is a racemic equal mixture of the dextrorotatory enantiomer and the levorotatory enantiomer. These enantiomers are mirror images of each other and rotate the plane of polarized light in opposite directions. The dextrorotatory form is known as d-norgestrel, and the levorotatory form is l-norgestrel (known as levonorgestrel). Levonorgestrel is the active isomer of norgestrel.
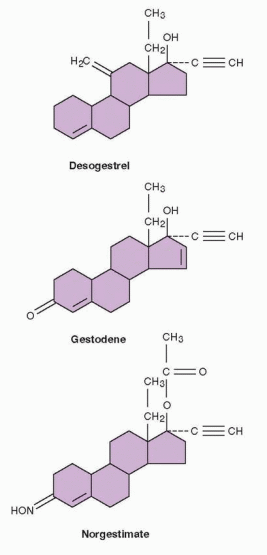 |
Desogestrel undergoes two metabolic steps before the progestational activity is expressed in its active metabolite, 3-keto-desogestrel, known as etonogestrel. This metabolite differs from levonorgestrel only by a methylene group in the 11 position. Gestodene differs from levonorgestrel by the presence of a double bond between carbons 15 and 16; thus, it is Δ-15 gestodene. It is metabolized into many derivatives with progestational activity, but not levonorgestrel. Several metabolites have the potential to contribute to the activity of norgestimate. Although norgestimate is a “new” progestin, epidemiologists included it in the oral contraceptive second-generation family because its activity was believed to be largely due to levonorgestrel and levonorgestrel metabolites.
35,36 Almost all of the biologic effects are attributed to the 17-deacetylated metabolite, now known as norelgestromin; the levonorgestrel metabolites are tightly bound to sex hormone-binding globulin (unlike norelgestromin) severely limiting their biologic activity.
37Definitions Used In Epidemiologic Studies
Low-Dose Oral Contraceptives — Products containing less than 50 μg ethinyl estradiol
First-Generation Oral Contraceptives — Products containing 50 μg or more of ethinyl estradiol
Second-Generation Oral Contraceptives — Products containing levonorgestrel, norgestimate, and other members of the norethindrone family and 20, 30, or 35 μg ethinyl estradiol
Third-Generation Oral Contraceptives — Products containing desogestrel or gestodene with 20, 25, or 30 μg ethinyl estradiol
Fourth-Generation Oral Contraceptives — Products containing drospirenone, dienogest, or nomegestrol acetate.
Probably the greatest influence on the effort that yielded the new progestins was the belief throughout the 1980s that androgenic metabolic effects were important, especially in terms of cardiovascular disease. Cardiovascular side effects are now known to be due to a doserelated stimulation of thrombosis by estrogen and not secondary to metabolic effects such as lipid changes. In the search to find compounds that minimize androgenic effects, however, the pharmaceutical companies succeeded.
The new progestins include desogestrel, gestodene, and norgestimate.
38 In regard to cycle control (breakthrough bleeding and amenorrhea), the new formulations are comparable with previous low-dose products. All progestins derived from 19-nortestosterone have the potential to decrease glucose tolerance and increase insulin resistance. The impact on carbohydrate metabolism of the previous low-dose formulations was very minimal, and the impact of the new progestins is negligible. Most changes are not statistically significant, and when they are, they are so subtle as to be of no clinical significance. The decreased androgenicity of the progestins in the newer products is reflected in increased sex hormone-binding globulin and decreased free testosterone concentrations to a greater degree than the older oral contraceptives. This difference could be of greater clinical value in the treatment of acne and hirsutism, but comparative clinical studies have indicated similar effects for all oral contraceptives.
39The new progestins, because of their reduced androgenicity, predictably do not adversely affect the cholesterol-lipoprotein profile. Indeed, the estrogen-progestin balance of combined oral contraceptives containing one of the new progestins even promote favorable lipid changes. Thus, the new formulations have the potential to offer protection against cardiovascular disease, an important consideration as we enter an era of women using oral contraceptives for longer durations and later in life. But one must be cautious regarding the clinical significance of subtle changes, and it is unlikely there will be a major impact.
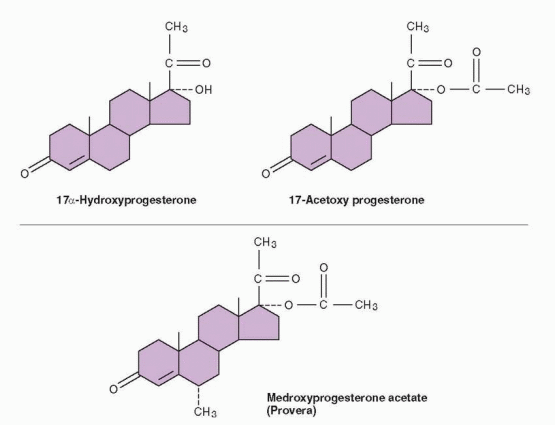 |
A second group of progestins became available for use when it was discovered that acetylation of the 17-hydroxy group of 17-hydroxyprogesterone produced an orally active but weak progestin. An addition at the 6 position is necessary to give sufficient progestational strength for human use, probably by inhibiting metabolism. Derivatives of progesterone with substituents at the 17 and 6 positions include the widely used medroxyprogesterone acetate. Chlormadinone and cyproterone acetate are progesterone derivatives with a 17a-acetoxy group, marketed in a combination with ethinyl estradiol.
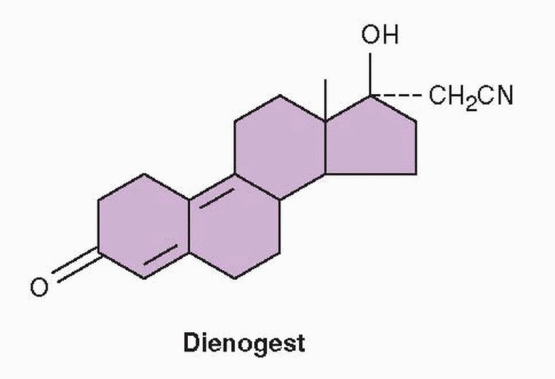
Dienogest is a 19-nortestosterone that has a cyanomethyl group instead of an ethinyl group in the 17 position and an additional double bond, combining the properties of both the 19-nortestosterone family and the derivatives of progesterone.
40 It exerts antiandrogenic activity and is used in a 2-mg dose combined with 30 μg ethinyl estradiol or estradiol valerate as an oral contraceptive.
41,42 and
43 The combination oral contraceptive with estradiol valerate (Qlaira, Natazia) uniquely consists of 4 phases, with the estradiol valerate dose decreasing from 3 mg to 1 mg over 26 days, and dienogest provided in a dose of 2 mg on days 3-7 and 3 mg on days 8-24. This phasic administration of estradiol valerate and dienogest provides ovulation inhibition and a bleeding profile comparable to that of a standard, low-dose, monophasic oral contraceptive.
42,43 and
44
 |
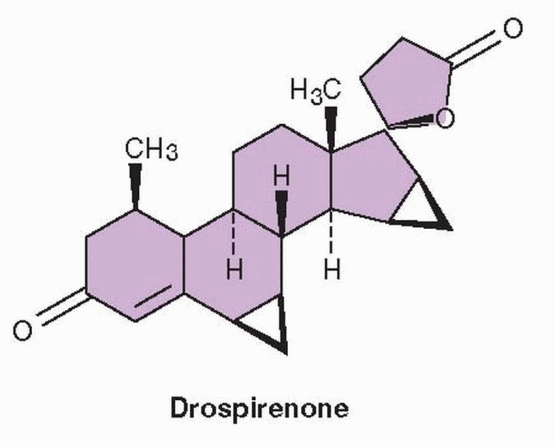
Drospirenone is a progestin that is an analogue of spironolactone. Its biochemical profile is very similar to progesterone, including a high affinity for the mineralocorticoid receptor that produces an antimineralocorticoid effect.
45,46 Contraceptive efficacy equal to that of other formulations is achieved in the combination of 3.0 mg drospirenone and 30 μg ethinyl estradiol (Yasmin). Because drospirenone is spironolactone-like with antiandrogenic and antimineralocorticoid activity, caution is recommended in regard to serum potassium levels, avoiding its use in women with abnormal renal, adrenal, or hepatic function. However, hyperkalemia and its complications have not been a clinical problem encountered with the use of a drospirenone-containing contraceptive in the general population.
47
 |
It has been suggested that the oral contraceptive that contains drospirenone is effective for treating premenstrual syndrome/premenstrual dysphoric disorder (PMDD). In an open-label, 1-year study of 326 women, Yasmin was associated with a significant reduction in scores assessing negative affect, water retention, and increased appetite during the
premenstrual and menstrual phases of their cycles.
48 A similar effect was observed in new users and in those who switched from other oral contraceptives. We have learned over the last decade that treatments for premenstrual syndrome must be studied in comparison with a placebo because of the powerful placebo response associated with this disorder. In a double-blind, placebo-controlled randomized trial, 82 women with established diagnoses of PMDD were assessed using the Calendar of Premenstrual Experiences scale.
49 A statistically significant reduction associated with Yasmin treatment was achieved in only one category, that measuring acne, appetite, and food cravings. The overall result was essentially not significant.
In a multicenter 2-year study in Europe of 900 women, Yasmin was compared to Marvelon (the same dose of ethinyl estradiol and 150 μg desogestrel).
50 Marvelon was associated with a small increase in body weight after the fifth cycle; the average body weight associated with Yasmin remained throughout the 2 years below the baseline level at the beginning of the study, but increased to a level above the baseline at the end of the study. The early weight loss amounted to only 1% of body weight and may reflect diuretic action. This study also observed a small reduction in premenstrual symptoms with Yasmin. The evidence, therefore, for a beneficial impact on PMDD with Yasmin is minimal.
The 24-day version of this oral contraceptive, Yaz (3 mg drospirenone and 20 μg ethinyl estradiol), demonstrated in a 3-month multicenter, double-blind, randomized trial symptomatic improvement in 450 women with PMDD.
51 The magnitude of the treatment response compared with placebo amounted to a need to treat 8 women to achieve at least a 50% reduction in symptom severity in a single patient.
These results suggest that modest beneficial results in treating PMDD may be achieved with any oral estrogen-progestin contraceptive administered in an extended regimen. We would expect similar results with the daily, continuous administration of any oral contraceptive.Nomegestrol acetate (NomAc), derived from progesterone with the elimination of the 19 carbon, is used in Europe in postmenopausal hormone therapy and recently combined with estradiol for contraception. The first 24 pills of a package contain 2.5 mg NomAc and 1.5 mg estradiol, and the last four are placebos in an extended regimen typical of newer oral contraceptives. NomAc has potent inhibitory effects on gonadotropin secretion, and no androgenic activity (in fact, it is somewhat antiandrogenic).
52 Unlike drospirenone, NomAc has no glucocorticoid or mineralocorticoid activity.
53 Its relative lack of endometrial effects has been associated with an increase in irregular bleeding in some clinical trials.
Different Formulations
The multiphasic preparation alters the dosage of both the estrogen and progestin components periodically throughout the pill-taking schedule. The aim of these new formulations is to alter steroid levels in an effort to achieve lesser metabolic effects and minimize the occurrence of breakthrough bleeding and amenorrhea, while maintaining efficacy. However, metabolic studies with the multiphasic preparations indicate no differences or very slight improvements over the metabolic effects of low-dose monophasic products.
An estrophasic approach (Estrostep) combines a continuous low dose of a progestin with a low, but gradually increasing dose of estrogen.
54 This approach minimizes estrogen exposure at the beginning of the cycle, yielding a low rate of side effects such as nausea. The increasing estrogen results in a marked increase in sex hormone-binding globulin that produces a very low androgenic state by reducing the bioavailability of circulating free androgens, and this formulation is very effective for treating acne.
55 56
Extended Regimens
Two clinical concerns prompted the development of an oral contraceptive regimen with a reduction in the pill-free interval: (1) breakthrough bleeding, and (2) ovarian activity during pill taking that could lead to ovulation and failure of contraception. Extending the active pill cycle by several days is aimed at decreasing breakthrough bleeding and spotting and reducing the length of withdrawal bleeding without compromising efficacy or safety, and perhaps increasing contraceptive protection by a greater suppression of ovarian activity. This strategy has produced several new 24-day products: Loestrin 24 Fe (1 mg norethindrone acetate/ethinyl estradiol 20 μg with 4 iron-containing placebo pills), Yaz (3 mg drospirenone/ethinyl estradiol 20 μg), and Minesse (60 μg gestodene/ethinyl estradiol 15 μg).
The traditional combination oral contraceptive pill, consisting of estrogen and progestin components, is given daily for 3 of every 4 weeks, for a total of 21 days. Despite multiple contraceptive actions, there has been concern that the current lower-dose products allow follicular development in some individuals, especially in those who metabolize and clear steroid hormones rapidly.
57 Even with greater follicular activity with the lowest-dose oral contraceptives; however, ovulation is still effectively prevented in most women.
58 Nevertheless, recognition of follicular growth emerging during the standard pill-free interval with oral contraceptives and awareness that ovarian activity is greater with the lowestdose estrogen formulations, along with the problem of breakthrough bleeding, provided the motivation to shorten the pill-free interval.
A move to low doses of estrogen in combined oral contraceptives has been fueled by a desire to minimize estrogen-linked, serious cardiovascular side effects. Breakthrough bleeding rates are higher with the lower-dose (20 μg ethinyl estradiol) oral contraceptives, although not dramatically.
59,60 and
61 Breakthrough bleeding is higher in women who smoke and in smokers who use formulations with 20 μg ethinyl estradiol.
62 Breakthrough bleeding gives rise to fears and concerns; it is aggravating, and even embarrassing. These are reasons why although breakthrough bleeding during oral contraceptive use is considered a
minor side effect, it can have a
major consequence: interruption of adherence to therapy resulting in unwanted pregnancies. A nationwide survey identified irregular bleeding as the primary reason for discontinuation of oral contraception.
63 It is important to emphasize that there is no evidence that the onset of bleeding is associated with decreased efficacy, no matter what oral contraceptive formulation is used, even the lower-dose products. Indeed, in a careful study, breakthrough bleeding did not indicate decreases in the contraceptive blood levels of the estrogen and progestin components.
64The most frequently encountered breakthrough bleeding occurs in the first few months of use as the endometrium adjusts to the pharmacologic impact of the oral contraceptive. The incidence is greatest in the first 3 months, ranging from 10 to 30% in the first month to less than 10% in the third. However, the differences among the various 21-day formulations containing 20 μg ethinyl estradiol are of minimal clinical significance. For this reason, the new approach evolved, increasing the number of days with active drug treatment to 24.
Ovarian follicles begin to grow during the 7-day pill-free interval in traditional regimens of oral contraceptives because FSH levels begin to rise after 4 pill-free days.
65 These follicles can reach impressive size; ovarian follicles more than 10 mm in diameter have the potential to continue growth as a dominant follicle.
66 The successful conversion to a dominant follicle marks the “selection” of a follicle destined to ovulate, the process whereby, with rare exception, only a single follicle succeeds.
67,68 However, follicles that reach sizes consistent with dominant follicles, even preovulatory follicles, are not assured of ovulation in women using oral contraceptives in a compliant fashion. Some stop growing, while others continue to grow but fail to ovulate, almost assuredly because of suppression of the LH surge.
69,70 and
71 Nevertheless, evidence indicates that dominant follicles can emerge, secrete preovulatory levels of estradiol, and ovulate, and in this case, contraceptive efficacy requires the other progestational actions.
58,71,72
Clinical Studies with Extended Regimens
The major clinical study with Loestrin 24 Fe was a 6-month, open-label, randomized, activecontrolled study in 32 centers in the U.S.
73 Nine hundred and thirty eight patients were randomized to either the 24-day product or the 21-day formulation (1 mg norethindrone acetate/ethinyl estradiol 20 μg) in a 4:1 ratio that yielded 705 women in the treated group and 181 in the comparison group available for analysis. A strength of this study was that it compared the 24-day product to an identical 21-day formulation. The study was not powered to determine a significant difference in efficacy comparing the two products. The Pearl index for the 24-day schedule was 1.82 (1.78 for subjects 35 years old and younger). The cumulative pregnancy rate for 6 months was 0.9%. These numbers are typical for all oral contraceptives.
The number of days with breakthrough bleeding or spotting was comparable in both groups, but the 24-day group demonstrated a steady decline in breakthrough bleeding/spotting days, so that in cycle 6 the mean number of bleeding days was significantly lower in the 24-day group (0.95 vs. 1.63). Among the women in the 24-day group, those who switched from another oral contraceptive had a lower mean number of bleeding days compared to new users, probably reflecting suppression of endometrial growth by the previous use. Each cycle with the 24-day product demonstrated a shorter duration of withdrawal bleeding (bleeding beginning after the last day of active drug intake), achieving statistical significance in the second cycle. Combining breakthrough bleeding and withdrawal bleeding, the total number of days over the entire 6 treatment cycles with bleeding was significantly less in the 24-day group: 18.6 for the 24-day schedule compared with 23.2 for the 21-day regimen.
A reasonable concern with extending the days of active treatment is the resulting increase in overall hormone exposure. The accumulative dosages over 6 cycles, however, were not notably different. The 24-day schedule totaled 144 mg of norethindrone acetate and 2.88 mg ethinyl estradiol, compared with 126 mg norethindrone acetate and 2.52 mg ethinyl estradiol in the 21-day group. There were no demonstrable differences in adverse events.
A 21-day product has been compared with extending the schedule to 23 days, using 75 μg gestodene/20 μg ethinyl estradiol.
74,75 and
76 The 23-day regimen produced a greater suppression of ovarian activity as measured by lower estradiol levels and less follicular activity as assessed by ultrasonography; however, the incidence of bleeding and spotting days was similar in the two treatment groups. The 23-day regimen was associated with shorter withdrawal bleeding periods compared with the 21-day schedule.
Ovarian activity was compared in a group of women using 60 μg gestodene/ethinyl estradiol 15 μg for 24 days compared to a group using the same product on the standard 21-day regimen.
77 The women using the 21-day regimen experienced greater follicular activity with larger follicles and higher estradiol levels. Breakthrough bleeding was more prevalent with the 24-day schedule; however, the number of treatment cycles in this small study was not large enough to assess bleeding control. A larger study compared the 24-day regimen of 60 μg gestodene/ethinyl estradiol 15 μg with a 21-day regimen using 150 μg desogestrel/ethinyl estradiol 20 μg and reported a greater incidence of breakthrough bleeding with the 24-day regimen; however the length of bleeding was shorter and the intensity of bleeding was reduced.
78 The 1-year overall incidence of breakthrough bleeding with this 24-day, 15 μg ethinyl estradiol product has been reported to be 19.3%.
79In a short study of only three treatment cycles, the 24-day 3 mg drospirenone/ethinyl estradiol 20 μg product was compared to a 21-day regimen of the same formulation.
80 The 24-day schedule was associated with greater follicular suppression and only one ovulation in cycle 3 compared with 4 ovulations in the 21-day regimen when the initial 3 tablets in the third month were substituted with placebos. A study of 12 women using this formulation for either 23 days or 24 days documented greater suppression of FSH, LH, inhibin B, and estradiol during the pill-free interval when compared to women using the same product for 21 days.
65
Clinical Recommendation
The 24-day regimen of low-dose oral contraceptives achieves its goals. Both bleeding and ovarian activity are reduced. Indeed, the two are related.
Diminished ovarian follicular activity is responsible for less fluctuation in endogenous estrogen levels, resulting in a more quiescent and stable endometrium. Extended (and continuous dosing) regimens compared with the standard 21-day regimen are associated with a decrease in menstrual discomfort, headaches, and bloating.
81,82 and
83Another clinical advantage of the 24-day regimen is a reduction in the risk of “escape” follicular activity if a patient inadvertently starts a new package 1 or 2 days late. Randomized studies that extended the pill-free interval by 2 or 3 days observed that women taking a 20 μg ethinyl estradiol formulation had a greater increase in follicular activity compared with women using a 35 μg ethinyl estradiol product.
84,85 Ovarian follicular activity is greater with products containing 20 μg ethinyl estradiol, and the maximal follicular size reached is larger. In one study, a greater proportion of women on a 20 μg product, around 30%, achieved follicular diameters of 15 mm or greater, compared with a 35 μg formulation when the pill-free interval was extended from 7 to 9 days.
85 Once follicles achieve a diameter greater than 10 mm, an increasing percentage go on to ovulate even in the presence of oral contraceptive treatment.
86 The lower-dose formulations produce less suppression of gonadotropin secretion, documented in these studies by higher FSH, LH, and estradiol blood levels among the users of the 20 μg ethinyl estradiol product.
Not only does the 24-day product allow a day or two grace period, but the extended hormone exposure suppresses gonadotropin and follicular activity to a greater degree. Thus, even in patients with good compliance, a greater reduction in follicular activity can reduce the possibility of breakthrough ovulations and contraceptive failure. This would be difficult and expensive to document because it would require a clinical trial with a very large number of patients.
A regimen is available that supplies a package containing the number of pills required for 84 days of daily administration, a reduction of menstrual frequency to 4 per year.
87 This approach includes Seasonale (20 μg ethinyl estradiol and 100 μg levonorgestrel), with 7 placebo pills after 84 active pills, and Seasonique, with 7 pills of 10 μg ethinyl estradiol after 84 active pills. The combination with 7 days of estrogen was a response to the discovery that 84 active pill days are rapidly followed by a rise in FSH with stimulation of follicular growth.
88 A slighter higher rate of breakthrough bleeding with this regimen improves with time, but overall, bleeding is less with Seasonique because of better FSH suppression at the end of the 84-day combination estrogen-progestin period.
89,90Steroid contraception in the currently used low doses has been demonstrated to be very safe for healthy women. Efforts to improve steroid contraception are now focusing on maximizing adherence to treatment and minimizing pregnancies from contraceptive failures. The 24-day regimen offers clinicians and patients the important advantage of reduced bleeding and the possible advantage of greater efficacy because of better compliance as well as a reduction in ovarian activity.
Continuous Dosing
More and more women are embracing the idea that fewer menstrual periods provide a welcome relief from bleeding and menstrual symptoms. Clinicians for years have prescribed unlimited daily oral contraceptives to treat conditions such as endometriosis, bleeding disorders, menstrual seizures, and menstrual migraine headaches, even to avoid bleeding in athletes and busy individuals. Many women do not require the periodic experience of
vaginal bleeding to assure themselves they are not pregnant. And of course, modern society is long past the notion that menstrual bleeding is a cleansing event, a detoxification. It is not necessary for women using oral contraceptives to experience any withdrawal bleeding. Monthly bleeding, periodic bleeding, or no bleeding—this is an individual woman’s choice. Any combination oral contraceptive can be used on a daily basis; even the lowest estrogen dose formulations provide excellent bleeding and side effect profiles in a continuous regimen.
82,91,92 and
93.
Oral contraceptives with the lowest doses of estrogen should be used for overweight and older women, but progestin-only methods are an even better choice.Eliminating a pill-free interval also reduces symptoms associated with menstruation, such as headaches, dysmenorrhea, and bloating.
94,95 and
96 As with the extended regimen, continuous dosing provides greater ovarian suppression, reducing the potential for breakthrough and escape ovulations.
97 A further benefit of continuous use is simplification of the pill-taking schedule with the potential of better compliance and a lower failure rate. Continuous dosing can also be achieved with the contraceptive vaginal ring and the contraceptive patch. The return of ovulation and achievement of pregnancy are not delayed after discontinuation of continuous dosing.
97,98
Generic Products
Generic products are therapeutically equivalent drugs, containing the same amount of active ingredients in the same concentration and dosage form. These products are less expensive, marketed by pharmaceutical companies after patent expiration of the original drug. Generic oral contraceptives need only meet the test of bioequivalence; studies to demonstrate efficacy, side effects, and safety are not required. Meeting the test of bioequivalence requires demonstration in a small number of subjects that absorption, concentrations, and time curves are comparable to the reference drug. The generic product will be approved if the bioequivalence testing ranges from 80 to 125% of the values for the reference drug (differences no greater than 20% lower or 25% higher). Approved, patented products must not vary more than ±10%; therefore a generic oral contraceptive could contain only 70% of the standard dose. In the lowest-dose oral contraceptives, this could impair efficacy. However, we should hasten to point out that there has been no evidence or even anecdotal suggestions that generic oral contraceptives have reduced efficacy or cause more side effects such as breakthrough bleeding. Patients should be forewarned that generic products differ in shape, packaging, and color.
Off Label Uses of Steroid Contraception
Steroid contraception is often used for noncontraceptive purposes. The list is long, including treatment of acne, dysmenorrhea, heavy or irregular vaginal bleeding, menses-associated mood changes, the polycystic ovary syndrome, and endometriosis. For most of the oral contraceptive’s 50-year history, all of these have been “off label” applications, but recently pharmaceutical companies have conducted trials to obtain label “indications” to use in advertising directed to both clinicians and consumers. In order to acquire such an indication, the company simply has to demonstrate that their formulation is better than a placebo at, for example, improving acne or relieving the symptoms of premenstrual dysphoric disorder. Because these trials usually compare a product to a placebo or just to another contraceptive formulation, the studies do not reveal whether the product receiving approval for an “indication” is really better than others. Prices and formularies restrict patient access to the full range of oral contraceptives
99; therefore, clinicians must make judgements by comparing findings from unrelated studies and experience to decide which pill to use for a specific purpose in an individual patient. In most cases, as we will emphasize, it is unlikely that there are major differences among similar products.
Potency
For many years, clinicians, scientists, medical writers, and even the pharmaceutical industry attempted to assign potency values to the various progestational components of oral contraceptives. An accurate assessment, however, has been difficult to achieve for many reasons. Progestins act on numerous target organs (e.g., the uterus, the mammary glands, and the liver), and potency varies depending on the target organ and end point being studied. In the past, animal assays, such as the Clauberg test (endometrial change in the rabbit) and the rat ventral prostate assay, were used to determine progestin potency. Although these were considered acceptable methods at the time, a better understanding of steroid hormone action and metabolism and a recognition that animal and human responses differ have led to greater reliance on data collected from human studies.
Historically, this has been a confusing issue because publications and experts used potency ranking to provide clinical advice. There is absolutely no need for confusion. Oral contraceptive progestin potency is no longer a consideration when it comes to prescribing oral contraception, because the potency of the various progestins has been accounted for by appropriate adjustments of dose. In other words, the biologic effect (in this case the clinical effect) of the various progestational components in current low-dose oral contraceptives is approximately the same. The potency of a drug does not determine its efficacy or safety, only the amount of a drug required to achieve an effect.
Clinical advice based on potency ranking is an artificial exercise that has not stood the test of time. There is no clinical evidence that a particular progestin is better or worse in terms of particular side effects or clinical responses. Thus oral contraceptives should be judged by their clinical characteristics: efficacy, side effects, risks, and benefits. Our progress in lowering the doses of the steroids contained in oral contraceptives has yielded products with little serious differences.
Metabolic Effects of Oral Contraception
Cardiovascular Disease
The Coagulation System
Thrombosis can be divided into two major categories, venous thromboembolism and arterial thrombosis. Venous thromboembolism includes both deep vein thrombosis and pulmonary embolism. Arterial thrombosis includes acute myocardial infarction and stroke.
The goal of the clotting mechanism is to produce thrombin, which converts fibrinogen to a fibrin clot. Thrombin is generated from prothrombin by factor Xa in the presence of factor V, calcium, and phospholipids. The vitamin K-dependent factors include factors
VII, IX, and X, as well as prothrombin. Antithrombin III is one of the body’s natural anticoagulants, an irreversible inhibitor of thrombin and factors IXa, Xa, and XIa. Protein C and protein S are two other major inhibitors of coagulation and are also vitamin K-dependent. Protein C, and its helper, protein S, inhibit clotting at the level of factors V and
VIII. Tissue plasminogen activator (t-PA) is produced by endothelial cells and released when a clot forms. Both t-PA and plasminogen bind to the fibrin clot. The t-PA converts the plasminogen to plasmin which lyses the clot by degrading the fibrin. Deficiencies of antithrombin III, protein C, and protein S are inherited in an autosomal dominant pattern, accounting for 10-15% of familial thrombosis. The most common inherited causes of venous thromboembolism are the factor V Leiden mutation, followed distantly by a mutation in the prothrombin gene.
106
Coagulation Factors: |
|
Factors that favor clotting when increased |
|
|
Fibrinogen |
|
|
Factors VII, VIII, X |
|
Factors that favor clotting when decreased |
|
|
Antithrombin III |
|
|
Protein C |
|
|
Protein S |
Fibrinolysis Factors: |
|
Factors that favor clotting when increased |
|
|
Plasminogen activator inhibitor-1 (PAI-1) |
|
Factors that favor clotting when decreased |
|
|
Antiplasmin |
An inherited resistance to activated protein C has been identified as the basis for about 50% of cases of familial venous thrombosis, due in almost all cases to a gene alteration recognized as the factor V Leiden mutation.
107,108 The factor V Leiden mutation is found in approximately 30% of individuals who develop venous thromboembolism.
109 Activated protein C inhibits coagulation by degrading factors V and VIII. One of the three cleavage sites in factor V is the precise site of a mutation known as the factor V Leiden mutation that substitutes glutamine instead of arginine at this site (adenine for guanine at nucleotide 1691 in the gene).
109 This mutation makes factor V resistant to degradation and activation in fibrinolysis. The entire clotting cascade is then resistant to the actions of the protein C system.
Heterozygotes for the factor V Leiden mutation have an 8-fold increased risk of venous thrombosis, and homozygotes have an 80-fold increased risk, and this risk is further enhanced by oral contraceptive use. The highest prevalence (3-4% of the general population) of factor V Leiden is found in Europeans, and its occurrence in populations not of European descent is very rare, perhaps explaining the low frequency of thromboembolic disease in Africa, Asia, and in Native Americans.
110 The mutation is believed to have arisen in a single ancestor approximately 21,000 to 34,000 years ago.
111 It has been suggested that this was a useful adaptation in heterozygotes in response to life-threatening bleeding, such as with childbirth.
The next most common inherited disorder after the factor V Leiden mutation is a mutation, a guanine to adenine change, in the gene encoding prothrombin.
106,112 The prevalence of this abnormality in the white population is estimated to range from 0.7 to 4%.
113 Oral contraceptive use has been reported to markedly increase the risk of venous thrombosis in carriers of the prothrombin mutation.
114 Perhaps other unidentified disorders make a contribution because an increased risk of venous thrombosis with oral contraceptives has been reported in women with elevated prothrombin levels despite an absence of the prothrombin gene mutation.
115The administration of pharmacologic amounts of estrogen as in high-dose oral contraceptives causes an increase in the production of clotting factors such as factor V, factor VIII, factor X, and fibrinogen.
116 The progestin component also influences the clotting factor responses.
117 Some studies of the blood coagulation system have concluded that both monophasic and multiphasic low-dose oral contraceptives have no significant clinical impact on the coagulation system. Slight increases in thrombin formation are offset by increased fibrinolytic activity.
118,119 Other studies of formulations containing 30 and 35 μg of ethinyl estradiol indicate an increase in clotting factors associated with an increase in platelet activity.
120 However, these changes are essentially all within normal ranges and their clinical significance is unknown.
117Smoking produces a shift to hypercoagulability.
121 A 20 μg estrogen formulation has been reported to have no effect on clotting parameters, even in smokers.
121,122 One study comparing a 20 μg product with a 30 μg product found similar mild procoagulant and fibrinolytic activity, although there was a trend toward increased fibrinolytic activity with the lower dose.
123 These mixed reports make it essential to base clinical decisions on the epidemiologic studies of clinical events.
There is no evidence of an increase in risk of cardiovascular disease among past users of oral contraception.124,125 and 126 In the Nurses’ Health Study, the Royal College of General Practitioners’ Study, and the Oxford Family Planning Association Study, long-term past use of oral contraceptives was not associated with an increase in overall mortality.127,128 and 129
Part of the concern for a possible lingering effect of oral contraceptive use was based on a presumed adverse impact on the atherosclerotic process, which would then be added to the effect of aging and, thus, would be manifested later in life. Instead, the findings have been consistent with the contention that cardiovascular disease due to oral contraception is secondary to acute effects, specifically estrogen-induced thrombosis, a dose-related event.
Venous Thromboembolism
Older epidemiologic evaluations of oral contraceptives and vascular disease indicated that venous thrombosis was an effect of estrogen, limited to current users, with a disappearance of the risk by 3 months after discontinuation.
130,131 Thromboembolic disease was believed to be a consequence of the pharmacologic administration of estrogen, and the level of risk was believed to be related to the estrogen dose.
132,133 and
134 Smoking was documented to produce an additive increase in the risk of arterial thrombosis,
135,136 and
137 but had no effect on the risk of venous thromboembolism.
138,139Is there still a risk of venous thromboembolism with the current low-dose (less than 50 μg ethinyl estradiol) formulations of oral contraceptives? In the first years of oral contraception, the available products, containing 80 and 100 μg ethinyl estradiol (an extremely high dose), were associated with a 6-fold increased risk of venous thrombosis.
140 Because of the increased risks for venous thrombosis, myocardial infarction, and stroke, lower dose formulations (less than 50 μg estrogen) came to dominate the market, and clinicians became more careful in their screening of patients and prescribing of oral contraception. Two forces, therefore, were at work simultaneously to bring greater safety to women utilizing oral contraception: (1) the use of lower dose formulations, and (2) the avoidance of oral contraception by high-risk patients. Because of these two forces, the Puget Sound study in the United States documented a reduction in venous thrombosis risk to 2-fold.
141 The new studies also reflect the importance of these two forces, but they still indicate an increased risk.
The World Health Organization (WHO) Collaborative Study of Cardiovascular Disease and Steroid Hormone Contraception was a hospital-based, case-control study with subjects collected from 21 centers in 17 countries in Africa, Asia, Europe, and Latin America.
142 As part of this study, the risk of
idiopathic venous thromboembolism associated with a formulation containing 30 μg ethinyl estradiol and levonorgestrel (doses ranging from 125 to 250 μg) was compared with the risk with preparations containing 20 or 30 μg ethinyl estradiol and either desogestrel or gestodene (data from 10 centers in 9 countries).
143 The users of the levonorgestrel formulations had an increased odds ratio of 3.5 compared with nonusers. Current users of a desogestrel product had an increased risk of 9.1 compared with nonusers, and with gestodene, the odds ratio was also 9.1. Thus, the increased risk for desogestrel and gestodene was 2.6 times that of levonorgestrel, when adjusted for body weight and height.
The Transnational Study on Oral Contraceptives and the Health of Young Women analyzed 471 cases of deep vein thrombosis and/or venous thromboembolism from the United Kingdom and Germany.
144 Second-generation oral contraceptives were defined as products containing 35 μg or less of ethinyl estradiol and a progestin other than desogestrel or gestodene. Comparing users of second-generation products to nonusers, the odds ratio was 3.2. Comparing users of desogestrel and gestodene products to users of second-generation oral contraceptives, the risk of venous thromboembolism was 1.5-fold greater.
A third major study was from Boston University, but the data were derived from the General Practice Research Database, a computerized system involving the general practitioners in the U.K.
145 Using this cohort, the death rate from pulmonary embolism, stroke, and acute myocardial infarction was calculated in the users of levonorgestrel, desogestrel, and gestodene low-dose oral contraceptives. Over a 3-year period, they collected a total of 15 unexpected idiopathic cardiovascular deaths in users of these products, a
nonsignificant change, and no
difference in the risk comparing desogestrel and gestodene with levonorgestrel. The risk estimates for venous thromboembolism (adjusted for smoking and body size) were about 2 times greater for desogestrel and for gestodene, compared with levonorgestrel uses. In an updated analysis from this same group and database, the findings were unchanged, except that smoking was found to be a risk factor for venous thromboembolism.
146 An American case-control study concluded that norgestimate- and levonorgestrel-containing oral contraceptives had similar risks for venous thromboembolism, but there was a small increase in risk associated with desogestrel.
147Similar results were reported when women with deep vein thrombosis in the Leiden Thrombophilia Study in the Netherlands were reanalyzed for their use of oral contraceptives.
148 As expected, the risk of deep vein thrombosis was markedly higher in women who were carriers of the factor V Leiden mutation and in women with a family history of thrombosis.
In Denmark, Lidegaard and colleagues performed a hospital-based, case-control study of women with confirmed diagnoses of venous thromboembolism in 1994 and 1995 (in Denmark, all women with this diagnosis are hospitalized, and therefore, very few, if any, cases were missed).
149 A 2-fold increased risk of venous thromboembolism was found in current users of oral contraceptives, regardless of estrogen doses ranging from 20 to 50 μg. The increased risk was concentrated in the first year of use.
Because there were more short-term users of the new progestins and more long-term users of the older progestins, adjustment for duration of use resulted in no significant differences between the different types of progestins. Those factors associated with an increased risk of thromboembolism included coagulation disorders, treated hypertension during pregnancy, family history of venous thromboembolism, and an increasing body mass index. Notably, conditions not associated with an increased risk of venous thromboembolism included smoking, migraine, diabetes, hyperlipidemia, parity, or age at first birth. There was still insufficient strength in this study to establish the absence or presence of a dose-response relationship comparing the 20 μg estrogen dose to higher doses; however, a 5-year update reported the following useful information:
150
The risk of venous thrombosis associated with current use of oral contraceptives declined with increasing duration of use.
The risk was slightly greater with desogestrel or gestodene.
Smoking more than 10 cigarettes per day increased the risk.
Oral contraceptives with 20 μg estrogen had a lower risk than products with 30-40 μg.
Progestin-only contraceptive products did not increase the risk.
Case-control studies using cases of venous thromboembolism derived from the computer records of general practices in the U.K. concluded that the increased risk associated with oral contraceptives was the same for all types, and that the pattern of risk with specific oral contraceptives suggested confounding because of “preferential prescribing” (defined later).
151,152 In these studies, matching cases and controls by year of birth eliminated differences between different types of oral contraceptives. A similar analysis based on 42 cases from a German database again found no difference between new progestin and older progestin oral contraceptives.
153 Thus, in these two studies, more precise adjustments for age eliminated a confounding bias. An assessment of the incidence of venous thromboembolism in the U.K. before and after the decline in third-generation progestin use could detect no impact on the statistics (neither an increase nor a decrease).
154A reanalysis of the Transnational Case-Control Study considered the duration and patterns of oral contraceptive use.
155,156 This reanalysis focused on first-time users of second- and third-generation oral contraceptives.
Statistical analysis with adjustment for duration of use in 105 cases who were first-time users could find no differences between second- and third-generation products. A similar reanalysis of the U.K. General Practice Database could demonstrate no difference between different oral contraceptive formulations.157A case-control study in Germany assessed the outcome when the cases were restricted to hospitalized patients compared to results when all cases, both in-hospital and out-of-hospital, were considered.
158 The conclusion indicated that hospital-based studies overestimated the risk of venous thromboembolism, and that there was no difference comparing progestins when all cases were included.
Former users discontinue oral contraceptives for a variety of reasons, and often are switched to what clinicians perceive to be “safer” products, a practice called
“preferential prescribing.”159,160 and 161 Individuals who do well with a product tend to remain with that product. Thus, at any one point in time, individuals on an older product will be relatively healthy and free of side effects—the
“healthy user effect.” This is also called
attrition of susceptibles because higher risk individuals with problems are gradually eliminated from the group.
162 Comparing users of older and newer products, therefore, can involve disparate cohorts of individuals.Because desogestrel- and gestodene-containing products were marketed as less androgenic and therefore “better” (a marketing claim not substantiated by epidemiologic studies), clinicians chose to provide these products to higher risk patients and older women.
159,160 In addition, clinicians switched patients perceived to be at greater risk for thrombosis from older oral contraceptives to the newer formulations with desogestrel and gestodene. Furthermore, these products were prescribed more often to young women who were starting oral contraception for the first time (these young women will not have experienced the test of pregnancy or previous oral contraceptive use to help identify those who have an inherited predisposition to venous thrombosis). These changing practice patterns exert different effects over the lifetime of a product, and analytical adjustments are extremely difficult.
The initial studies were impressive in their agreement. All indicated increased relative risks associated with desogestrel and gestodene compared with levonorgestrel. Nevertheless, all of the early studies, somewhat similar in design, were influenced by the same unrecognized biases. Persistent errors will produce consistent conclusions.
Forty cases of venous thrombosis in drospirenone (Yasmin) users (two of which were fatal) were reported in Europe in 2002.
163 The Dutch College of General Practitioners issued a statement encouraging clinicians not to prescribe Yasmin. However, this is the similar story we experienced with “third-generation” progestins, only to learn that preferential prescribing and the healthy user effect probably biased the early case-control studies. In postmarketing surveillance, only one case of venous thrombosis occurred in a million cycles of Yasmin compared with five among users of other oral contraceptives.
163 In a subsequent monitoring study, the incidence of venous thromboembolism in new users was comparable to that seen with other low-dose oral contraceptives.
164 The European Active Surveillance Study (EURAS) was a large cohort study that enrolled
only new users of oral contraceptives containing a variety of progestins, including drospirenone and levonorgestrel.
165 The incidence of cardiovascular events was similar for all progestins. An American cohort study also focused on new users of oral contraceptives, and thromboembolism occurred at a similar low rate comparing drospirenone users with other oral contraceptives.
166The Danish investigators continued their interest in hormonal contraception and venous thrombosis, performing a national cohort study using the reliable Danish national registries of events from 1995 to 2005.
167 As in the earlier Danish case-control study,
150 the risk of venous thrombosis in current users of oral contraceptives decreased with duration of use and with the dose of estrogen, and was slightly higher with products containing desogestrel, gestodene, drospirenone, and cyproterone. Did this study escape the problems of preferential prescribing and the healthy user effect (attrition of susceptibles)? The incidence of thrombotic events in the comparator group (levonorgestrel users) was lower than that
reported in other studies, suggesting that this group did demonstrate a healthy user effect. The study was not limited to new users, a requirement in order to avoid the confounding due to attrition of susceptibles. The authors argued that preferential prescribing was not prevalent in Denmark after 1995 (but they offered no proof of this contention), and they further argued that the fact that the use of other medications was similar comparing levonorgestrel and drospirenone suggested a similar level of health and a lack of preferential prescribing. This study was unable to control for body mass index (BMI) or family history of thrombosis, two important markers for women at high risk of venous thrombosis. Preferential prescribing remains a possible confounder in the Danish study; however, the problem of a healthy user effect is even more likely.
Acase-controlstudy from the Netherlands also reported higher risks of venous thrombosis in users of desogestrel, gestodene, drospirenone, and cyproterone compared with levonorgestrel users.
168 The authors supported their results by citing findings from their own institution that users of oral contraceptives containing drospirenone and cyproterone have lower levels of free protein S and free tissue factor pathway inhibitor associated with greater resistance to activated protein C compared with levonorgestrel users.
169 The relative risks in this study were surprisingly high, higher than all other reports involving low-dose oral contraceptives. Once again, the healthy user effect was a likely confounder in that the study was not limited to new users. The authors claimed to compensate for the attrition of susceptibles by analyzing only short-term users. Even though the validity of this approach can be debated, the results indicated nonsignificant increased risks with drospirenone and cyproterone compared with levonorgestrel, and any conclusion was limited by a small number of short-term users. In this study, the risk associated with products containing 20 μg ethinyl estradiol was not increased.
An international study is underway, the International Active Surveillance study of women taking Oral Contraceptives (INAS-OC), designed to record cardiovascular events in a cohort of more than 80,000 oral contraceptive users.
170 Two-year and 5-year follow-up reports are anticipated.
The risk of venous thrombosis associated with modern oral estrogen-progestin contraceptives is increased about 2-fold, but manifested primarily in the first years of use and concentrated in overweight women.165,171,172 The risk, which increases with increasing body weight and age, is influenced in a major way by the estrogen dose, and the difference among progestin products is small, either real but not meaningful clinically or a reflection of biases and confounders. The impact of smoking on the risk of venous thrombosis is less than that on the risk of arterial thrombosis, but smoking, especially heavy smoking, may act synergistically with oral contraceptives.173
The risk of venous thromboembolism in the general population is now considered to be higher than previously estimated because of the prevalence of modern diagnostic methods. The new studies also indicate that the risk associated with low-dose oral contraceptives is lower than previously reported, and more prevalent in high-risk individuals (obesity and inherited or acquired thrombophilias). Oral contraceptives with the lowest doses of estrogen should be used for overweight and older women, but progestin-only methods are an even better choice.
Venous Thromboembolism and Thrombophilias
An inherited resistance to activated protein C, the factor V Leiden mutation, may account for a significant portion of the patients who experience venous thrombosis while taking oral contraceptives.
The factor V Leiden mutation is the most common inherited coagulation problem, transmitted in an autosomal-dominant fashion.
107,174 Heterozygotes have a 6- to 8-fold increased risk of venous thromboembolism, and homozygotes an 80-fold increased risk. Oral contraceptive users who have this mutation have been reported to have a 30-fold increased risk of venous thrombosis.
175,176 Some have effectively argued, however, that this increase has been overestimated, and it is closer to 10-15-fold.
177 The risk of developing venous thrombosis is greatest in the initial months of use, and it has been suggested that venous thrombosis occurring in the first month of exposure should make the clinician suspect the presence of a clotting disorder.
178Should screening for the factor V Leiden mutation (or for other inherited clotting disorders) be routine prior to prescribing contraceptives? The carrier frequencies of the Leiden mutation in the American population (the percentages are similar in men and women) are as follows:
179
Caucasian Americans |
5.27% |
Hispanic Americans |
2.21% |
Native Americans |
1.25% |
Black Americans |
1.23% |
Asian Americans |
0.45% |
These estimates are consistent with European assessments, indicating that this is a trait carried in people of European origin. About 1 in 5,000 individuals is homozygous for the Leiden mutation. In the United States, of the approximately 12 million women currently using oral contraceptives, about 540,000 are likely to carry the factor V Leiden mutation. However, because the incidence rate of venous thromboembolism is so low (5-10 per 10,000 young women per year), the number of women required to be screened to prevent one event is prohibitively large. The prevalence of all deficiencies is only about 0.5% in the asymptomatic population, and only one-third of patients at risk are detected by the present tests.
180Furthermore, because only a small number of women even with the Leiden mutation (less than 1 in 1,000) have a clinical event (99.85% of the individuals who test positive will NOT have a clinical event!), the finding of a positive screening test, especially considering the high rate of false-positive tests, would be a barrier to the use of oral contraceptives, and a subsequent increase in unwanted pregnancies would likely follow. Pregnancy, of course, has a much greater risk of venous thromboembolism than oral contraceptives. Most experts believe that screening for inherited disorders should be pursued only in women with a previous episode of venous thromboembolism or a close positive family history (parent or sibling) of venous thrombosis.
The second most prevalent inherited thrombophilia is the prothrombin gene
20210A mutation. A combination of the prothrombin gene mutation and the Leiden mutation is found
in about 2% of venous thromboembolism cases.
181 Less frequent are genetic defects in coagulation inhibitors (antithrombin, protein C, and protein S), but these defects carry a substantial increase in risk.
Acquired thrombophilias include the presence of antiphospholipid antibodies (lupus anticoagulant and anticardiolipin) usually associated with autoimmune diseases.
The inherited and acquired thrombophilias predispose to venous thromboembolism in a synergistic manner with estrogen-containing contraceptives. However, the actual incidence of events is low, and identification of a thrombophilia does not predict a clinical event.
Arterial Thrombosis
Because the incidence of cerebral thrombotic attacks (thrombotic strokes and transient ischemic attacks) among young women is higher than venous thromboembolism and myocardial infarction, and death and disability are more likely, cerebral arterial thrombosis is the most important possible side effect. Nevertheless, the incidence is low, and a small increase in the very low incidence of stroke in young women carries with it little increase in absolute risk. Because the incidence of cerebral thrombotic attacks is higher in women over age 40, we should do our best, as the following discussion will indicate, to make sure estrogen-progestin contraceptive users over age 40 are in good health and without significant risk factors for cardiovascular disease (especially hypertension, migraine with aura, and smoking).
Although it has been difficult to establish arterial thrombosis dose-response relationships with estrogen because these events are so rare, modern, lower estrogen doses are related to the risk of myocardial infarction and thrombotic strokes in case-control studies.
182,183 Thus, a rationale for advocating low-dose estrogen oral contraceptives continues to be valid.
Arterial Thrombosis—Myocardial Infarction
A population-based, case-control study analyzed 187 cases of myocardial infarction in users of low-dose oral contraceptives in the Kaiser Permanente Medical Care Program.
184There was no statistically significant increase in the odds ratio for myocardial infarction in current oral contraceptive users compared with past or never users.
In the Transnational case-control study of myocardial infarctions collected from 16 centers in Austria, France, Germany, Switzerland, and the U.K. cigarette smoking carried a higher risk for myocardial infarction than oral contraceptives, and
nonsmoking users of oral contraceptives had no evidence of an increased risk.185,186 In addition, there was an indication that patient screening is important in minimizing the impact of hypertension on the risk of myocardial infarction. Similar results were reported in a case-control study based on subjects in England, Scotland, and Wales, and another in America.
187,188In the WHO multicenter study, there were 368 cases of acute myocardial infarction.
189 Factors associated with an increased risk of myocardial infarction included smoking, a history of hypertension (including hypertension in pregnancy), diabetes, rheumatic heart disease, abnormal blood lipids, and a family history of stroke or myocardial infarction. Duration of use and past use of oral contraceptives did not affect risk. Although there was about a 5-fold overall increased odds ratio of myocardial infarction in current users of oral contraceptives, essentially all cases occurred in women with cardiovascular risk factors. There was no apparent effect of increasing age on risk; however, there were only 12 cases among oral contraceptives users less than 35 years old. There was no apparent relationship
with estrogen dose, and there was no apparent influence of type or dose of progestin, but the rare occurrence of this condition produced such small numbers that there was insufficient statistical power to accurately assess the effects of progestin type, and estrogen and progestin doses.
The conclusion of this study was that the risk of myocardial infarction in women who use oral contraceptives is increased only in smokers.In a Danish case-control study of acute myocardial infarction in young women, a statistically significant increase in risk was noted only in current users of 50 μg ethinyl estradiol.183 There was a progressive increase in risk with the number of cigarettes smoked, (accounting for 80% of the acute myocardial infarctions in young women), increasing body mass index, treated hypertension, treated hypertension in pregnancy, diabetes mellitus, hyperlipidemia, frequent migraine, and family history of myocardial infarction. However, only family history of myocardial infarction and smoking affected the risk associated with oral contraceptives; no influence on oral contraceptive risk was apparent with diabetes, hypertension, and heart disease. No differences could be demonstrated according to type of progestin.
A case-control study from the Netherlands found that the risk of myocardial infarction was highest among users of oral contraceptives who smoked, had diabetes mellitus, or who were hypercholesterolemic.
190 The risk of myocardial infarction was not affected by the presence of the factor V Leiden mutation or the prothrombin gene mutation.
The Women’s Lifestyle and Health Cohort Study is a prospective cohort study of 106,841 Norwegian and Swedish women, started in 1991, specifically designed to assess the longterm health effects of hormonal contraceptives.
191 Low-dose (less than 50 μg ethinyl estradiol) oral contraceptives were not associated with an increased risk of myocardial infarction. All previous cohort studies date back to oral contraceptive use with higher doses of estrogen in the 1970s and 1980s. For example, in the report from the Nurses’ Health Study in 1988, an increased risk of myocardial infarction was found in current users.
192Case-control studies of low-dose estrogen oral contraceptives have concluded that an increased risk of arterial disease occurs only in women who have hypertension or are smokers.
124,184,185,189,190 The cohort studies don’t help us with this important issue because the numbers are too small for definitive analyses of subgroups. Nevertheless, British and Finnish cohorts were reported to have increased risks of developing myocardial infarction in oral contraceptive users who smoked.
193,194
Arterial Thrombosis—Stroke
Older case-control and cohort studies indicated an increased risk of cerebral thrombosis among current users of high-dose oral contraceptives.
196,197 and
198 However, thrombotic stroke did not appear to be increased in healthy, nonsmoking women with the use of oral contraceptives containing less than 50 μg ethinyl estradiol.
197,198 A case-control study of all 794 women in Denmark who suffered a cerebral thromboembolic attack during 1985-1989 concluded that there was an almost 2-fold increased relative risk associated with oral contraceptives containing 30-40 μg estrogen, and the risk was significantly influenced by both smoking and the dose of estrogen in additive (not synergistic) fashion.
137 A case-control analysis of data collected by the Royal College of General Practitioners’ Oral Contraception Study concluded that current users were at increased risk of stroke (with a persisting effect in former users); however, this outcome was limited mainly to smokers and to formulations with 50 μg or more of estrogen.
198A population-based, case-control study of 408 strokes from the California Kaiser Permanente Medical Care Program found no increase in risk for either ischemic stroke or hemorrhagic stroke.
199 The identifiable risk factors for ischemic stroke were smoking, hypertension, diabetes, elevated body weight, and low socioeconomic status. The risk factors for hemorrhagic stroke were the same plus greater body mass and heavy use of alcohol.
Current users of low-dose oral contraceptives did not have an increased risk of ischemic or hemorrhagic stroke compared with former users and with never users. There was no evidence for an adverse effect of increasing age or for smoking (for hemorrhagic stroke, there was a suggestion of a positive interaction between current oral contraceptive use and smoking, but the numbers were small, and the result was not statistically significant).
The Transnational study analyzed their data for ischemic stroke in a case-control study of 220 ischemic strokes in the U.K., Germany, France, Switzerland, and Austria.
200 Overall, there was a 3-fold increase in the risk of ischemic stroke associated with the use of oral contraceptives, with higher risks observed in smokers (more than 10 cigarettes per day), in women with hypertension, and in users of higher dose estrogen products. No differences were observed comparing second- and third-generation progestins. A Dutch case-control study also found no differences comparing second and third generation progestins.
201 A case-control study from the state of Washington concluded that there is no increased risk of stroke in current users of low-dose oral contraceptives.
202 A pooled analysis of the data from California and Washington concluded that low-dose oral contraceptives are not associated with an increase in the risk of stroke.
203The World Health Organization data on stroke come from the same collaborative study that yielded the publications on venous thromboembolism. The results with stroke were published as two separate reports, one on ischemic stroke and the other on hemorrhagic
This hospital-based, case-control study from 21 centers in 17 countries accumulated 697 cases of ischemic stroke, 141 from Europe and 556 from developing countries.
204 The overall odds ratio for ischemic stroke indicated about a 3-fold increased risk. In Europe, however, the risk was statistically significant only for higher-dose products, and
NOT statistically significant for products with less than 50 μg ethinyl estradiol. In developing countries, there was no difference in risk with low-dose and higher dose oral contraceptives. This is believed to be due to the strong influence of hypertension. In Europe, it was uncommon for women with a history of hypertension to be using oral contraceptives; however, this was not the case in developing countries. Duration of use and type of progestin had no impact, and past users did not have an increased risk, but smoking 10 or more cigarettes daily exerted a synergistic effect with oral contraceptives, increasing the risk
of ischemic stroke, approximating the effect of hypertension and oral contraceptives. The risk was greater in women 35 years and older; however, this, too, was believed to be due to an effect of hypertension.
Thus, the conclusion of this study was that the risk of ischemic stroke is extremely low, concentrated in those who use higher dose products, smoke, or have hypertension.In the WHO study on hemorrhagic stroke, there were 1,068 cases.
205 Current use of oral contraceptives was associated with a slightly increased risk of hemorrhagic stroke only in developing countries, not in Europe. This again probably reflects the presence of hypertension, because the greatest increased risk (about 10- to 15-fold) was identified in current users of oral contraceptives who had a history of hypertension. Current cigarette smoking also increased the risk in oral contraceptive users, but not as dramatically as hypertension. For hemorrhagic stroke, the dose of estrogen had no effect on risk, and neither did duration of use
or type of progestin.
This study concluded that the risk of hemorrhagic stroke due to oral contraceptives is increased only slightly in older women, probably occurring only in women with risk factors such as hypertension.A second Danish case-control study included thrombotic strokes and transitory cerebral ischemic attacks analyzed together as cerebral thromboembolic attacks.
182 In this study, the 219 cases during 1994 and 1995 included 146 cases of cerebral infarction and 73 cases of transient ischemic attacks. There was a dose-response relationship with estrogen in the dose ranges of 20, 30-40, and 50 μg ethinyl estradiol, although the number of 20 μg users (5 cases, 22 controls) was not sufficient to establish a lower risk at this lower dose. This analysis claimed a reduced risk associated with desogestrel and gestodene; however, the odds ratio did not achieve statistical significance. An updated 5-year report of the Danish case-control study indicated that the odds ratio of cerebral thrombosis decreased from a high of 4.5 with 50 μg estrogen pills to 1.6 with 20-40 μg pills.
206 Hypertension increased the risk 5-fold, migraine 3.2 times, diabetes 5.6 times, hyperlipidemia and coagulation disorders about 12-fold.
The Norwegian-Swedish Women’s Lifestyle and Health Cohort Study supports the conventional wisdom of the last decade that low-dose oral contraceptives do not increase the myocardial infarction or stroke in healthy, nonsmoking women, regardless of age.
207 Screening for hypertension is especially important in that it is a major risk factor for stroke associated with oral contraceptive use.
In the absence of hypertension, the effect of smoking in women under age 35 is too small to be measured. In the presence of hypertension, it is currently believed that with medical control of blood pressure and close follow-up (blood pressure monitoring every 3 months), nonsmoking women under age 35 and otherwise healthy can use low-dose oral contraception.
Conclusion—Myocardial Infarction and Stroke
Oral contraceptives containing less than 50 μg ethinyl estradiol do not increase the risk of myocardial infarction or stroke in healthy, nonsmoking women, regardless of age.
The effect of smoking in women under age 35 is, as we have long recognized, not detectable in the absence of hypertension. After age 35, the subtle presence of hypertension makes analysis difficult, but the Kaiser study indicates that increasing age and smoking by themselves have little impact on the risk of stroke in low-dose oral contraceptive users. The screening of patients in the Kaiser program was excellent, resulting in few women with hypertension using oral contraceptives. There is no reason to doubt that these conclusions apply as well to the transdermal and vaginal methods of steroid contraception.
Epidemiologic studies fail to find any substantial risk of ischemic or hemorrhagic stroke with low-dose oral contraceptives in healthy, young women. The WHO study did find evidence for an adverse impact of smoking in women under age 35; the Kaiser study did not. This difference is explained by the confounding effect of hypertension, the major risk factor identified. In the WHO study, a history of hypertension was based on whether a patient reported ever having had high blood pressure (other than in pregnancy) and not validated by medical records. In the Kaiser study, women were classified as having hypertension if they reported using antihypertensive medication (less than 5% of oral contraceptive users had treated hypertension, and there were no users of higher dose products). In the WHO study, the effect of using oral contraceptives in the presence of a high-risk factor is apparent in the different odds ratios when European women who received good screening from clinicians were compared with women in developing countries who received little screening; therefore, more women with cardiovascular risk factors in developing countries were using oral contraceptives. The studies indicate that hypertension should be a major concern, especially in regards to the risk of stroke.
Over the years, there has been recurring discussion over whether to provide oral contraceptives over-the-counter on a nonprescription basis. The data in the WHO study make an impressive argument against such a move. The increased risk of myocardial infarction was most evident in developing countries where 70% of the cases received their oral contraceptives from a nonclinical source. Deprived of screening, women with risk factors in developing countries were exposed to greater risk.
Smoking
Smoking continues to be a difficult problem, not only for patient management, but for analysis of data as well. In large U.S. surveys in 1982 and 1988, the decline in the prevalence of smoking was similar in users and nonusers of oral contraception; however, 24.3% of 35- to 45-year-old women who used oral contraceptives were smokers!
208 In this group of smoking, oral contraceptive-using women, 85.3% smoked 15 or more cigarettes per day (heavy smoking). Despite the widespread teaching and publicity that smoking is a contraindication to oral contraceptive use over the age of 35, more older women who use oral contraceptives smoke and smoke heavily, compared with young women. This strongly implies that older smokers are less than honest with clinicians when requesting oral contraception, and this further raises serious concern over how well this confounding variable can be controlled in case-control and cohort studies. Studies documenting the effects of smoking bans on hospital admissions for myocardial infarctions indicate a rapid reduction in 6 months to a year.
209,210 A former smoker must have stopped smoking for at least 6 consecutive months and preferably 12 months to be regarded as a nonsmoker. Women who have nicotine in their bloodstream obtained from patches or gum should be regarded as smokers.
Lipoproteins and Oral Contraception
The balance of estrogen and progestin potency in a given oral contraceptive formulation can potentially influence cardiovascular risk by its overall effect on lipoprotein levels. Oral contraceptives with relatively high doses of progestins (doses not used in today’s low-dose formulations) do produce unfavorable lipoprotein changes.
211 The levonorgestrel triphasic exerts no significant changes on HDL-cholesterol, LDL-cholesterol, apoprotein B, and no change or an increase in apoprotein A. The monophasic desogestrel and desogestrel pills have a favorable effect on the lipoprotein profile, while the triphasic norgestimate and gestodene pills produce beneficial alterations in the LDL/HDL and apoprotein B/apoprotein A ratios.
212,213,214 and
215 Like the triphasic levonorgestrel pills, norethindrone multiphasic pills have no significant impact on the lipoprotein profile over 6-12 months.
216 In summary, studies of low-dose formulations indicate that the adverse effects of progestins are limited to the fixed-dose combination with a dose of levonorgestrel that exceeds that in the multiphasic formulation or in the low-dose products. The formulation that contains 100 μg levonorgestrel and 20 μg ethinyl estradiol produces short-term changes in the lipid profile that are similar to those seen with other low-dose oral contraceptives, and with long-term use, the levels revert to those observed at baseline before treatment.
217An important study in monkeys indicated a protective action of estrogen against atherosclerosis, but by a mechanism independent of the cholesterol-lipoprotein profile. Oral administration of a combination of estrogen and progestin to monkeys fed a high-cholesterol, atherogenic diet decreased the extent of coronary atherosclerosis despite a reduction in HDL-cholesterol levels.
218,219 and
220 In somewhat similar experiments, estrogen treatment markedly prevented arterial lesion development in rabbits.
221,222 and
223 In considering the impact of progestational agents, lowering of HDL is not necessarily atherogenic if accompanied by a significant estrogen impact. These animal studies help explain why older, higher dose combinations, which had an adverse impact on the lipoprotein profile did not increase subsequent cardiovascular disease.
124,127 The estrogen component provided protection through a direct effect on vessel walls, especially favorably influencing vasomotor and platelet factors such as nitric oxide and prostacyclin.
This conclusion is reinforced by angiographic and autopsy studies. Young women with myocardial infarctions who have used oral contraceptives have less diffuse atherosclerosis than nonusers.
224,225 Indeed, a case-control study indicated that the risk of myocardial infarction in patients taking older, high-dose levonorgestrel-containing formulations is the same as that experienced with pills containing other progestins.
124In the past two decades, we were subjected to considerable marketing hype about the importance of the impact of oral contraceptives on the cholesterol-lipoprotein profile. If indeed certain oral contraceptives had a negative impact on the lipoprotein profile, one would expect to find evidence of atherosclerosis as a cause of an increase in subsequent cardiovascular disease. There is no such evidence. Thus, the mechanism of the cardiovascular complications is undoubtedly a short-term acute mechanism—thrombosis (an estrogen-related effect).
Remember too that the new progestins, because of their reduced androgenicity, predictably do not adversely affect the cholesterol-lipoprotein profile. The estrogen-progestin balance of combined oral contraceptives containing one of the new progestins even promotes favorable lipid changes.
38 Thus, the new formulations have the potential to offer protection against cardiovascular disease, an important consideration as we enter an era of women using oral contraceptives for longer durations and later in life. But one must be cautious regarding the clinical significance of subtle changes, and as with the older progestins and
adverse changes in lipoproteins, it is unlikely that oral contraceptives will have a clinically meaningful beneficial effect on the incidence of coronary heart disease.
Hypertension
Oral contraceptive-induced hypertension was observed in approximately 5% of users of higher dose pills. More recent evidence indicates that small increases in blood pressure can be observed even with 30 μg estrogen, monophasic pills, including those containing the new progestins. However, an increased incidence of clinically significant hypertension has not been reported.
226,227,228 and
229 The lack of clinical hypertension in most studies may be due to the rarity of its occurrence. The Nurses’ Health Study observed an increased risk of clinical hypertension in current users of low-dose oral contraceptives, providing an incidence of 41.5 cases per 10,000 women per year.
230 Therefore, an annual assessment of blood pressure is still an important element of clinical surveillance, even when low-dose oral contraceptives are used. Postmenopausal women in the Rancho Bernardo Study who had previously used oral contraceptives (probably high-dose products) had slightly higher
(2-4 mm Hg) diastolic blood pressures.
231 Because past users do not demonstrate differences in incidence or risk factors for cardiovascular disease, it is unlikely this blood pressure difference has an important clinical effect.
Variables such as previous toxemia of pregnancy or previous renal disease do not predict whether a woman will develop hypertension on oral contraception.
232 Likewise, women who have developed hypertension on oral contraception are not more predisposed to develop toxemia of pregnancy. Overall, there is no evidence that previous oral contraceptive users have an increased risk of hypertension during a subsequent pregnancy.
233, 234 The exception is the Nurses’ Health Study, which indicated that recent users for a long duration (8 or more years) have a 2-fold increased risk of preeclampsia, a finding that was based on a small number of cases.
235 These epidemiologic associations are hard to establish because of the role of underlying hypertension in pregnancy-induced hypertension and the difficulty in assessing the efficacy of hypertension screening in oral contraceptive users.
The mechanism for an effect on blood pressure is thought to involve the renin angiotensin system. The most consistent finding is a marked increase in plasma angiotensinogen, the renin substrate, up to 8 times normal values (on higher dose pills). In nearly all women, excessive vasoconstriction is prevented by a compensatory decrease in plasma renin concentration. If hypertension does develop, the renin-angiotensinogen changes take 3-6 months to disappear after stopping combined oral contraception.
One must also consider the effects of oral contraceptives in patients with preexisting hypertension or cardiac disease. Women on oral contraceptives and with uncontrolled hypertension have an increased risk of arterial thrombosis.
189,204,205 Women with treated hypertension using oral contraceptives have been reported to have poor control of blood pressure with higher diastolic pressures.
236 In our view, with medical control of the blood pressure and close follow-up (at least every 3 months), the clinician and the nonsmoking patient who is under age 35 and otherwise healthy may choose low-dose oral contraception. Close follow-up is also indicated in women with a history of preexisting renal disease or a strong family history of hypertension or cardiovascular disease. It seems prudent to suggest that patients with marginal cardiac reserve should utilize other means of contraception. Significant increases in cardiac output and plasma volume have been recorded with oral contraceptive use (higher dose pills), probably a result of fluid retention.
Epidemiologic studies reinforce the belief that the risks of arterial and venous thrombosis are a consequence of the estrogen component of combination oral contraceptives. Current evidence does not support an advantage or disadvantage for any particular formulation, except for the greater safety associated with any product containing less than 50 μg ethinyl estradiol. Although it is logical to expect the greatest safety with the lowest dose of estrogen, the rare occurrence of arterial and venous thrombosis in healthy women makes it unlikely that there will be any measurable differences in the attributable incidence of clinical events with all low-dose products.
The epidemiologic data emphasize the importance of good patient screening. The occurrence of arterial thrombosis is essentially limited to older women who smoke or have cardiovascular risk factors, especially hypertension. The impact of good screening is evident in the repeated failure to detect an increase in mortality due to myocardial infarction or stroke in healthy, nonsmoking women.
129,145,195 Although the risk of venous thromboembolism is
slightly increased, the actual incidence is still relatively rare. The overall mortality rate with venous thromboembolism is about 1%, probably less with oral contraceptives, because most deaths from thromboembolism are associated with trauma, surgery, or a major illness. The minimal risk of venous thrombosis associated with oral contraceptive use does not justify the cost of routine screening for coagulation deficiencies. Nevertheless, the importance of this issue is illustrated by the increased risk of a very rare event, cerebral sinus thrombosis, in women who have an inherited predisposition for clotting and use oral contraceptives.
106,237
If a patient has a close family history (parent or sibling) or a previous episode of idiopathic thromboembolism, an evaluation to search for an underlying abnormality in the coagulation system is warranted. It has been reported that family history of venous thromboembolism has low predictive value.
238 Another study indicated that testing for thrombophilia did not allow for prediction of recurrent events, but risk factors such as family history did provide prediction.
239 The conservative recommendation for a high-risk woman considering exposure to exogenous estrogen stimulation is to rule out an underlying thrombophilia. The following measurements are recommended, and abnormal results require consultation with a hematologist regarding prognosis and prophylactic treatment. The list of laboratory tests is long, and because this is a dynamic and changing field, the best advice is to consult with a hematologist. If a diagnosis of a congenital deficiency is made, screening should be offered to other family members.
Hypercoaguable Conditions |
Thrombophilia Screening |
Antithrombin III deficiency |
Antithrombin III |
Protein C deficiency |
Protein C |
Protein S deficiency |
Protein S |
Factor V Leiden mutation |
Activated protein C resistance ratio |
Prothrombin gene mutation |
Activated partial thromboplastin time |
Antiphospholipid syndrome |
Hexagonal activated partial thromboplastin time |
|
Anticardiolipin antibodies |
|
Lupus anticoagulant |
|
Fibrinogen |
|
Prothrombin G mutation (DNA test) |
|
Thrombin time |
|
Homocysteine level |
|
Complete blood count |
Combination estrogen-progestin contraception is contraindicated in women who have a history of idiopathic venous thromboembolism, and also in women who have a close family history (parent or sibling) of idiopathic venous thromboembolism. These women will have a higher incidence of congenital deficiencies in important clotting measurements, especially antithrombin III, protein C, protein S, and resistance to activated protein C.
240 Such a patient who screens negatively for an inherited clotting deficiency might still consider the use of oral contraceptives, but this would be a difficult decision with unknown risks for both patient and clinician, and it is more prudent to consider other contraceptive options. Other risk factors for thromboembolism that should be considered by clinicians include an acquired predisposition such as the presence of lupus anticoagulant or malignancy, and immobility or trauma. Varicose veins are not a risk factor unless they are very extensive.
140 Progestin-only methods, including implants, depot medroxyprogesterone acetate, and the levonorgestrel-releasing IUS are recommended for high risk women and for women who are anticoagulated.
The conclusion once again is that low-dose oral contraceptives are very safe for healthy, young women. By effectively screening older women for the presence of smoking and cardiovascular risk factors, especially hypertension, we can limit, if not eliminate, any increased risk for arterial disease associated with low-dose oral contraceptives. And it is very important to emphasize that there is no increased risk of cardiovascular events associated with duration of use (long-term). In large cohort studies, the risk of overall mortality comparing
users and nonusers of oral contraceptives is identical.
127,128 and
129 It is worth noting again that these conclusions likely also apply to transdermal and vaginal estrogen-progestin contraception.