11.1 The newborn infant
stabilization and examination
Dr Neil Campbell began this Chapter in the fifth edition of this book, thus:
Neonatal transition
Circulation
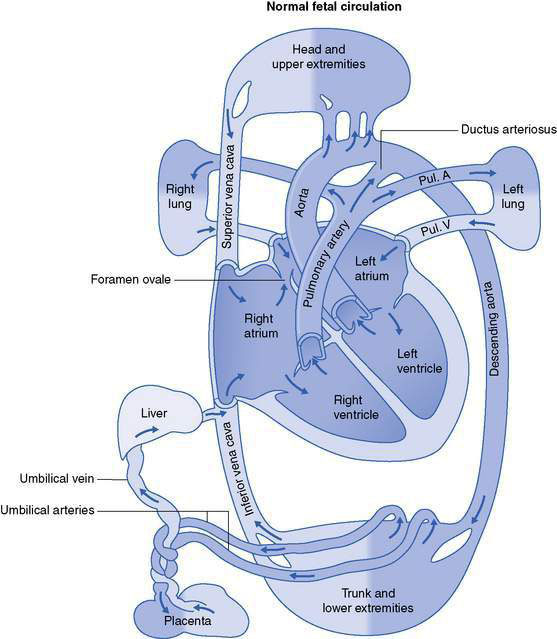
In utero there is high pulmonary vascular resistance such that only 10–15% of the cardiac output goes through the pulmonary circulation. Most of the cardiac output bypasses the lungs by flowing right-to-left across the foramen ovale or through the ductus arteriosus (Fig. 11.1.1). With the infant’s first breath the pulmonary vascular resistance falls and blood flows to the lungs; with cord clamping the peripheral vascular resistance rises and the foramen ovale is kept shut; and with the rise in partial pressure of oxygen (Pao2) and withdrawal of prostaglandins produced by the fetoplacental unit, the ductus closes. In some babies with persistent pulmonary hypertension, these changes do not occur and there continues to be a right-to-left shunt at the atrium and ductus. Such infants are tachypnoeic and remain cyanosed.
Respiration
The fetal lungs are filled with fluid that has been secreted by the pulmonary epithelium. There is a net outward movement of this fluid into the amniotic fluid with breathing movements during fetal life. Hormonal changes, including a rise in catecholamines, occurring with the onset of labour, lead to the reabsorption of some of this fluid from the alveolar sacs. Most remaining fluid is squeezed out of the lungs during passage of the chest through the birth canal (and can be seen as clear fluid around the nose and mouth at birth). With normal chest recoil the infant’s lungs fill with air, surfactant is released from the type II pneumocytes, lowering surface tension in the alveoli, and the residual lung volume is established with the first few breaths. Infants born by caesarean section, prior to labour, are more likely to have retained lung fluid (transient tachypnoea of the newborn; see Chapter 11.3).
Temperature control
Newborn infants have a larger surface area compared to their weight than do adults and can become cold rapidly. They are wet at birth and lose heat through water evaporation, as well as via radiation, conduction and convection if not clothed. After birth, hormonal changes lead to heat production from non-shivering thermogenesis in the brown adipose tissue. Core temperature is normally maintained at 36.5–37°C. Hypothermia will increase oxygen consumption required for basal metabolism and is a not uncommon cause of tachypnoea (see Chapter 11.3). Hypothermia may also occur in conditions such as sepsis.
Metabolism
The fetus is dependent on the maternal supply of glucose via the placenta, and glycogen stores are laid down in the liver, muscle and heart as gestation increases. At birth the maternal glucose supply ceases and the infant’s glucose levels fall over the next 1–2 hours before hormonal mechanisms bring about a rise from mobilized stores. Infants who have delay in feeding, preterm, growth retarded and sick infants, as well as infants of diabetic mothers, are all at increased risk of significant hypoglycaemia (see Chapter 11.2).
Gastrointestinal
The fetus swallows amniotic fluid, which is rich in growth factors. After birth, coordination of sucking and swallowing is readily established on day 1, and healthy term infants demand to feed from the breast eight or more times a day. Meconium should be passed by all infants by 48 hours of age. Before birth, unconjugated bilirubin is excreted by the placenta. During the transition to hepatic conjugation and excretion of bilirubin, all infants have raised serum bilirubin levels to some degree (see Chapter 11.2). All newborn infants have low levels of vitamin K-dependent clotting factors. Intrinsic vitamin K production follows bacterial colonization of the gut. This occurs in the first few days, but the vitamin K deficiency state carries with it a risk of haemorrhage (see below).
Neonatal stabilization and resuscitation
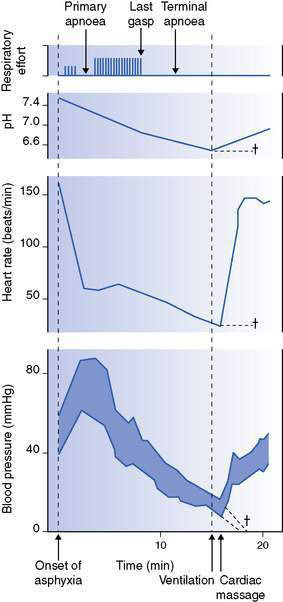
Animal experiments carried out in the 1960s looked at the effects of acute, total asphyxia on heart rate (HR) and breathing (Fig. 11.1.2). At delivery by caesarean section (simply to control the situation), air breathing was prevented totally by occlusion of the airway. There was an initial period of gasping followed by cessation of breathing (primary apnoea), then a further period of gasping and finally no breathing (terminal apnoea).
Apgar scores
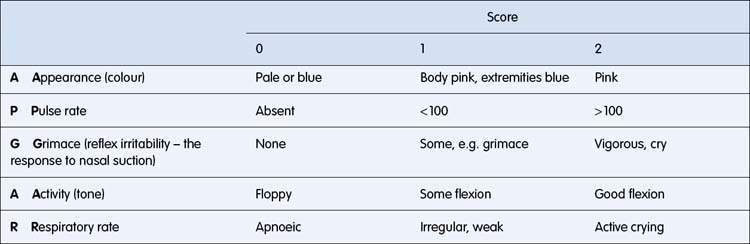
Since the 1950s the Apgar score (Table 11.1.1) has been used to assess the infant’s condition at birth and to differentiate those who are vigorous or depressed. Many health jurisdictions require the Apgar score at 1 and 5 minutes to be recorded.
There are many problems with the Apgar score:
• Different observers will record slightly different scores.
• There are clearly many routes to a score of 5 or 6 (for example), and both this and the infant’s condition should be fully described.
• Vigorous infants should not be suctioned (as this may lead to vagus-induced bradycardia or even vocal cord occlusion) and so a score of 2 is awarded for ‘reflex irritability’ by inference. Such infants will all have scores of 9–10.
• The score is much less satisfactory in very preterm or ventilated infants.
• For an individual infant there is little correlation between the Apgar score and pH or lactate values, and unfortunately neither is good at predicting the long-term outcome.
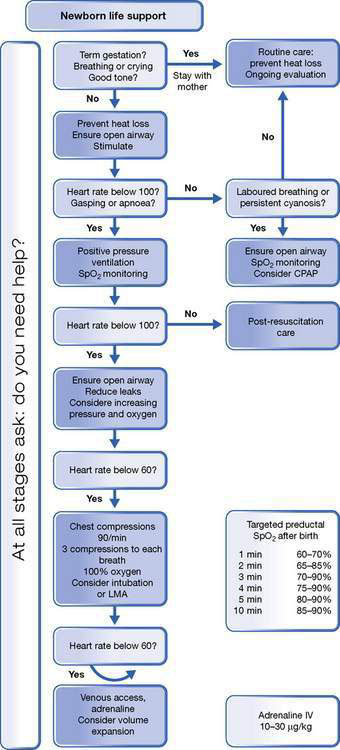
Basic care and stabilization (Fig. 11.1.3)
• Check the maternal and obstetric history.
• Check the equipment: infant overhead warmer, air and oxygen supply, suction apparatus, self-inflating bag and appropriately sized masks and/or pressure device and T-piece, intubation equipment, umbilical catheter, drugs.
• Start stopwatch when infant’s body is free from the mother.
• Assess the infant rapidly, particularly tone, breathing and HR (use stethoscope on chest):
1. The infant is vigorous and crying. Leave alone (but dry and wrap, or place in skin to skin contact with mother).
2. Infant cyanosed, irregular respirations, HR > 100. Gentle stimulation, check head in neutral position (avoid neck flexion and hyperextension) to open the airway, gentle oropharyngeal suction if obvious obstruction. Most respond.
3. Still inadequate respirations or apnoea, or HR < 100: (30 seconds from birth). Three to five slow (3 seconds) breaths then bag and mask at 40–60 per minute. Have pop-off valve or manometer set at 30 cmH2O, but lower pressures are probably adequate. Check the response: should be visible chest movement and increase in HR.
4. HR < 60 and not increasing, inadequate respirations or apnoea. Proceed to ADVANCED resuscitation. Call for help. Intubate, if skilled. Otherwise continue with bag and mask, check head in neutral position with jaw thrust, check chest movement and air entry. Give 3 cardiac compressions (see below) to 1 breath. Consider drugs: IV adrenaline (epinephrine).
Studies have shown the appropriate technique to achieve the most effective use of a face mask during neonatal resuscitation (Fig. 11.1.4).
• Hold the newborn head in a neutral position (A).
• Gently roll the mask on to the face from the tip of the chin (B, C).
• Hold the mask with thumb and index finger at the top where the silicone is thickest (D).
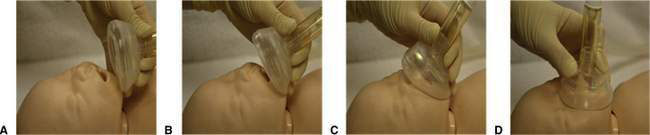
Fig. 11.1.4 Technique for using face mask in neonatal resuscitation.
(From Schilleman K, Witlox RS, Lopriore E et al 2010 Leak and obstruction with mask ventilation during simulated neonatal resuscitation. Arch Dis Child Fetal Neonatal Ed 2010;95:F398–F402, with permission.)
• With the thumb and index finger apply even downward pressure on top of the mask (D).
• Do not let the fingers encroach on to the skirt of the mask.
• The other fingers perform a chin lift with upward pressure.
Additional notes
Chest compressions
• It is essential that chest compressions follow a period of adequate lung inflation – which is generally the most effective.
• Chest compressions must not divert attention from ongoing lung inflation, or compromise adequate ventilation.
• The correct method is with hands encircling the chest and thumbs on the lower third of the sternum, compressing about one-third of the depth of the chest.
• One large survey of over 30 000 births suggested that only around 1 per 1000 infants ‘need’ cardiac compressions. It is likely that chest compressions are overused.