Renal Tumors
Renal tumors are the second most common abdominal tumor seen in infants and children behind neuroblastoma. They represent a wide spectrum from benign to extremely malignant tumors (Box 65-1). These tumors include: Wilms tumor (WT) (also referred to as nephroblastoma or renal embryoma), renal cell carcinoma (RCC), clear cell sarcoma of the kidney (CCSK), rhabdoid tumor of the kidney (RTK), congenital mesoblastic nephroma (CMN), cystic renal tumor, and angiomyolipoma. Advances in the management of these tumors have been significant over the past six decades since Sidney Farber first administered dactinomycin (Actinomycin D) for advanced-stage WTs.1 Multidisciplinary and multi-institutional prospective randomized cooperative group trials in both North America and Europe by the Children’s Oncology Group (COG-formerly the National Wilms Tumor Study Group [NWTSG]) and the Société Internationale d’Oncologie Pédiatrique (SIOP) have produced a large body of evidence-based knowledge to establish the optimal risk-based treatments (Table 65-1). The goal of this chapter is to describe the early history of WT, followed by a discussion of the etiologic factors in renal tumor formation, molecular genetics, pathologic subtypes and premalignant syndromes, and current treatment algorithms for children.
Wilms Tumor
History
The first descriptions of WT have been variably attributed to either Rance in 1814 or Wilms in 1899.2–4 The first known specimen of this tumor was preserved by the British surgeon John Hunter between 1763 and 1793.5 This specimen of a bilateral tumor in a young infant remains in the Hunterian Museum of the Royal College of Surgeons in London. Carl Max Wilhelm Wilms was a German pathologist and surgeon. Wilms’ name became indelibly linked to this tumor in children after publication of his comprehensive monograph in 1899 titled ‘Die Mischgeschwülste der Niere’ which described seven children with nephroblastoma as part of a monograph on ‘mixed tumors of the kidney’.3,4 In it he proposed that tumor cells originate during the development of the embryo. The first successful nephrectomy was probably performed by Thomas Jessop at the General Infirmary in Leeds, England on the 7th of June, 1877 on a 2-year-old child with hematuria and a tumor of the kidney.6,7
Resection was recognized early as an effective treatment for WT and remains as the cornerstone of all therapies. However, at the beginning of the 20th century, survival was approximately 5% and operative mortality was high. In 1916 radiation therapy was added as an adjuvant therapy by Friedlander.8 In the 1930s, Ladd and Gross described the principles of operative therapy for WT, including transperitoneal exposure and early ligation of the renal pedicle.7,9 They stressed the need to remove the perirenal fat to resect lymphatic extensions and to avoid rupture of the renal capsule, principles we continue to follow today. From 1931 to 1939, survival from resection alone, involving ligation of the renal pedicle before removal, was 32% at the Children’s Hospital in Boston.1,10 Gross and Neuhauser later proposed the routine addition of abdominal radiation to WT therapy and reported an estimated 47% cure.11 Thus in 1940,1 most patients began to receive postoperative irradiation to the renal fossa. This decreased the local recurrence rate, but did not significantly affect the incidence of pulmonary metastases or improve the long-term survival. Under Gross’s tutelage, pediatric surgeons in North America generally performed primary resection of WT, while in Europe, the Paris school led by Schweisguth and Bamberger reported early success with preoperative irradiation, establishing a precedent for initial adjuvant therapy.12
WT was the first malignancy in which the importance of adjuvant treatment was recognized. This principle was espoused by Sidney Farber decades before it would be applied to other pediatric and adult solid tumors.1 The concept of adjuvant therapy ‘was based upon the supposition that in the children with WT who died, the tumor must have metastasized already at the time of discovery of the primary tumor’, although no evidence of spread was available. Dactinomycin was the first active agent identified for the treatment of WT. Of the 53 patients who had no demonstrable metastases on admission treated with combined regimen of operation, local radiation, and dactinomycin from 1957 to 1964, an 89% 2-year disease-free survival was reported,1 a very reasonable rate of survival even today. In patients with metastases identified at presentation, 18 of 31 (58%) were alive and free of disease more than 2 years later. In the early 1960s, vincristine sulfate was identified as an active agent against WT and was added to the standard therapy.13
Epidemiology
WT is the most frequent tumor of the kidney in infants and children. Its incidence is 7.6 cases for every million children younger than 15 years, or one case per 10,000 infants.14 This translates into 600 to 650 cases a year in North America. It is less common in East Asian populations than in white children, but is more frequent in black children.15,16 The mean age at diagnosis is 36 months, with most children presenting between the ages of 12 and 48 months. Tumors tend to occur earlier in boys than girls. WT incidence decreases over the age of 10 and is less common under 6 months of age. However, it still constitutes 20% of all renal tumors in children less than 6 months. Bilateral Wilms tumors (BWT) occur in 4–13% of patients.15,17
WT occurs in several well-described syndromes. These comprise about 10% of all WT cases, and include sporadic aniridia, isolated hemihypertrophy, the Denys–Drash syndrome (nephropathy, renal failure, male pseudohermaphroditism, and WT), genital anomalies, Beckwith–Wiedemann syndrome (BWS; visceromegaly, macroglossia, omphalocele, and hyperinsulinemic hypoglycemia in infancy), and the WAGR complex (WT with aniridia, genitourinary malformations, and mental retardation). These congenital syndromes have helped identify the genetic and etiological mechanisms inherent in developing a WT. For example, children with WAGR syndrome, which is associated with a chromosomal defect in 11p13, are at a 30% higher risk of developing WT than a normal child. Aniridia is usually diagnosed at birth and helps identify these children.18 BWS is a second common syndrome associated with WT. BWS affects 1 in 14,000 children. These children are at an increased risk of several types of embryonal tumors, including WT. The most frequently observed tumors in BWS are WT and hepatoblastoma, which comprise 43% and 12% of reported associated cancers, respectively.19,20 The risk for malignant development is greatest in the first decade of life. Three large studies of children with BWS reported tumor frequencies of 7.1% (13/183), 7.5% (29/388), and 14% (22/159).19,21–23
Molecular Genetics of Wilms Tumor
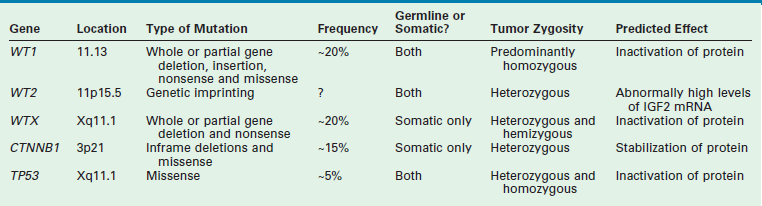
WT is an embryonal tumor that was originally thought to fit the model proposed by Knudson’s ‘two-hit’ hypothesis for cancer development.24 However, the genesis of WT has been shown to be far more complex. Multiple mutated WT genes have been identified as well as areas of loss of genetic material and allelic uniqueness (loss of heterozygosity) that are important to tumor development. A few of these genetic anomalies have been evaluated in clinical trials to assess their prognostic significance and are now being used in conjunction with traditional factors, such as stage, to determine the intensity of therapy.19,25 A detailed discussion of all the genetic anomalies associated with WT is beyond the scope of this chapter; however, a summary of the key genes and genetic anomalies is presented in Table 65-2.
TP53
TP53 mutations in WT are almost exclusively found in tumors with anaplastic histology. Seventy-five per cent of anaplastic WT have p53 mutations.26,27 Interestingly, some tumors can contain both anaplastic and favorable histology. Studies in these tumors demonstrate that the p53 mutation is found only in the areas of anaplasia and not in the area of favorable histology.26 This implies that p53 mutations may be essential for anaplastic progression.
CTNNB1
CTNNB1 plays a central role in the Wnt signal transduction pathway. The Wnt signaling pathway describes a network of proteins known for their roles in embryogenesis and cancer. Deregulation of CTNNB1 has been linked to several malignancies. CTNNB1 mutations have been reported to occur in 15% of WT.28,29 The mutations resulted in areas of phosphorylation and degradation of beta catenin, which is highly correlated with WT1 mutation and the WTX gene.
WTX
The WTX gene (also known as AMER1 for adenomatous polyposis coli (APC) membrane recruitment 1) was found to be mutated in 15 of 51 (29%) WT tested, making it the most common known gene mutation in WT.30 Localization of WTX on the X-chromosome results in complete inactivation by a single mutational event in males, and in females if the active X-chromosome is affected. The exact clinical impact of WTX on WT development remains to be defined.
WT1 (11p13)
WT1 gene was the first gene to be linked with WT development. Evidence suggests that WT1 may act as a tumor suppressor gene or oncogene and is located at chromosome 11p13.31,32 Wild-type WT1 is important for normal cell development and survival. WT1 is involved in cell growth, differentiation, and apoptosis.33 Patients heterozygous for WT1 germline mutations are predisposed to WT, and WT1 is inactivated in tumors. This implies the loss of WT1 function is associated with enhanced cell viability and/or proliferation. Ablation of WT1 at the initial stages of kidney development results in apoptosis and renal agenesis, indicating that it has a crucial role in maintaining cell viability. In some leukemias, the increased expression of WT1 compared with normal bone marrow cells, along with some reports of WT1 expression being a marker of poor prognosis, suggest that WT1 functions as an oncogene.26 By contrast, observations of WT1 inactivating mutations in leukemias suggest it functions as a tumor suppressor gene. It should be emphasized that children with congenital syndromes and WT make up a very small proportion of the total WT population. However, loss of heterozygosity (LOH) is seen in 30–40% of WT patients in the region of WT1. Based on this observation, it would be expected that many patients with sporadic WT would have a mutation in the WT1 gene. Surprisingly, the incidence of mutations of WT1 associated with WT in the sporadic form of the disease is low (10–20%).34,35
Loss of Heterozygosity
LOH refers to loss of genetic material and allelic uniqueness. A major aim of the fifth National Wilms Tumor Study (NWTS-5) was to determine if tumor-specific LOH for chromosomes 11p, 1p, or 16q was associated with an adverse prognosis for children with favorable-histology (FH) WT, a finding suggested in earlier retrospective studies.25,36,37 The results of NWTS-5 demonstrated that outcomes for patients with LOH at 1p and 16q were at least 10% worse than those without LOH (Fig. 65-1). These findings were used as determinants for the intensity of therapy on the current COG renal tumor studies.
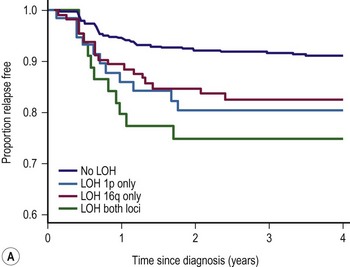
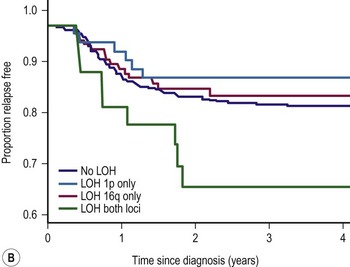
FIGURE 65-1 (A) Relapse-free survival with loss of heterozygosity (LOH) at chromosomes 1p and 16q for patients with stage I/II Wilms tumor of favorable histology. (B) Relapse-free survival for patients with stage III/IV Wilms tumor with favorable histology and LOH at chromosome 1p and 16q. (From Grundy PE, Breslow N, Li S. Loss of heterozygosity for chromosomes 1p and 16q is an adverse prognostic factor in favorable-histology Wilms’ tumor: A report from the National Wilms’ Tumor Study Group. J Clin Oncol 2005;23:7312–21, with permission from the American Society of Clinical Oncology.)
Pathologic Precursors: Nephrogenic Rests, Nephroblastomatosis, and Multicystic Dysplastic Kidneys
Nephrogenesis in the normal kidney is usually complete by 34 to 36 weeks gestation. The presence of nephrogenic rests (NRs; persistent metanephric tissue in the kidney after the 36th week of gestation) has been associated with the occurrence of WT. NR are considered precursor lesions to WT. However, only a small number develop clonal transformation resulting in the development of WT. The presence of multiple or diffuse NRs is termed nephroblastomatosis.
These NRs may occur in a perilobar (PLNRs) or intralobar (ILNRs) location, and may be single or multiple (Fig. 65-2). In children with aniridia or the Denys–Drash syndrome, the lesions are primarily ILNRs, whereas children with hemihypertrophy or the BWS have predominantly PLNRs (Table 65-3).38 NRs may be further classified by their growth phase, which has been separated into three phases; (1) incipient or dormant nephrogenic rests which show few well-formed tubular structures, but no evidence of proliferation and no mitoses; (2) hyperplastic nephrogenic rests which are composed of epithelial elements with nodular expansive growth; and (3) sclerosing rests which consist of stromal and epithelial elements with few blastemal nephrogenic elements. Most NRs are dormant or in the sclerosing phase, and the majority will spontaneously resolve. Hyperplastic NR can produce masses as large as conventional WT and can present a diagnostic dilemma. NRs are rare in the general population. In an autopsy series of infants younger than 3 months, nine of 1,035 infants (0.87%) had PLNRs, while ILNRs occurred in only 2 of 2000 cases (0.1%).39
TABLE 65-3
Association of Nephrogenic Rests with Wilms Tumor Predisposition Syndromes and Congenital Anomalies
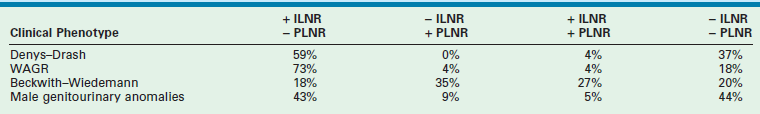
ILNR, intralobar nephrogenic rests; PLNR, perilobar nephrogenic rests; +, present; −, absent.
From Breslow NE, Beckwith JB, Perlman EJ, et al. Age distributions, birth weights, nephrogenic rests, and heterogeneity in the pathogenesis of Wilms tumor. Pediatr Blood Cancer 2006;47:260–7.
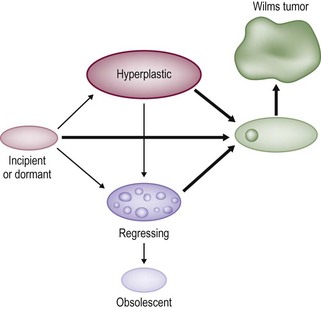
FIGURE 65-2 Diagrammatic depiction of nephrogenic rests and their classification. Thick arrows indicate tumor induction. (From Beckwith JB. Precursor lesions of Wilms’ tumor: Clinical and biological implications. Med Pediatr Oncol 1993;21:158–68.)
Pathologic distinction between NR and WT can be very difficult. In fact, it may be impossible to distinguish a hyperplastic NR from a WT based on an incisional or needle biopsy specimen that does not include the margin between the NR and the remaining kidney.38,40 Most hyperplastic nodules lack a pseudocapsule at their perpiphery, whereas most WTs will have one. Thus, if the biopsy specimen does not contain the lesion and its margin, it will be difficult to differentiate between these two lesions. In these situations, the current recommendation is to use the term ‘nephrogenic process, consistent with a WT or a nephrogenic rest’.40,41 This is one reason that it is recommended to avoid percutaneous biopsies in children with renal masses.
The incidence of NRs is about 100 times greater than that of WT (1/10,000 infants). In a review of cases of WT reported in the NWTS-4, 41% of the unilateral WTs were associated with NRs, whereas in children with synchronous bilateral WT, the incidence of NRs was 99%.38 These were primarily PLNRs. A child with a WT and NRs in the resected specimen is at increased risk of developing a metachronous tumor in the contralateral kidney.42 In a child under one year of age, this risk is very significant, and these children need to be followed very carefully with sequential ultrasound (US) examinations. It is very difficult to diagnose NRs on imaging. Although magnetic resonance imaging (MRI) may be helpful, there is no gold standard.
Diffuse hyperplastic perilobar nephroblastomatosis (DHPLN) is a unique category of nephroblastomatosis in which the rests form a thick rind around the periphery of the kidney. Infants with DHPLN may initially present with large unilateral or bilateral flank masses (Fig. 65-3). A characteristic radiographic finding is massively enlarged kidneys that maintain their normal configuration and lack evidence of necrosis. As with the isolated NRs, proliferation of the thin rind of NRs on the periphery of the kidney will preserve the normal configuration of the kidney, but result in marked enlargement of its size. This is in contrast to WT, in which the normal renal configuration and collecting system are generally distorted. The optimal diagnosis and management of a child with DHPLN is unclear. In a 2006 study of 52 children and 33 cases of DHPLN, histologic examination alone was unable to establish a diagnosis in 21 children that underwent biopsy at the time of initial presentation.41 In the same study, 24 children developed WT during long-term follow-up with eight being anaplastic. Thirteen had single tumors and 11 were multiple. Treatment was variable and included surgery, radiation and chemotherapy, or expectant observation. The three patients who received no therapy developed WT 10 months after diagnosis. Chemotherapy may decrease the proliferative element in NRs, but it is unclear whether it prevents the development of malignancy. A current COG study includes a treatment protocol to address some of the questions with respect to DHPLN and its optimal therapy.
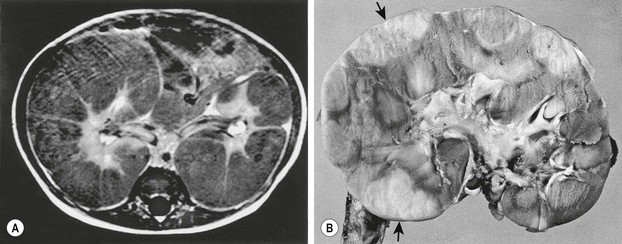
FIGURE 65-3 (A) MR image of a 10-month-old infant who presented with large bilateral flank masses demonstrates a picture characteristic of diffuse hyperplastic perilobar nephroblastomatosis (DHPLN) with extensive involvement of the entire cortex of the kidney with no evidence of necrosis and general preservation of the shape of the kidney. (B) A resected kidney with a similar pattern of DHPLN reveals extensive involvement of the periphery of the cortex by severely hypertrophied nephrogenic rests (arrows). Resection of such kidneys should be avoided because, in most cases, the hypertrophy will resolve and the kidney will have excellent preservation of its function. (B courtesy of J. B. Beckwith, by permission.)
An increased risk of WT arising in multicystic dysplastic kidneys has been suggested. This is primarily based on case reports and all children have been under the age of four. In contrast, a report describing 1041 infants and children with multicystic dysplastic kidneys found no case associated with a WT.43 The authors concluded that routine prophylactic nephrectomy was not needed in children with multicystic dysplastic kidneys. Furthermore, a review of the NWTS pathology files identified only three cases of dysplastic kidneys in more than 7,000 children with WT over a 26-year interval.44 Thus, prophylactic nephrectomy in children with multicystic dysplastic kidney for malignancy is not justified. However, screening ultrasound in these children until age 4 is suggested.43,44
Pathology
WTs are currently divided into those with ‘favorable’ histology and those with ‘unfavorable’ histology. Unfavorable histology proved to be the most important factor in patient outcome in NWTS-1, a finding that has remained consistent through the current trials.5,45 Fortunately, favorable histology comprises 90% of the tumors. These tumors are the ‘classic WT’ consisting of three elements: blastemal, stromal, and epithelial tubules (Fig. 65-4). Tumors contain various proportions of each of these elements. Triphasic are the most characteristic, but biphasic and monophasic lesions also occur.46 The proportion of these three elements in WTs have been studied, but have not been shown to predict outcomes. Under the current treatment protocols, they are not utilized to determine therapy.46,47 Abnormal mucinous or squamous epithelium, skeletal muscle, cartilage, osteoid, or fat are less frequent elements found in WT.5
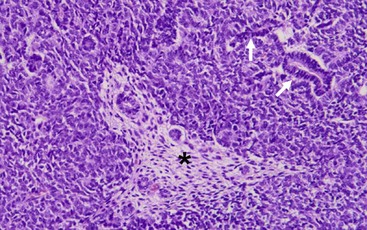
FIGURE 65-4 The classic ‘triphasic’ histologic pattern (blastemal, epithelial, and mesenchymal derivatives) of a Wilms tumor is seen on this H&E slide. There is a predominance of small undifferentiated blastemal cells in the image that surround a few neoplastic duct and tubular epithelial structures (solid arrows). In the center is an island (asterisk) of spindle-shaped fibroblastic mesenchymal cells that surround a few tubules. The neoplastic cells in the image lack the requisite features of an anaplastic variant (see Fig. 65-5).
Unfavorable tumors are those with focal or diffuse anaplasia.5,48,49 Anaplasia is defined by multipolar polyploid mitotic figures, marked nuclear enlargement (giant nuclei with diameters at least three times those of adjacent cells), and hyperchromasia (Fig. 65-5).50 Determining whether a tumor has diffuse or focal anaplasia is important for prognosis and therapy.48 Focal anaplasia is defined as the presence of one or a few sharply localized regions of anaplasia within a primary tumor, the majority of which contains no nuclear atypia. Tumor designated as diffuse anaplasia must have at least one of the following four criteria: anaplastic cells outside of the kidney, presence of anaplasia in a random kidney biopsy, anaplasia in more than one region of the kidney, or anaplasia in one region with extreme nuclear pleomorphism in another site. Anaplasia occurs primarily in children older than 2 years. In NWTS-1, 66.7% of patients with anaplasia experienced relapse and 58.3% died of their tumor.45 Of note, there was a higher frequency of relapse and death in the ‘diffuse’ subgroup. Additionally, anaplasia in extrarenal tumor sites and a predominantly blastemal tumor pattern were both adverse prognostic factors.50 Moreover, anaplasia is a marker of resistance to therapy, not of aggressive behavior.46,47,50 TP53 deletion on chromosome 17 has been associated with anaplasia. However, a recent study demonstrated recurrent anaplasia-specific genomic loss and under-expression in several additional regions, most strikingly 4q and 14q, suggesting anaplasia is not linked solely to deletions of chromosome 17, but is associated with loss at multiple loci.51
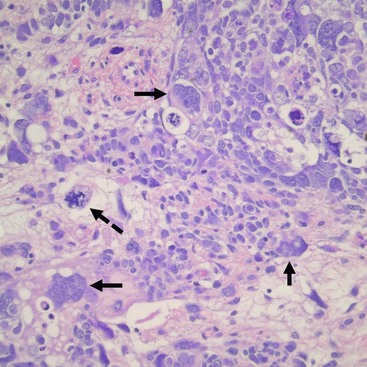
FIGURE 65-5 Salient features of the anaplastic Wilms’ tumor variant with H&E staining. Anaplasia is defined by three requisite criteria that are all depicted in the image: atypical mitotic figures (dotted arrow) (often tripolar or multipolar); nuclear enlargement (solid arrows) to greater than three times the size of resident cell nuclei of the same type; and nuclear hyperchromicity present in some of the cells. Enlargement and hyperchromicity reflect the fact that anaplastic Wilms tumors contain nearly twice the amount of DNA as normal cells with duplication of many whole chromosomes in each cell.
CCSK and malignant RTK were grouped in the initial NWTSG studies with the unfavorable histology WT. They are now considered distinct entities from WT, based on their pathologic appearance, biologic behavior, and response to different therapies.52,53 CCSK is a highly malignant tumor with an unusual proclivity for bony metastasis. It generally appears as a large unifocal and unilateral tumor with a homogeneous mucoid, tan, or gray/tan cut surface, often with foci of necrosis or prominent cysts.52 This tumor invades and surrounds the renal parenchyma rather than compresses the margin into a pseudocapsule as is seen in a WT. Its classic appearance is that of a deceptively bland tumor with uniform oval nuclei with a delicate chromatin pattern, a prominent nuclear membrane, and sparse, poorly stained vacuolated ‘water-clear’ cytoplasm with indistinct cell membranes. Although the cells generally appear in cords or nests divided by an arborizing network of vessels and supporting spindle cell septa, there are variations and nine major histologic patterns have been identified.52,53 Recently, a translocation t(10;17) and deletion (14q) have also been described in CCSK, suggesting that they may play a role in its pathogenesis.54 The cell of origin of this tumor is not known. In addition to osseous metastases, CCSKs also have a significant incidence of metastases to the brain. Long-term follow-up is important as one report found 30% of the relapses occur more than two years after diagnosis.55
Malignant RTK occur in young infants with a median age of 11 months.56 Most (85%) cases occur within the first two years of life. It is the most aggressive and lethal of all pediatric renal tumors, and fortunately only accounts for 2% of renal tumors. Grossly these tumors are unencapsulated and invasive with characteristic involvement of the perihilar renal parenchyma. Histologically, RTKs are characterized by monomorphous, discohesive, rounded to polygonal cells with acidophilic cytoplasm and eccentric nuclei containing prominent large ‘owl eye’ nucleoli reminiscent of skeletal muscle, but lacking its cytoplasmic striations, ultrastructural features, and immunochemical markers.57 A large periodic acid–Schiff (PAS)-positive hyaline cytoplasmic inclusion occurs in a variable population of tumor cells and is a hallmark of this tumor.58 Ultrastructural examination reveals parallel cytoplasmic filamentous inclusions packed in concentric whorled arrays, a distinctive feature of this tumor, which suggests a neuroectodermal origin. The tumor tends to infiltrate the adjacent renal parenchyma rather than to compress it. These tumors are notable for the occurrence of a second primary tumor in the midline of the brain, resembling medulloblastoma.59 A consistent deletion (22q11-12) has been described in both renal and extrarenal rhabdoid tumors.60,61 These deletions delineate an area of overlap at the site of the hSNF5/INI1 gene, and tumors have biallelic alterations or deletions of this gene.62–64 For all renal tumors except RTK, immunohistochemical staining for the wild type INI-1 protein shows nuclear positivity. In renal and extra renal rhabdoid tumors, this is absent.64
Staging
Two principal staging systems are used for children with WT (Table 65-4). The COG system is based on pretreatment findings prior to administration of chemotherapy or radiotherapy. Patients are given a local stage and a disease stage. The local stage defines the extent of abdominal disease, while the disease stage considers both the local extent of disease and distant metastasis. Both factors determine therapy with the use of local radiation therapy to the tumor bed based on the local stage, and the intensity of chemotherapy based on the disease stage.65
SIOP protocols generally recommend chemotherapy followed by nephrectomy, with surgicopathologic staging occurring at the time of nephrectomy. The SIOP classification was revised in 2010 based on a review of the histologic appearance of the tumors at resection (post neoadjuvant chemotherapy) and the corresponding outcomes.66,67 Tumors are now classified on SIOP protocols as completely necrotic (low-risk tumor), blastemal (high-risk tumor), and other histology (intermediate-risk tumors).
Prognostic Factors
The current prognostic factors used in COG trials are histology, stage, age, tumor weight, response to therapy and LOH at 1p and 16q. The two most important factors continue to be the histology and the stage of the tumor.25,68,69 The details and prognostic significance of tumor pathology has been previously discussed in the Pathology section. The tumor stage is determined by the results of the imaging studies, the findings at operation, and the histological findings.
Rapid response to chemotherapy is a prognostic category that is being evaluated in patients who present with stage IV disease due to lung metastasis. The goal in these patients is to avoid lung radiation. Response to therapy is also being assessed in BWTs. Also, LOH at both 1p and 16q is now being used for determination of therapy in the current COG renal tumor studies.25
Clinical Presentation and Diagnosis
The differential diagnosis for a malignant abdominal mass in a child includes WT, neuroblastoma, hepatoblastoma, rhabdomyosarcoma and lymphoma. WT is often noted during a bath or by the pediatrician at a routine visit. This is in marked contrast to neuroblastoma, which is seen in the same age group, but frequently presents with pain, often from osseous metastasis. WT also may be associated with hematuria (gross in 18.2% and microscopic in 24.5% of patients), but with a much lower frequency than is seen with RCC.16 Twenty to 25% of patients present with hypertension, and 10% with fever. Occasionally, a child may have abdominal trauma and demonstrate pain out of proportion to what is expected, and an abdominal mass is found that cannot be attributed to the trauma.
Radiographic imaging is performed to determine the anatomic location and extent of the mass. WT can extend into the renal vein (11% of cases) or the vena cava (4% of cases) (Fig. 65-6).70,71 Embolization of a caval thrombus to the pulmonary artery can be lethal, and the presence of a thrombus must be identified preoperatively to prevent this occurrence. Ultrasound has been the recommended screening study for an abdominal mass to determine its site of origin and to assess for possible intravascular or ureteral extension. However, a 2012 COG study demonstrated computed tomography (CT) can accurately identify cavo-atrial tumor thrombus as well.72 Routine Doppler ultrasound evaluation, after CT has already been performed, may not be needed. A CT/MRI scan of the abdomen will confirm a renal origin to the mass, and whether there are bilateral tumors. Historically, bilateral renal explorations were recommended by NWTSG protocols because early generations of CT scans missed 7–10% of bilateral lesions. However, with new helical CT scans only 0.25% of bilateral tumors are missed.73 Based on these results, bilateral exploration is not recommended on current protocols from the COG.
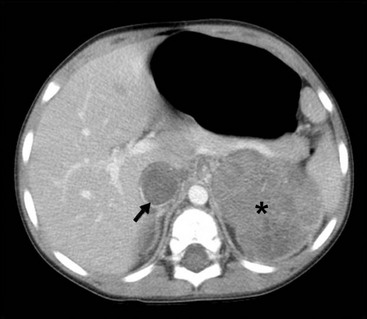
FIGURE 65-6 A transverse cut of a CT scan demonstrating a left Wilms tumor (asterisk) that extends into the inferior vena cava (arrow).
A neuroblastoma will generally indent the kidney whereas WTs arise from within the kidney and distort its internal configuration. Also, a thin lip of renal parenchyma can often be seen extending over the neoplasm in a WT, known as the claw sign (Fig. 65-7). Unfortunately, there are no characteristic radiographic findings of WTs that allow accurate diagnosis. Intra-abdominal staging has been difficult to assess radiographically unless extensive lymph node involvement or intrahepatic metastases are present. A recent study by Khanna and associates, using the COG database of 3,000 cases, found the preoperative imaging sensitivity and specificity of ultrasound in detecting WT rupture to be 53.7% and 88.4%, respectively, compared to the findings at operation.74 The common sites of metastatic spread are the lungs and the liver; hence CT scans of the chest should be included in the staging studies.
Surgery
Surgical Details
The essential tasks that are required of the surgeon, irrespective of when the WT resection occurs, are: (1) safe resection of the tumor; (2) accurate staging of the tumor; (3) avoidance of complications that ‘upstage’ the tumor (rupture or unnecessary biopsy); and (4) accurate documentation of the operative findings and details of the procedure in the operative notes. Tumor spill, failure to biopsy lymph nodes (for both unilateral and bilateral tumors), incomplete tumor removal, failure to assess for extrarenal tumor extension, and operative complications will adversely impact patient survival.75–77
Although WTs are usually initially seen as a large mass, most are resectable (Fig. 65-8). Renal tumors should be excised through an adequate subcostal or thoracoabdominal incision. Struggling through an inadequate incision will often result in tumor rupture, which increases both the stage of the tumor and the risk for intra-abdominal recurrence.77 A flank incision should not be used for resection of pediatric renal tumors because of the limited exposure it provides. Initial exploration of the abdomen includes inspection for hepatic metastases or intraperitoneal spread. The vena cava, if it is accessible, should be palpated to assess for intravascular extension of tumor. The colon is then mobilized off the anterior aspect of the kidney and the renal mass. Although early descriptions of the operative technique recommended initial control of the renal hilum, this is often not feasible with extremely large tumors and must await mobilization of the mass to allow safe exposure of the hilum. Premature attempts at vascular control, particularly for left-sided tumors, may result in ligation of the superior mesenteric artery.78 The renal mass should not be biopsied unless the decision is made to not proceed with a complete resection. Biopsy will produce contamination of the peritoneum and increase the tumor stage to stage III.
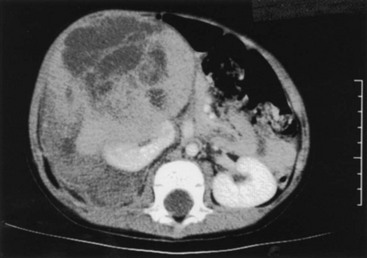
FIGURE 65-8 CT scan of a 3-year-old boy who presented with a history of abdominal pain, fever, and a right flank mass. The tumor is very large with extension outside the renal capsule into the retroperitoneal tissues posterior to the kidney. Despite its size, this lesion was able to be completely resected. Lymph nodes and specimen margins were negative for tumor. This tumor was therefore stage II, and the infant was able to avoid both anthracycline therapy and abdominal irradiation because of the complete resection.
Biopsy of lymph nodes in the renal hilum and along the vena cava and aorta is critical for adequate staging. Even in children with stage IV disease, local staging is vital because it will determine whether abdominal radiation therapy is used in children treated on COG protocols. Studies have demonstrated that the surgeon’s gross evaluation and assessment of lymph nodes does not reliably correspond with the histologic involvement of tumor in the nodes, with false-negative and false-positive rates of 31.3% and 18.1%, respectively.79 An increased incidence of local recurrence was seen in NWTS-4 children in whom biopsy of lymph nodes was not performed, particularly in stage I cases.77 This suggests that under treatment of local disease in these children, due to inadequate staging, resulted in an increased frequency of local recurrence. Although grossly involved lymph nodes are generally resected, an extensive retroperitoneal lymph node resection has not been demonstrated to improve local control.80 There has recently been increased interest in determining the extent of node dissection required for both WT and other renal tumors, although to date the data are inconclusive and retrospective.81,82 As the tumor is being mobilized, the ureter is palpated for tumor and is resected close to the bladder. Gross hematuria in children with WT is infrequent, but its occurrence suggests involvement of the renal pelvis with possible extension down into the ureter.83 Cystoscopy should be considered in these children to identify bladder involvement to avoid transection of the tumor thrombus during division of the ureter. Complete resection of the ureteral extension prevents this finding from increasing the local stage of the tumor. If the tumor involves the upper pole of the kidney, the adrenal gland is generally resected to provide adequate margins around the tumor. With lower-pole lesions, the adrenal gland may be preserved.
The factors associated with an increased risk of local recurrence include stage III disease, unfavorable histology, failure to biopsy the lymph nodes, and tumor rupture during operation.77 Tumor rupture and biopsy of the lymph nodes are the factors that the surgeon can impact. Multiple regression analysis adjusting for the combined effects of histology, lymph node involvement, and age reveal that tumor spillage remained significant and was greatest in children with stage II disease who received less intensive therapy. Most tumor ruptures occur during mobilization of the posterior aspects of the tumor where it is adherent to the diaphragm. This can be best prevented by the use of an adequate incision for exposure and resection of a segment of the adherent diaphragm if necessary. In a review of 2,000 cases of WT, size of tumor greater than 15 cm was the most significant risk for tumor rupture.84 Tumor rupture/spillage results in a stage III classification which results in additional chemotherapy and RT in a child who otherwise might have stage II disease.
‘Spill’ refers to a break in the tumor capsule during resection whether accidental, unavoidable, or by design. Spill is also considered to have occurred if the tumor is transected when the renal vein or ureter is divided. In COG protocols, a preoperative or intraoperative biopsy (needle or open) are stage III criteria. This is not true for patients on SIOP protocols where fine needle or Tru-cut biopsies are stage II criteria and incisional biopsies mean stage III. ‘Rupture’ refers to either the spontaneous or post-traumatic rupture of the tumor preoperatively with the result that tumor cells are disseminated throughout the peritoneal or retroperitoneal space.85 Blood in the peritoneal cavity may be a sign of rupture. When found, a thorough examination of the tumor surface is mandated. Rupture is also considered to have occurred if the tumor penetrates the renal capsule, with tumor extending into the peritoneal cavity. When encountered, all of these situations make the child stage III (COG and SIOP) and must be carefully documented in the operative note.
Resection of adjacent organs (liver, spleen, or pancreas) or resection of massive WT are discouraged because such extensive resections are associated with a significant increase in surgical complications.76,86 In this situation the surgeon should only sample the primary tumor.
Prior to operation, coagulation studies should be obtained because acquired von Willebrand disease (vWD) has been reported in WT and other malignancies.87,88 A single prospective study of 50 WT patients found the incidence of acquired vWD was 8%.88 However, the true incidence and prevalence is unknown. Historically, when coagulation abnormalities were identified, they were felt to be clinically insignificant.89
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree