Cryptosporidiosis
Luis F. Barroso II and Richard L. Guerrant
Cryptosporidium species are tiny (2–6 μm), obligate intracellular parasites related to other coccidian protozoan, including Toxoplasma, Cyclospora, Isospora, Plasmodium, Eimeria, and Sarcocystis. Cryptosporidium species primarily infect the gastrointestinal tract of a variety of vertebrate hosts, including humans.1 Host range is largely a function of species, as a given species of parasite most efficiently maintains infection within a few species of hosts.
Over 20 species of Cryptosporidium have been described, but there is some debate as to the exact number of species.2 Two species, C hominis and C parvum, account for nearly all of the disease caused by Cryptosporidium species in humans and are the species of public health significance. C parvum was formerly divided into two genotypes (genotypes I and II), but current classification separates genotype I into a separate species known as C hominis.3
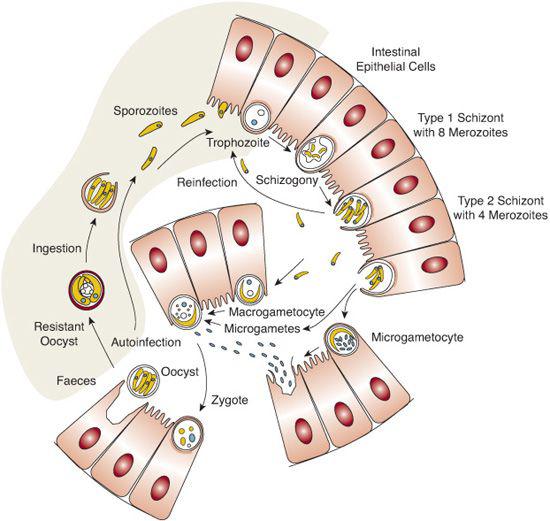
Cryptosporidium completes its life cycle within a single host1,11 (Fig. 345-1). Infection occurs after ingesting the sporulated, thick-walled oocysts. Excystation occurs in the small intestine after exposure to bile salts and pancreatic enzymes, releasing four sporozoites. These sporozoites penetrate a surface epithelial cell in the intestinal mucosa and form an intracellular parasitophorous vacuole. They then differentiate into uninuclear trophozoites, which undergo asexual replication (merogony) to form type I meronts. The type I meront can then autoinfect other surface epithelial cells or differentiate into a type II meront. The type II meront then undergoes gametogomy, producing both microgametocytes and macrogametocytes. These gametocytes fertilize to produce oocysts. The life cycle is complete when the oocysts undergo sporogomy, resulting in infectious sporozoites within the oocysts. Approximately 80% of the oocysts produced in this fashion are environmentally resistant, thick-walled cysts that are excreted in the feces. The remaining 20% are thin-walled cysts that undergo another autoinfective stage. The autoinfectious stages are important features in the parasite’s life cycle and accounts for persistent and occasionally severe disease, even when a low inoculum of cysts are ingested.
 EPIDEMIOLOGY
EPIDEMIOLOGY
Cryptosporidia are ubiquitous in the environment and are found in several sources of untreated surface water. During the AIDS epidemic, cryptosporidia were recognized as a significant enteric pathogen, causing severe disease in patients with advanced AIDS. Currently, studies show that the incidence of AIDS-associated cryptosporidiosis has declined with the advent of highly active antiretroviral therapy (HAART).12,13
The prevalence of disease is higher in developing countries due to less sanitary conditions that promote fecal-oral transmission and the lack of safe water sources. The seroprevalence in developing countries is as high as 75% by age 4, in contrast to the United States, where seroprevalence is approximately 15%.14 Cryptosporidiosis is more common in children than adults. Daycare attendance is a risk factor, and outbreaks have been reported in childcare settings, likely through secondary person-to-person transmission.15,16 The principal mode of transmission is fecal-oral.
The oocyst is particularly resistant to disinfectants, including chlorine, bleach, ethanol, and many other hospital, industrial, and household chemicals.1,11,17 However, 6% hydrogen peroxide is an effective cysticide. Cryptosporidium is resistant to standard chlorine concentrations in public water systems, and it often escapes filtration systems. The largest waterborne outbreak occurred in 1993 in Milwaukee, Wisconsin, with approximately 400,000 people infected and 69 deaths.18
 PATHOPHYSIOLOGY
PATHOPHYSIOLOGY
In the immunocompetent host, Cryptosporidium primarily infects the proximal small bowel. The exact mechanism whereby Cryptosporidium causes diarrhea is unknown. The organism disrupts epithelial tight junctions in several in vitro epithelial cell models,21 resulting in a loss of barrier function. Experiments have failed to reveal any exotoxin or virulence factor produced by Cryptosporidium, although the organism clearly causes malabsorption. Pathological findings include loss of intestinal epithelium, villous atrophy with loss of epithelial architecture and surface area, and infiltration of the lamina propria with mononuclear and polymorphonuclear cells.11 Various stages of the parasite can be seen immediately below the brush border of epithelial cells in biopsy samples.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


