Immunology and Immunodeficiencies
Rebecca H. Buckley
Division of Pediatric Allergy and Immunology, Duke University Medical Center, Durham, North Carolina 27710.
CELLULAR AND HUMORAL BASES OF THE IMMUNE RESPONSE
Cells of the Immune System
The immune system consists of lymphoid and phagocytic cells, their secreted products, and the complement proteins. Lymphocytes that have the ability to differentiate into antibody-forming cells are referred to as B cells because of their origin in the bone marrow. T cells are so named because they develop in the thymus. Although T cells do not produce antibodies, they play a central role in all specific immune responses through interactions with other immune cells by their cell surface molecules and by producing soluble factors (cytokines) that influence the function of many types of cells (1,2). They and the other cells of the immune system also bear adhesion or costimulatory molecules on their surfaces that facilitate cell–cell interaction, a necessary component of immune responses (3). Adhesion molecules are also crucial for the trafficking of immune cells to lymphoid organs and tissues, as well as to sites of inflammation. In contrast to T and B lymphocytes, which have antigen-specific recognition capabilities, natural killer (NK) cells mediate nonantigen-specific cytotoxicity; they originate from both the bone marrow and the thymus. NK cells are believed to assist in protection against viral infections, in tumor surveillance, and in immunoregulation. The immune system thus has a crucial role in all host defense. Its ability to recognize foreign antigens also accounts for its ability to reject organ and tissue grafts, and to produce allergic reactions. In addition, the immune system serves to protect against autoimmune diseases and malignancy.
Development of Cells and Organs of the Immune System
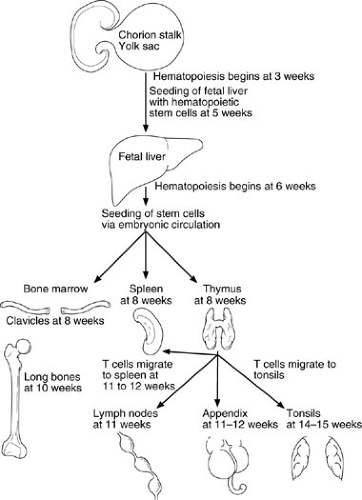
Stem cells appear in the yolk sac and fetal liver by 5 weeks’ gestation; they later reside in the bone marrow, where they remain throughout life. Lymphoid and phagocytic cells arise from these stem cells. Lymphoid stem cells differentiate into T, B, or NK cells, depending on the organs or tissues to which they migrate (Fig. 15-1). The initial migration and development, as well as the continued differentiation of lymphoid cells throughout life, occur through the interaction of the previously mentioned lymphocytic and tissue cell–surface adhesion molecules (Table 15-1), and under the influence of cytokines (Table 15-2) secreted by the involved cells (1,3). Primary lymphoid organ (thymus, bone marrow) development begins during the middle of the first trimester of gestation and proceeds rapidly; secondary lymphoid organ (spleen, lymph nodes, tonsils, Peyer’s patches, lamina propria) development soon follows. This process is almost complete by the end of the first trimester of gestation (4).
Clusters of Differentiation Classification of Human Leukocyte Differentiation Antigens
The World Health Organization has developed an official classification of well-characterized surface molecules present on immune cells (3). Each molecule is given a number that is preceded by the letters CD, which stand for clusters of differentiation (Table 15-1). This terminology is used throughout this chapter in referring to the cells of the immune system and other cells with which they interact.
Immune Cellular Function
T Cells
Two of the main functions of T cells are (1) to provide cytotoxic activity against facultative intracellular pathogens (mycobacteria, fungi) and virally infected cells or tumor
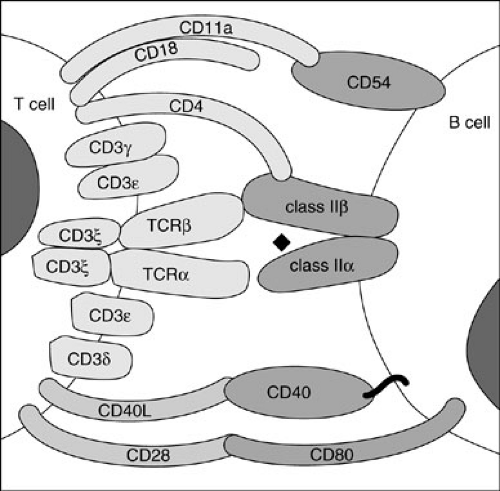
cells, and (2) to provide cytokines (Table 15-2) to help B cells make antibodies. For the T cell to carry out these functions, it first must bind to the target cell or to the antigen-presenting cell (APC), respectively. For high-affinity binding of T cells to target cells or APCs, several molecules on T cells in addition to TCRs bind to molecules on their respective target cells (Fig. 15-2). CD8 molecules on cytotoxic T cells bind to MHC class I molecules on target cells. CD4 molecules on helper T cells bind to MHC class II molecules on APCs. Lymphocyte function antigen 1 (LFA-1) molecules on T cells bind to intracellular adhesion molecule 1 (ICAM-1 or CD54) on APCs. CD2 molecules on T cells bind to LFA-3 or CD58 on APCs. With the adhesion of cytotoxic T cells to their targets, they are stimulated to kill the target; and with the adhesion of helper T cells to APCs, they are stimulated to produce cytokines, which stimulate B cells and other cells. Many other adhesion pairs participate in these important cell–cell interactions.
cells, and (2) to provide cytokines (Table 15-2) to help B cells make antibodies. For the T cell to carry out these functions, it first must bind to the target cell or to the antigen-presenting cell (APC), respectively. For high-affinity binding of T cells to target cells or APCs, several molecules on T cells in addition to TCRs bind to molecules on their respective target cells (Fig. 15-2). CD8 molecules on cytotoxic T cells bind to MHC class I molecules on target cells. CD4 molecules on helper T cells bind to MHC class II molecules on APCs. Lymphocyte function antigen 1 (LFA-1) molecules on T cells bind to intracellular adhesion molecule 1 (ICAM-1 or CD54) on APCs. CD2 molecules on T cells bind to LFA-3 or CD58 on APCs. With the adhesion of cytotoxic T cells to their targets, they are stimulated to kill the target; and with the adhesion of helper T cells to APCs, they are stimulated to produce cytokines, which stimulate B cells and other cells. Many other adhesion pairs participate in these important cell–cell interactions.
B Cells
In the primary antibody response, native antigen is carried to a lymph node draining the site of entry, taken up by specialized cells called follicle-stimulating cells (FSCs), and expressed on their surfaces. Virgin B cells bearing surface immunoglobulin specific for that antigen then bind to the antigen; if the affinity of the B-cell surface immunoglobulin for the antigen present on the FSCs is high, and if other signals are provided by activated helper T cells, the B cell develops into an antibody-producing plasma cell. If the affinity is not high enough, or if T-cell signals are not received, the B cell dies by apoptosis. The signals provided by activated helper T cells include those from cytokines they secrete (IL-4, IL-5, IL-6, IL-10, and IL-13) (1); (Table 15-2) and the signal from a surface T-cell molecule, CD154, which binds to CD40 on the B-cell surface (5) (Fig. 15-2). Cross-linking of CD40 on B cells or allowing CD40 to interact with CD154 in the presence of certain cytokines causes the B cells to undergo proliferation and to initiate immunoglobulin synthesis. In the primary immune response, usually only IgM antibody is made, and most of it is of relatively low affinity. Some B cells become memory B cells during the primary immune response. These cells have switched their immunoglobulin genes so IgG, IgA, or IgE antibodies of higher affinity are formed on a secondary exposure to the same antigen. The secondary immune response occurs when these memory B cells again encounter that antigen. Plasma cells form, just as in the primary response; however, many more cells are rapidly generated, and IgG, IgA, and IgE antibodies of increased affinity are produced. The exact pattern of isotype response to antigen varies, depending on the type of antigen and the cytokines present in the microenvironment.
Natural Killer Cells
NK lymphocytes are derived from bone marrow stem cells and are defined by their functional capacity to mediate nonantigen-specific cytotoxicity. NK cell activity has been found in human fetal liver cells by 8 to 11 weeks’ gestation. Unlike T and B cells, NK cells do not rearrange antigen-receptor genes during their development. All NK cells express CD56, and most also bear CD16 [Fcγ receptor III (FcγRIII)] on their cell surfaces. Because NK cells share surface antigens with T and myeloid cells, the lineage relation of NK cells is still unclear. NK cells have been found in the thymus, but most experimental studies suggest that thymic processing is not necessary for NK cell development. After release from bone marrow, NK cells enter the circulation or migrate to the spleen; there are few NK cells in lymph nodes. The percentage of NK cells in cord blood is usually lower than in the blood of children and adults (constituting 10% of lymphocytes), but the absolute number of NK cells is about the same, owing to a higher lymphocyte count. The capacity of cord blood NK cells to mediate target lysis in either NK cell assays or antibody-dependent cell-mediated cytotoxity assays is roughly two-thirds that of adults (4).
TABLE 15-1 Clusters of Differentiation (CD) Classification of Some Lymphocyte Surface Molecules.a | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lymphoid Organs
At the time of birth, lymphoid organs are proportionally small but microscopically well developed; they mature rapidly during the postnatal period. The neonate’s thymus is about two-thirds its mature weight, and it reaches that weight by the end of the first year of life. Most lymphoid structures also appear to be mature histologically by the end of the first year. Peripheral blood absolute lymphocyte counts reach a peak by 9 months of age. All lymphoid tissues continue to enlarge and even exceed adult dimensions during the prepubertal years, then all except the spleen undergo involution coincident with puberty. The spleen gradually increases in size during maturation and does not reach full weight until adulthood. The mean number of Peyer patches is one-half the adult number at birth and gradually increases until the adult mean number is exceeded during adolescent years (4).
Phagocytic Cells
The principal circulating phagocyte is the neutrophil, whereas the predominant phagocytic cell in tissues is the macrophage. Monocytes are derived from the bone marrow; subsequently, they can differentiate into tissue macrophages. Bone marrow neutrophil maturation is influenced by several factors secreted by stromal cells, monocytes, and T lymphocytes (1). These cytokines include IL-7, IL-3, granulocyte colony-stimulating factor, and granulocyte-monocyte colony-stimulating factor. Mature neutrophils migrate from the bone marrow into the circulation. To carry out their roles, phagocytes must be produced in adequate numbers and mobilized at the appropriate time. To perform their functions, neutrophils must marginate from the intravascular compartment, be deformable enough to undergo diapedesis, migrate along a chemotactic gradient to the site of inflammation, phagocytose, and have adequate lysosomal constituents and oxidative metabolism to kill microorganisms. A defect in any of these characteristics can lead to increased susceptibility to infection (6).
Phagocytosis is the process by which cells ingest particles encountered in their microenvironment so as to contain or destroy them (6). It is facilitated by a variety of substances known as opsonins. The most important opsonins are heat-stable IgG antibodies and the heat-labile complement components C3 and C5. Particles, such as bacteria, are coated with these substances; interaction with phagocytic cell membrane receptors for the opsonins (IgG and C3b) initiates ingestion of the particles. They then enter
a phagocytic vacuole that ultimately fuses with specific and azurophilic granules. Destruction of microorganisms requires generation of reactive oxygen molecules through a phagocytic cell respiratory burst. These oxygen radicals interact with myeloperoxidase and a halide ion to generate hypochlorite, which is extremely toxic to organisms. Azurophilic granules contain lytic enzymes, including acid hydrolase, lysozyme, and myeloperoxidase. Specific granules contain lysozyme and lactoferrin. Movement and fusion of granules is dependent on the microtubular system. The combination of these aerobic and anaerobic neutrophil bactericidal systems effects destruction of the bacterium.
a phagocytic vacuole that ultimately fuses with specific and azurophilic granules. Destruction of microorganisms requires generation of reactive oxygen molecules through a phagocytic cell respiratory burst. These oxygen radicals interact with myeloperoxidase and a halide ion to generate hypochlorite, which is extremely toxic to organisms. Azurophilic granules contain lytic enzymes, including acid hydrolase, lysozyme, and myeloperoxidase. Specific granules contain lysozyme and lactoferrin. Movement and fusion of granules is dependent on the microtubular system. The combination of these aerobic and anaerobic neutrophil bactericidal systems effects destruction of the bacterium.
|
Complement Proteins
The complement system consists of more than 20 native serum proteins, 12 of which act sequentially to amplify the effects of antibody-mediated immunity, cell-mediated immunity, and phagocytosis, and to generate elements of the inflammatory response (Fig. 15-3) (7). This system is one of the major effector arms in host defense. It can effect a primitive nonantigen-specific defense against invading microorganisms, but it can also be triggered by antibody to provide antigen-specific host defense. The molecular weights of the 12 serum proteins that act sequentially in the complement cascade vary from 24,000 to 400,000 kd. Their serum concentrations vary from about 150 mg/dL to nanogram amounts. The gene encoding properdin is on the short arm of the X chromosome, but the genes encoding all the other complement proteins are on autosomal chromosomes.
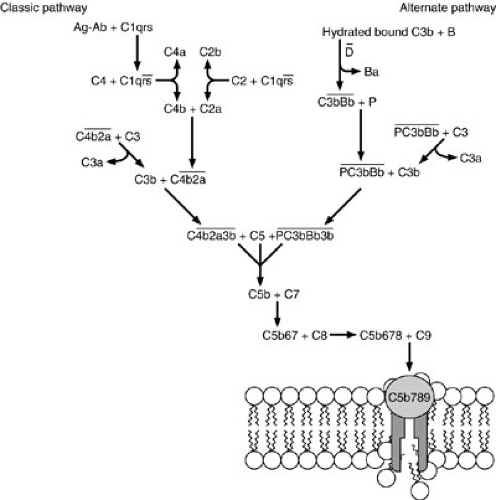 FIGURE 15-3. Classic and alternative pathways of complement activation, showing the central role of C3. Cleavage fragments are indicated by letters after the component numbers. |
Three major known functions of the complement system are to (1) cause lysis of organisms and cells; (2) promote opsonization of particles by coating them with peptides recognized by specific receptors on phagocytic cells; and (3) generate inflammatory peptides, which cause smooth muscle dilatation, immigration of inflammatory cells (chemotaxis), and histamine release. Two major pathways of complement activation are known. The more primitive pathway, termed the alternative pathway because of its discovery after the original pathway, is important as a first line of defense against invasion. This pathway functions in the absence of antibody, but it can also function more rapidly in the presence of antibody.
PRIMARY IMMUNODEFICIENCY DISEASES
Primary, or genetically determined, immunodeficiencies can involve any component of the immune system. They are considered to be rare, but the true incidences and prevalences of these diseases are unknown because there is no screening for any of these defects at any time of life, anywhere in the world. It is likely that they are far more common than appreciated. More than 100 immunodeficiency syndromes have been described since the mid-1950s (8,9,10). Until the mid-1990s, there was little insight into the fundamental problems underlying a majority of these conditions. It is impressive that the underlying genetic defects have now been identified in more than 50 of these diseases
(9). Most are recessive traits, some of which are caused by mutations in genes on the X chromosome and others by mutations on autosomal chromosomes.
(9). Most are recessive traits, some of which are caused by mutations in genes on the X chromosome and others by mutations on autosomal chromosomes.
Defects Resulting in Antibody Deficiency
Defects affecting antibody production occur more often than any other type. Selective absence of IgA is the most common, with a reported incidence ranging from 1 in 333 to 1 in 16,000 people in different races.
X-linked (Bruton) Agammaglobulinemia
Most boys afflicted with X-linked agammaglobulinemia (XLA) are well during the first 6 to 9 months of life owing to protection by maternally transmitted IgG antibodies (8). Thereafter, they frequently develop infections with high-grade extracellular pyogenic organisms, such as pneumococci, streptococci, and Haemophilus sp, unless given antibiotics or immunoglobulin therapy. Mycoplasma infections are also frequent. Chronic fungal infections are not usually present, and Pneumocystis carinii pneumonia does not usually occur unless there is an associated neutropenia. Viral infections are self-limiting, except for hepatitis viruses and enteroviruses. The latter susceptibility suggests a primary role for secretory IgA antibody in host defense against hepatitis and enteroviruses because normal T-cell function has been present in all XLA patients with persistent enterovirus infections reported thus far.
The diagnosis of XLA is confirmed when serum concentrations of IgG, IgA, IgM, and IgE are very low and there is a family history of similarly affected lateral maternal male relatives (8). Tests for antibodies to blood group substances and to diphtheria, tetanus, H. influenzae, or pneumococcal vaccine antigens are useful in distinguishing this disorder from transient hypogammaglobulinemia of infancy. Hypoplasia of adenoids, tonsils, and peripheral lymph nodes is the rule; germinal centers are not found, and plasma cells are rare. Pre-B cells are present in the bone marrow, but blood B lymphocytes are usually absent. The abnormal gene in XLA was mapped to q22 on the long arm of the X chromosome and found to encode a B-cell protein-tyrosine kinase, now named Bruton tyrosine kinase (BTK) in honor of the discoverer of this condition (11). Carriers can be detected by the finding of nonrandom X-chromosome inactivation in B cells or by mutation analysis (11).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree