Food Allergy
Maria Garcia-Lloret
Food allergies, defined as adverse immune responses to food proteins, are an increasingly common concern in the pediatric age group. Food allergy is not one disease, but a spectrum of clinicopathological disorders.1 As such, its manifestations differ significantly, depending on the immune mechanism involved and the affected target organ, ranging from the prototypical acute urticaria/angioedema to chronic conditions such as eczema or failure to thrive. Currently, there are no tests that can reliably predict the severity of a food allergic reaction, which may vary with similar exposures and even in the same individual. As a whole, fatalities are rare, but they do occur. Teenagers are particularly vulnerable because they undertake unnecessary risks and may ignore the warming signs of an impending severe reaction.
Because a diagnosis of food allergy entails a considerable nutritional and social burden for the affected children and their families, all efforts should be geared to ensure that a true food hypersensitivity is the cause of the patient’s complaints. This is not an easy task, given the protean clinical manifestations of these disorders and the recognized pitfalls of the routine laboratory tests. In some instances, a double-blind placebo controlled food challenge (DBPCFC) may be necessary. This costly and at times cumbersome procedure is at present the only gold standard for the diagnosis of food allergy.
Food allergies are often the first step in what has been termed the atopic march, involving the sequential development of different allergic diseases in the same child.2 While many children will outgrow their food allergies before their sixth birthday, for others this will remain a lifelong concern. For the vast majority of food hypersensitivities, there are presently no curative treatments. Current management of these conditions relies on careful avoidance of the offending food(s) and initiating therapy to curtail symptoms in case of accidental exposures. Yet, reinstructing the immune system to tolerate food allergens is an attainable goal, as demonstrated by the success of allergen-specific immunotherapy in the treatment of respiratory allergies. Growing evidence from a number of clinical trials suggests that the same can be achieved in food-allergic patients, which could radically change the way in which these patients are managed in the near future.3
EPIDEMIOLOGY
Food allergies are far more prevalent in developed countries than in the developing world. In the United States, the overall prevalence of food allergy has been estimated in 3.5% of the general population, with roughly twice as many children than adults afflicted by these disorders.4 Like other allergic diseases, food allergies appear to be on the rise. The prevalence of peanut allergy, for instance, has more than doubled in the last 10 years, both in the United States and in Great Britain.5 The interaction of genetic, dietary, and environmental factors appears central to the recent increase in food allergies.
Food allergies have a strong genetic component. Studies in twins show that 7% of dizygotic and 64% of monozygotic twins share a peanut allergy, and siblings from a peanut-allergic child are 6 times more likely to develop this condition. Susceptibility to peanut allergy has also been linked to HLA class II polymorphisms.1,6
Dietary customs may influence sensitization to food allergens, although there is no consensus regarding the impact that the timing of introduction of particular food into the child’s diet and its amount have on the immune responses that lead to overt clinical allergy. Significant differences in the prevalence of peanut allergy have been reported in countries with a similar consumption of peanuts, such as Israel and Great Britain (0.04% and 5%, respectively). In addition, geographical variations in the cooking or processing of foods can affect their allergenic properties: roasting peanuts (customary in the United States) tends to increase their allergenicity and boiling or frying decreases it.5,7
Nonoral exposure to food allergens can result in sensitization. In particular, low-dose antigen exposure through the skin has been shown to promote systemic IgE responses in several animal models. The rise in the prevalence of peanut allergy has been linked to the widespread usage of peanut oil–containing emollients in the treatment of common dermatological problems of infancy (ie, eczema, diaper rash). These findings suggest that food allergies could develop through casual skin contact in children with preexisting inflammatory conditions of the skin.7
Environmental and lifestyle factors also affect the development of food allergies. The former would fall under the umbrella of the “hygiene hypothesis” that links the paucity of infections during childhood in the developed world to an increased risk of allergy. Conversely, the higher number of infections and/or higher levels of endotoxin exposure in children from rural or developing communities seems to protect against the development of atopic diseases, including food allergies.
IMMUNOPATHOGENESIS
Food allergy can be viewed as a breach in the normal tolerance to innocuous food antigens. The fetus and the young infant may be “physiologically” predisposed to develop allergic responses in that their immune responses are geared toward the TH2 phenotype. A switch toward TH1 predominance occurs shortly after birth, driven in part by the bacterial colonization of the gut and the skin as well by the exposure to nutrients and other immune regulatory elements present in the maternal milk. Infants who develop atopic disease show early consolidation of TH2 20 responses to allergens, often in the first year of life before disease is manifest.1,6
The importance of commensal bacteria in the generation of oral tolerance is controvesial. Initial observations that tolerance to orally administered antigens cannot be induced in mice reared under germ-free conditions were exciting but further work suggests that some8,9 breast milk is also an important source of immunomodulatory factors, including cytokines and immunoglobulins. Transforming growth factor-β is multifunctional cytokine that has been linked to protection against allergy in infancy in several epidemiological studies.10,11 Transforming growth factor-β may promote tolerance by favoring the induction of regulatory T cells (Treg), which are central to the maintenance of immune tolerance. Development of a key subset of Treg cells, the CD4+CD25+ T cells, is critically dependent on the expression of the transcription factor Foxp3. Males with mutations in the FOXP3 gene exhibit a disease of immune dysregulation, polyendocrinopathy, enteropathy, a complex syndrome accompanied autoimmunity and severe food allergies.12 Males with milder (hypomorphic) FOXP3 mutations may only present with severe food allergies that persist into adulthood.
At the gastrointestinal level, factors such as the incomplete digestion of protein antigens and the increased permeability of the intestinal mucosa that allows the passage of larger antigenic loads have been proposed to explain the increased predisposition of young infants to develop food allergies. The low levels of secretory IgA typical in infancy may also play a role because IgA is thought to promote immune exclusion by entrapping dietary antigens and microorganisms in the mucus.
CLINICAL MANIFESTATIONS
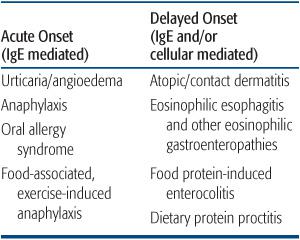
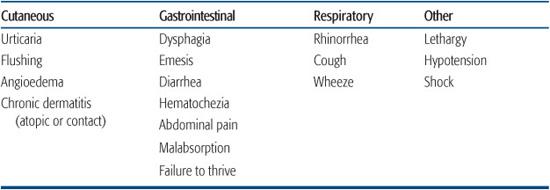
Food allergies can be grouped in two general categories: IgE-mediated and non-IgE-mediated (Table 194-1). IgE-mediated reactions are typically of rapid onset with clinical symptoms usually developing within minutes to a few hours of the ingestion of the offending food. These reactions exhibit the characteristic pattern of a type I hypersensitivity and are set in motion by the release of mast cell granules triggered by the cross-linking of IgE and its receptors by the food antigen. Skin manifestations (ie, flushing, urticaria, and/or angioedema) occur in more than two thirds of affected children, but some may present with respiratory or gastrointestinal manifestations exclusively (Table 194-2).4 Mild to moderate angioedema is not unusual. Progression to full-blown anaphylaxis with cardiovascular collapse may occur. An IgE-triggered reaction in the oral mucosa underlies oral allergy syndrome, in which subjects complain of pruritus of the mouth and/or throat when they eat raw vegetables and fruits. This syndrome develops exclusively in the older child or teenager with prior pollen sensitization and is due to heat-labile cross-reactive antigens present in plant and food13 antigens. Usually, these foods are well tolerated when cooked.
In non-IgE-mediated food hypersensitivity reactions, T-cell–mediated mechanisms provide the predominant pathogenic stimulus that drives the clinical manifestations. In some of these, there may be findings of concomitant sensitization (ie, detection of food-specific IgE), but symptoms of a type I hypersensitivity reaction as such are usually absent. A T-cell–mediated delayed type of hypersensitivity (type IV) has been suggested in this group supported by the positive patch test reaction to foods described in some patients with atopic dermatitis as well in patients with eosinophilic esophagitis.14,15
Food allergy has been implicated in the development of atopic dermatitis in the pediatric age group. Infants and young children may exhibit worsening symptoms when exposed to particular foods, either directly or through breast milk. Flares can occur within hours or days of exposure to these foods. The rash usually improves following an elimination of known food allergens. However, there is little evidence that use of exclusion diet in unselected individuals with atopic eczema is useful.16,17
The clinical phenotypes of allergic gastrointestinal disorders are discussed further in Chapter 411. Food protein–induced enterocolitis (FPIES) is a disease of infancy, typically triggered by the ingestion of whole cow’s milk–based formulas, although it can also be triggered by soy and solid food proteins. FPIES affects both the small bowel and the colon, with diffuse inflammation of the gastrointestinal tract, mild villous atrophy, and crypt abscesses. Clinically, it is characterized by profuse vomiting and diarrhea that develops within a few hours of ingestion of the allergenic food and may progress to dehydration and shock in 20% of patients.15
In contrast to FPIES, which is a severe disorder that requires prompt diagnosis management, infants with dietary protein proctocolitis usually look well and thrive but have presenting symptoms of the passage of frequent mucusy and sometimes overtly bloody stools.19,20 Some infants with allergic proctocolitis are breast-fed and become sensitized as a result of maternally ingested protein excreted in breast milk. Cow’s milk and soy products are the most commonly implicated allergens, and elimination of these foods from the child’s or the maternal diet usually results in the resolution of the symptoms.14,15
Table 194-1. Food Hypersensitivity Disorders
Eosinophilic esophagitis is typically a disease of the older child or adolescent, but symptoms may develop as early as the second or third year of life. Complaints of recurrent vomiting and abdominal pain are common. Coughing and choking can also be found in the initial stages progressing to overt dysphagia and, in the most severe cases, food impaction. Many of these children have concomitant symptoms of environmental allergies.15 Food allergy, particularly cow’s milk allergy, has been shown to manifest with constipation in some children but the prevalence of this disorder is unclear.21
DIAGNOSIS
The importance of a thorough clinical history in the diagnosis of food allergy cannot be overstated. Children may present with a variety of complaints that mimic other common pediatric disorders, and the diagnosis of a food allergy can be overlooked. Conversely, patients or caretakers may see food as a causal factor in situations where the likelihood of a true food allergy is very low. Non-immune–mediated adverse food reactions (ie, digestive enzyme deficiencies, GI infections, food aversions, etc) are common in the pediatric age group and often can be excluded on the basis of the clinical history alone.
A history of prior exposure is not required because allergic reactions to foods have been reported to occur at the first apparent ingestion in more than 50% of cases.1,4,6 Factors such as the amount and the type of food ingested as well as the time to the development of the symptoms need to be taken into consideration. A detailed food diary may help clarify these factors as well as provide an assessment of potential nutritional deficiencies.
The diagnostic workup of a suspected IgE-mediated food allergy includes a skin prick test and/or the measurement of serum food-specific IgE antibodies. Both modalities have similar sensitivity, estimated to be over 85%. Specificity, however, falls below the 40%, mostly due to the fact that they are both a measure of allergic sensitization, which may or may not correlate with clinical allergy. This can only be assessed by means of an oral challenge, which continues to be the gold standard for the diagnosis of food allergies. These tests are routinely performed safely in the allergists office.22,23 Serological tests also provide a reproducible quantitative assessment of allergic sensitization. For some common food allergens, studies have suggested cut-off values for serum food-specific IgE antibodies that can predict the likelihood of developing a systemic allergic reactions in any particular patient.24,25
Table 194-2. Clinical Manifestations of Food Hypersensitivity
While immediate type clinical reactions to food can quite easily be identified by the patient’s history or measurement of specific IgE in combination with positive oral food challenges, non-IgE and or delayed reactions still present diagnostic difficulties. In vitro determinations of allergen-specific T-cell responses, while scientifically sound, are still at the investigational level. The atopy patch test with foods represents another potential avenue, but its use is still nonstandarized and is not routinely performed in most centers.25
THERAPY
Current management of food allergies relies on the careful elimination of the offending food from the diet and the prompt institution of therapeutic measures to stall the development of severe reactions in case of accidental exposures. The latter measures are only pertinent to the IgE-mediated reactions, because non-IgE-mediated reactions rarely evolve into acute life-threatening conditions.
Elimination of offending foods from the diet sounds like a trivial exercise but due to often surprising uses of various food products in industrial food preparation processes, and problems of cross-contamination in food-processing facilities, this can be very challenging. Instruction about the fastidious avoidance of specific allergens by reading of labels (note that the components of commercial food products often changes without notice) often requires education of the parents by a dietitian or other provider with specific expertise in appropriate counseling of families. In children elimination diets carry a risk of inducing malnutrition with poor growth, due to the elimination of essential nutrients. In cases of multiple food allergy, monitoring by an experienced pediatric dietitian to assure adequate nutrient intake is useful.26
All patients with a history of a systemic reaction to foods should be prescribed self-injectable epinephrine with a formulation appropriate for the patient’s age and instructed on its use. This drug should be employed promptly in the case of an impending anaphylactic reaction. Patients should carry a dose of it at all times. For young children, parents and other caretakers should be provided with written instructions as to when and how to administer the drug. Milder reactions, defined as those involving exclusively the skin or the gastrointestinal system, can be managed with oral antihistaminics. The indication of glucocorticoids for the treatment of acute food allergic reactions is controversial. Although steroids are usually prescribed in the urgent care setting, there is no consistent evidence that they prevent the development of late-phase reactions and should not be routinely indicated.
The use of oral immunotherapy for desensitization to food allergens is currently an area of intense investigation.3 Although promising, the safety and effectiveness of this approach is yet to be established in infants and children.
REFERENCES
See references on DVD.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


