Ritual
Subject
Instrument
Sign
Sorry cut
Men
Knife
Laceration of arms, thighs
Women
Stone, club (nulla nulla), stick
Scalp laceration, scar, scarring alopecia
Teenage girls
Knife
Laceration to hips and calves
Nice marks
Prepubertal and teenage girls
Glowing twigs, cigarettes
Burns on dorsal hands and forearms
Ceremonial
Various
Piercings
Nasal septum, earlobes
Burning leaves
Posterior shoulder burns, scars
Environment
Plants and insects can cause persistent papular eruptions, local and systematized allergic reactions due to contact, foreign body reactions, toxins, and hypersensitivity.
Betel nut and tobacco chewing and kava ingestion may begin in adolescence
Water exposures may lead to contact hypersensitivity, toxic reactions, infestations, and infections.
Plant and insect exposure can result in solitary, clustered, or patterned papular, urticarial or vesiculobullous changes and may subsequently result in secondary infection or allergic reactions that may be localized or generalized (Fig. 48.1). Common Australian pathogenic insects include midges (sandflies), mosquitoes, ticks, fleas, mites, black flies, march flies, leeches, blister beetles, and ants.

Fig. 48.1
Insect bites presenting as (a) clustered papules in exposed skin of a 9 month old boy and (b) as an isolated resolving bullous lesion in a boy in urban south-eastern Australia (Royal Children’s Hospital Melbourne)
Bindii (Jo Jo) dermatitis results from impregnation of the spine of the bindii seed (Soliva pterosperma) into the skin, usually of palms, soles, elbows, knees, and buttocks of young boys during spring and summer [18]. Allergic and possibly foreign body reactions result in 1–2 mm red micropapules and papulopustules that may initially be itchy or tender, but often remain relatively asymptomatic for months, resolving towards winter and not responding to standard eczema treatments [18].
Other hairy plants that cause reactions include prickly pear, Hibiscus, Malachra fasciata, and Cionachne cyathapoda [17]. Plant contact allergy and toxic reactions (e.g. to latex from the Mangrove plant) are also more common in indigenous children, possibly because of increased exposure [19]. Caterpillar hair dermatitis may be underdiagnosed in indigenous communities, characterized by recurrent papulourticarial reactions relating to airborne spread of hair. An indoor outbreak has been reported in Australia originating from an infested Eucalyptus tree [17, 20].
Older adolescents may adopt adult practices like betel nut and tobacco chewing and kava ingestion (kava is a sedative drink produced from the roots of the plant Piper methysticum). Betel nut and tobacco chewing can cause psychoactive and autonomic effects, gum and teeth staining, malignancies (oropharyngeal and pancreatic), peptic ulcer, and heart disease. Kava has been associated with a cephalocaudal progressive ichthyosis, facial edema, and hepatotoxicity [21].
Water hazards include coral dermatitis, starfish and sea-urchin toxic dermatitis, and reactions from fish and marine sponges. Cercarial dermatitis results from cutaneous penetration by avian or non-avian schistosomes (e.g. Austrobilharzia terrigalensis , A. variglandis), and it is characterized by severe itch of exposed skin. This contrasts with the pruritic papules under clothing that characterizes seabather’s eruption from the nematocysts of larvae of cnidarians (sea anemones and thimble jellyfish) and also seaweed dermatitis.
Climate can adversely affect the skin of indigenous people preferentially living in harsh rural terrains. Miliaria is common in infants due to heat. Lip biting and chewing (triggered by mucosal dryness) is common in young males [17]. Heat, humidity, and possibly hair structure predisposes to trichomycosis axillaris from Corynebacteria in young indigenous adults of both sexes. Proximal axillary (and rarely pubic hair) develop yellow, occasionally red and uncommonly black, concretions that may be associated with malodor and hair brittleness. Wood’s lamp examination may fluoresce yellow or blue-white. Differentiation from lice and powder can also be made by demonstration of Gram-positive bacilli and by incubation on blood agar at 37 °C after immersion of hair in 70 % alcohol [17].
Nutrition
Urbanization, the dislocation of resettlement, and poor dietary awareness have led to increased obesity in indigenous compared with nonindigenous Australians. A study of rural indigenous children showed the prevalence of overweight children being 22.1 % and 20.7 % for girls and boys, respectively, with corresponding figures for obesity of 5.1 and 5.8 % [22]. Pseudoacanthosis nigricans is therefore common, presenting with velvety pigmentation of the nape of neck and axillae (Fig. 48.2). Skin tags may subsequently occur. Underlying conditions such as polycystic ovarian syndrome and diabetes (which is increasing in indigenous children in Australia and the USA) should also be considered [23].

Fig 48.2
An 11 year old boy from south-eastern urban Australia with obesity, early pseudoacanthosis nigricans, severe combined immunodeficiency and pityriasis rosea, 9 years after stem cell transplant (Royal Children’s Hospital, Melbourne)
Infection
Infectious diseases represent the overwhelming burden of skin morbidity in which indigenous children are overrepresented.
Scabies and Pyoderma
Epidemiology/Demographics
In some indigenous Australian communities, scabies prevalence is 30–50 % of children and that of pyoderma/impetigo is 10–70 %
64 % of children presenting with scabies have concurrent bacterial infection
The endemic, concurrent, recurrent, and complicated nature of infections have led to community-specific management protocols
High rates of Staphylococcus aureus skin and soft tissue infections—and particularly methicillin-resistant Staphylococcus aureus (MRSA)—have been documented in indigenous people from Australia, Canada, USA, and New Zealand [24–30]. Increased rates of diabetes and renal disease may predispose to invasive staphylococcal disease. Scabies frequently coexists with and also predisposes to these bacterial skin infections.
Scabies has a prevalence of 30–50 % in some communities of Aboriginal Australian children and it underlies 70 % of pyogenic skin infections [31–33] (Fig. 48.3). Pyoderma/impetigo occurs in 10–70 % of Aboriginal children in remote communities [7, 31]. Group A Streptococcus pyogenes (GAS) is found in 80–93 % of the swabs for impetigo [7, 31]. Of note, whereas Group C and Group G Streptococcus can also cause similar skin and soft tissue infections and are more frequently isolated in the throat than are GAS, they are rarely identified in skin sores and usually then only together with GAS or Staphylococcus aureus [34] (Figs. 48.4 and 48.5). Staphylococcus aureus (including community-acquired MRSA) is increasing in incidence in more affluent urban areas, where Staphylococcus aureus prevalence now approximates GAS amongst indigenous Australians [35, 36].
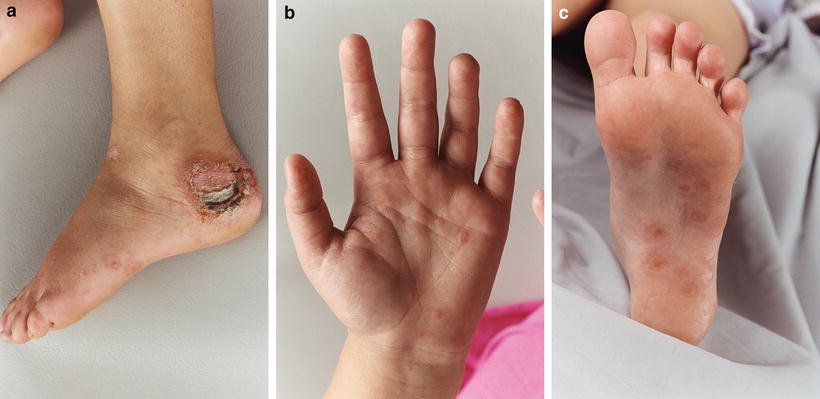

Fig. 48.3
Concurrent scabetic, Streptococcus pyogenes and Staphylococcus aureus infection, (a) before and (b) after treatment with cotrimoxazole twice-daily over 3 days in an 11 year old boy living in a 3 bedroom home with nine family members in Northern Australia (credit: Dr Asha Bowen)

Fig. 48.4
(a) A scalp sore in a 2 year old girl living in a 4 bedroom house with 11 relatives in remote Northern Australia. Group C streptococcus and Staphylococcus aureus were identified. (b) After treatment with intramuscular benzathine penicillin G (credit: Dr Asha Bowen)

Fig. 48.5
(a) Group G Streptococcus and Staph aureus complicating tinea pedis in an 8 year old girl from urban south-east Australia, leading to an erythema multiforme like id response of (b) palms and (c) soles (Royal Children’s Hospital, Melbourne)
Of children affected by scabies, 10 % develop scabies by 6 weeks of age, 50 % by 6 months, and 68 % by 1 year [7]. Similarly, 10 % of children with impetigo have been infected before 9 weeks of age [7]. The ubiquity of pyoderma in indigenous Australian communities previously raised the defined threshold of pyoderma treatment to ‘six infected sores’, but now treatment is recommended for all sores [37]. 64 % of children presenting with scabies have concurrent bacterial infection [7].
Clinical Presentation
Concurrence of scabies, streptococcal, and staphylococcal infection is common
Atypical presentations of scabies may result from effects of tropical climate, immunosuppression, and chronic eczematization
Group A Streptococcus is the most important cause of pyoderma and can lead to rheumatic fever, acute poststreptococcal glomerulonephritis (APSGN), and acute renal failure
Scabies in indigenous children show typical scabetic burrows, pruritic papules, nodules, and a propensity for finger webs, flexor aspects of wrists, extensor aspects of elbows, periumbilical skin, buttocks, ankles, male genitalia, and female periareolar regions. Infants are often affected on the scalp, neck, axillae, and groin. However, burrows are often not visible for children living in tropical locations [38]. Furthermore, chronic eczematization and immunosuppression can lead to modified and atypical presentations. Crusted scabies is particularly seen in the immunosuppressed (due to primary immunodeficiency, immunosuppressive medications, and HIV or HTLV-1 infection) as well as in children with leprosy. Scabies acquired from animals with mange are rare and self-limiting due to host-specificity of mites. Fissuring predisposes to secondary infection with GAS and S. aureus, leading to impetigo and cellulitis. Head lice infestation and tinea also predispose to skin GAS infection in indigenous children [39].
Differential diagnoses include bites (midges, fleas, bedbugs), bacterial infections (folliculitis, impetigo), tinea, viral exanthems, and papular urticaria.
Treatment
Scabies
An overview of the management of scabies in indigenous Australian children is outlined in Table 48.2. Sensitivity in communicating with families is critical to optimize compliance, which is often poor in indigenous communities (Appendix). A 5 % permethrin is usually the first-line treatment, applied for 8–12 h overnight. A second 8–12 h application after 1 or 2 weeks is particularly important for children [40]. Clothing and other possible fomites should be hot cycle washed or removed from body contact for >72 h. Concurrent treatment of household contacts is critical. Under 2 months of age, many practitioners use crotamiton or 5–10 % sulfur in paraffin, given the limited data on permethrin safety.
Table 48.2
Treatment approach to scabies in indigenous Australian pediatric population (aged over 6 months)
First line therapy | 5 % permethrin creama | Apply to whole body overnight (sparing head, except in infants) and repeat application in 1 week |
|---|---|---|
First line therapy | Treat family and close contacts | Utilize topical permethrin application as above |
First line therapy | Treat clothing, bedding | Hot wash clothing and bedding or remove from body contact for >72 h |
Second line therapyb | Oral ivermectin | 200 μg/kg orally on two occasions, 1 week apart |
Adjunctive therapy | Moderately potent corticosteroid | For post-scabetic pruritus which can persist for 2–8 weeks after eradication therapy |
Ivermectin is an alternative treatment. Although licensed in France, it is not licensed for routine use in many countries (e.g. Australia, Canada, UK) and does not work on all stages of the mite cycle. Increasing in vitro resistance of Sarcoptes scabiei to permethrin and ivermectin has been reported [41, 42]. Clinical resistance should be particularly suspected if symptoms persist for several weeks in nonendemic areas. For crusted scabies, up to seven doses of ivermectin combined with topical therapy (permethrin and keratolytics) may be needed to eradicate the mites [43].
Streptococcus and Staphylococcus
Intramuscular benzathine penicillin is the treatment of choice in GAS endemic communities due to poor compliance with oral therapy and the imperative to treat GAS to prevent serious sequelae, despite potential staphylococcal antibiotic resistance. Given low rates of indigenous throat GAS carriage, pyoderma is considered responsible for most cases of rheumatic fever as well as APSGN in Aboriginal children. Oral phenoxymethylpenicillin can be considered if compliance is good. Antistaphylococcal beta-lactams are used in areas with staphylococcal predominance. Alternative oral regimens may be indicated in the case of antibiotic resistance and penicillin allergy (Fig. 48.6). Topical antibiotics are not used in endemic areas.
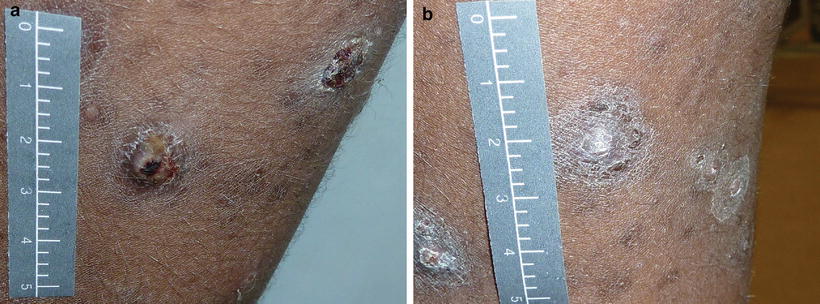

Fig. 48.6
Sores of an 8 year old girl from Central Australia infected with scabies, head lice, tinea and Strep pyogenes. She shared a two bedroom house with nine relatives. Treated with cotrimoxazole daily for 5 days and photographed (a) day 0 and (b) day 7 (credit: Dr Asha Bowen)
Boils (furuncles) are usually related to S. aureus and are best treated with incision and drainage. Flucloxacillin is used additionally in children (older adults with obesity and liver disease may have an increased risk of hepatotoxic complications). Nasal carriage is treated in chronic S. aureus carriers who experience recurrent infections, using 5–10 days of topical nasal mupirocin. Persistent reinfection will require determination of sensitivities and possibly repeated nasal mupirocin treatment or consideration of oral rifampicin. Bleach baths are an affordable and effective alternative therapy for persistent reinfection.
Cellulitis in Aboriginal communities is usually treated with procaine penicillin and elevation of the affected limb, although better S. aureus cover is required in urban communities [40]. Melioidosis and sporotrichosis also cause cellulitis and may require exclusion in atypical, refractory disease and particularly in adults with risk factors like diabetes, renal failure, and alcohol abuse.
Pyoderma prevention requires address of housing, education, and lifestyle problems. Introduction of swimming pools into communities has helped reduce infection rates, likely by reducing skin bacteria counts [44].
Efficacy of Treatment
A recent study in North Australia combined annual skin checks over a 3-year period with treatment of identified scabies cases and their household contacts with 5 % permethrin (or 10 % crotamiton for infants under 2 months) and treatment of impetigo (according to current guidelines) [37]. The study reported a 40 % drop in the rates of pyoderma especially in 3–14 year olds but no change in overall scabies prevalence, given poor treatment compliance.
In endemic communities, compliance with treatment is especially low amongst contacts, males, and in high scabies burden households [45]. Reasons cited for noncompliance include perceptions that treatment is of low priority, a fixed belief that treatment is only necessary for the index case and that topical therapy is too uncomfortable to apply.
Other Infections
Mycobacterium
Increased prevalence of tuberculosis, relative to the nonindigenous population, has been identified in indigenous people from Canada, Australia, New Zealand, and USA [46]. Cutaneous tuberculosis (TB), however, is an uncommon presentation of extrapulmonary TB and may only be found in 1–2 % of cases. There are a wide variety of clinical presentations of cutaneous TB. The presentation in skin relates to whether skin involvement is due to exogenous infection (primary inoculation into skin) or endogenous infection (due to the spread of TB within an individual). Furthermore, the immune response to TB can precipitate skin changes known as tuberculids.
The crude incidence rate of TB in the Australian indigenous population is 6.6/100,000. This is seven times that of the nonindigenous population and there are even higher rates in rural regions (13-fold in the Northern Territory) [47]. Similar increased relative risk is found in the New Zealand and US indigenous population, whereas the indigenous population of Canada carry a 35.2-fold increased risk compared to their nonindigenous counterparts. Australian Aboriginal children also carry a higher risk than non-Aboriginal children, but the incidence is much lower than adults. This suggests that differing environmental exposures (overcrowded housing, prisons) may contribute to the overall increased risk in Aboriginal Australians.
Unlike TB, a recent study in the Northern Territory did not find increased atypical mycobacterial disease in Aboriginal groups [48]. In this study, all cases of atypical mycobacterial infection in Aboriginal children presented with lymphadenitis in children under 10 years of age. Risk factors for skin infection with M. fortuitum, M. chelonae, M. abscessus, M. marinum and M. ulcerans in indigenous adults include HIV, chronic lung disease, smoking and alcoholism. Whereas M. scrofulaceum was previously the main cause of non-TB pediatric lymphadenitis in Aboriginal children, mycobacterium avium complex now accounts for 50 % of cases. Surgical excision alone is usually successful. However, tuberculous lymphadenitis is six times more common than non-tuberculous lymphadenitis, so unless proven otherwise, treatment must be directed towards TB.
Mycobacterium ulcerans, sometimes termed ‘Daintree ulcer’ or ‘Bairnsdale ulcer’ in Australia, is endemic in some regions of Queensland and Southeastern Australia and can present as a firm, painless nodule which breaks down to form a painless necrotic ulcer. The incidence of infection is increasing in Australia, though this likely reflects improved diagnosis [49]. M. ulcerans is best treated with surgical excision, with consideration of antibiotic therapy (with rifampicin and amikacin) for extensive disease.
Leprosy is caused by the obligate intracellular pathogen Mycobacterium leprae and is transmitted through close contact. Leprosy is a chronic granulomatous condition that primarily affects skin, mucous membranes, peripheral nerves, eyes, and testes. In the majority of patients, hypoesthesia is the first sign of disease, particularly of the extremities. Leprosy was not endemic to Australia prior to European settlement; however, the introduction of the disease in the late 1800s caused an epidemic across Northern Australia. Prevalence in some Aboriginal communities was determined to be 10 % in the 1950s with an overall prevalence of 270 per 100,000 indigenous Australians [50]. By 1997 the prevalence had fallen to 4/100,000 and it is now rare in Australia. Fifty-four patients were reported with leprosy in Australia between 2005 and 2010; however, indigenous patients represented only a minority, with the majority of patients being migrants from leprosy-endemic countries [51, 52]. Indigenous patients do continue to present with leprosy to healthcare practitioners in rare instances and clinicians must remain alert to the disease [53].
Melioidosis
Melioidosis is caused by the Gram-negative bacterium Burkholderia pseudomallei and is endemic to Northern Australia and parts of South East Asia. Sporadic reports of disease have also come from Central and South America, the Middle East, and Africa. 80 % of Thai children have antibodies by age 4, and a 3-year-old Australian case has been reported [54]. Soil or surface water is the usual source, although animals can also be infected and transmit disease by contact. The incidence in indigenous Australians is 25.5/100,000, four times the average north Australian incidence [55]. Other risk factors include diabetes mellitus, renal disease, and alcoholism, thus most cases occur in adults. About 80 % of cases occur during the wet season. 50 % of patients present with pneumonia, 6 % with osteomyelitis, and 6 % with skin and soft tissue infection (furuncles, cellulitis). Genitourinary, neurological, and gastrointestinal systems can also be affected. Chronic cases can mimic tuberculosis. Mortality in one series of 47 cases (including adults) was 21 %. Treatment includes ceftazidime and meropenem and must be started early.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


