The mother may also experience higher obstetrical morbidity and mortality rates. These also increase with the number of fetuses (Mhyre, 2012; Wen, 2004). In a study of more than 44,000 multifetal pregnancies, Walker and colleagues (2004) reported that, compared with singletons, the risks for preeclampsia, postpartum hemorrhage, and maternal death were increased twofold or more. The risk for peripartum hysterectomy is also increased, and Francois and associates (2005) reported this to be threefold for twins and 24-fold for triplets or quadruplets. Finally, these mothers are at increased risk for depression compared with women with a singleton pregnancy (Choi, 2009).
MECHANISMS OF MULTIFETAL GESTATIONS
Twin fetuses usually result from fertilization of two separate ova–dizygotic or fraternal twins. Less often, twins arise from a single fertilized ovum that divides–monozygotic or identical twins. Either or both processes may be involved in the formation of higher numbers. Quadruplets, for example, may arise from as few as one to as many as four ova.
 Dizygotic versus Monozygotic Twinning
Dizygotic versus Monozygotic Twinning
Dizygotic twins are not in a strict sense true twins because they result from the maturation and fertilization of two ova during a single ovulatory cycle. Moreover, from a genetic perspective, dizygotic twins are like any other pair of siblings. On the other hand, monozygotic or identical twins, although they have virtually the same genetic heritage, are usually not identical.
As discussed subsequently, the division of one fertilized zygote into two does not necessarily result in equal sharing of protoplasmic material. Monozygotic twins may actually be discordant for genetic mutations because of a postzygotic mutation, or may have the same genetic disease but with marked variability in expression. In female fetuses, skewed lyonization can produce differential expression of X-linked traits or diseases. Furthermore, the process of monozygotic twinning is in a sense a teratogenic event, and monozygotic twins have an increased incidence of often discordant malformations (Glinianaia, 2008). For example, in a study of 926 monozygotic twins, Pettit (2013) reported a 12-fold increase in the prevalence of congenital heart defects, but 68 percent of affected infants had a normal sibling. Accordingly, dizygotic or fraternal twins of the same sex may appear more nearly identical at birth than monozygotic twins.
Genesis of Monozygotic Twins
The developmental mechanisms underlying monozygotic twinning are poorly understood. Minor trauma to the blastocyst during assisted reproductive technology (ART) may lead to the increased incidence of monozygotic twinning observed in pregnancies conceived in this manner (Wenstrom, 1993).
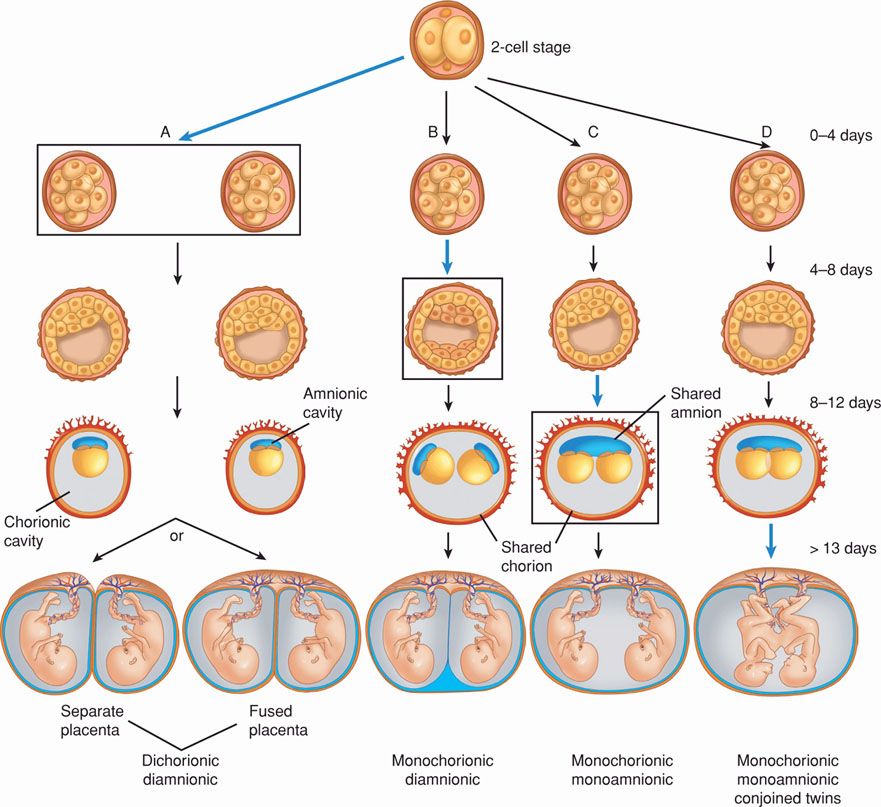
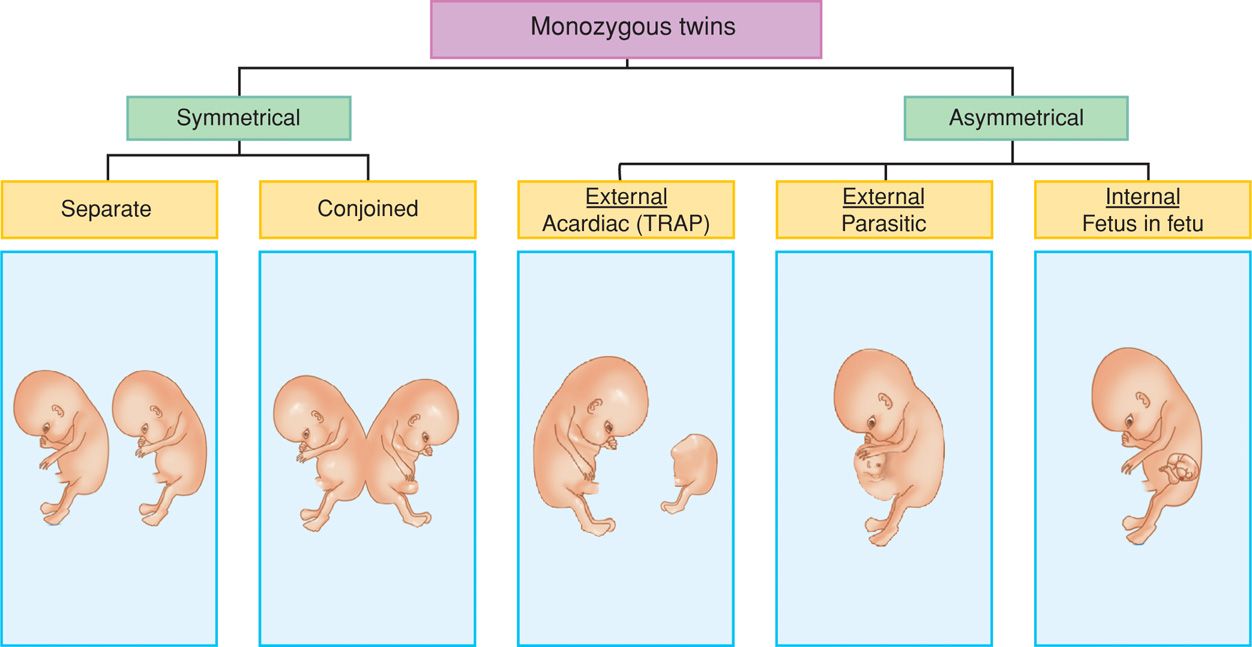
The outcome of the monozygotic twinning process depends on when division occurs. If zygotes divide within the first 72 hours after fertilization, two embryos, two amnions, and two chorions develop, and a diamnionic, dichorionic twin pregnancy evolves (Fig. 45-1). Two distinct placentas or a single, fused placenta may develop. If division occurs between the fourth and eighth day, a diamnionic, monochorionic twin pregnancy results. By approximately 8 days after fertilization, the chorion and the amnion have already differentiated, and division results in two embryos within a common amnionic sac, that is, a monoamnionic, monochorionic twin pregnancy. Conjoined twins result if twinning is initiated later.
FIGURE 45-1 Mechanism of monozygotic twinning. Black boxing and blue arrows in columns A, B, and C indicate timing of division. A. At 0 to 4 days postfertilization, an early conceptus may divide into two. Division at this early stage creates two chorions and two amnions (dichorionic, diamnionic). Placentas may be separate or fused. B. Division between 4 and 8 days leads to formation of a blastocyst with two separate embryoblasts (inner cell masses). Each embryoblast will form its own amnion within a shared chorion (monochorionic, diamnionic). C. Between 8 and 12 days, the amnion and amnionic cavity form above the germinal disc. Embryonic division leads to two embryos with a shared amnion and shared chorion (monochorionic, monoamnionic). D. Differing theories explain conjoined twin development. One describes an incomplete splitting of one embryo into two. The other describes fusion of a portion of one embryo from a monozygotic pair onto the other.
It has long been accepted that monochorionicity incontrovertibly indicated monozygosity. Rarely, however, monochorionic twins may in fact be dizygotic (Hack, 2009). Mechanisms for this are speculative, but Ekelund and coworkers (2008) found in their review of 14 such cases that nearly all have been conceived after ART procedures.
Superfetation and Superfecundation
In superfetation, an interval as long as or longer than a menstrual cycle intervenes between fertilizations. Superfetation requires ovulation and fertilization during the course of an established pregnancy, which is theoretically possible until the uterine cavity is obliterated by fusion of the decidua capsularis to the decidua parietalis. Although known to occur in mares, superfetation is not known to occur spontaneously in humans. Lantieri and colleagues (2010) reported a case after ovarian hyperstimulation and intrauterine insemination in the presence of an undiagnosed tubal pregnancy. Most authorities believe that alleged cases of human superfetation result from markedly unequal growth and development of twin fetuses with the same gestational age.
Superfecundation refers to fertilization of two ova within the same menstrual cycle but not at the same coitus, nor necessarily by sperm from the same male. An instance of superfecundation or heteropaternity, documented by Harris (1982), is demonstrated in Figure 45-2. The mother was sexually assaulted on the 10th day of her menstrual cycle and had intercourse 1 week later with her husband. She was delivered of a black neonate whose blood type was A and a white neonate whose blood type was O. The blood type of the mother and her husband was O.
 Frequency of Twinning
Frequency of Twinning
Dizygotic twinning is much more common than monozygous splitting of a single oocyte, and its incidence is influenced by race, heredity, maternal age, parity, and, especially, fertility treatment. By contrast, the frequency of monozygotic twin births is relatively constant worldwide—approximately one set per 250 births, and this incidence is generally independent of race, heredity, age, and parity. One exception is that zygotic splitting is increased following ART (Aston, 2008).
The “Vanishing Twin”
The incidence of twins in the first trimester is much greater than the incidence of twins at birth. Studies in which fetuses were evaluated with sonography in the first trimester have shown that one twin is lost or “vanishes” before the second trimester in up to 10 to 40 percent of all twin pregnancies (Brady, 2013). The incidence is higher in the setting of ART.
Monochorionic twins have a significantly greater risk of abortion than dichorionic twins (Sperling, 2006). In some cases, the entire pregnancy aborts. In many cases, however, only one fetus dies, and the remaining fetus delivers as a singleton. Undoubtedly, some threatened abortions have resulted in death and resorption of one embryo from an unrecognized twin gestation. It has been estimated that 1 in 80 births are multifetal, whereas 1 in 8 pregnancies begin multifetal followed by spontaneous reduction of one or more embryos or fetuses (Corsello, 2010).
Dickey and associates (2002) described spontaneous reduction in 709 women with a multifetal pregnancy. Before 12 weeks, one or more embryos died in 36 percent of twin pregnancies, 53 percent of triplet pregnancies, and 65 percent of quadruplet pregnancies. Interestingly, pregnancy duration and birthweight were inversely related to the initial gestational sac number regardless of the final number of fetuses at delivery. This effect was most pronounced in twins who started as quadruplets. Chasen and coworkers (2006) reported that spontaneous reduction of an in vitro fertilization (IVF) twin pregnancy to a singleton pregnancy was associated with perinatal outcomes intermediate between IVF singleton pregnancies and IVF twin pregnancies that did not undergo spontaneous reduction.
In one analysis of 41 cases of spontaneous reduction, higher values of pregnancy-associated plasma protein A (PAPP-A) and free β-human chorionic gonadotropin (β-hCG) were identified (Chasen, 2006). Gjerris and colleagues (2009) compared 56 cases of “vanishing twin” to 897 singletons after ART and did not identify any differences in first-trimester serum markers as long as the reduction was identified before 9 weeks. If diagnosed after 9 weeks, the serum markers were higher and less precise than in singleton ART gestations. Therefore, the diagnosis of vanishing twin should be excluded to avoid confusion during maternal serum screening for Down syndrome or neural-tube defects (Chap. 14, p. 283).
Factors That Influence Twinning
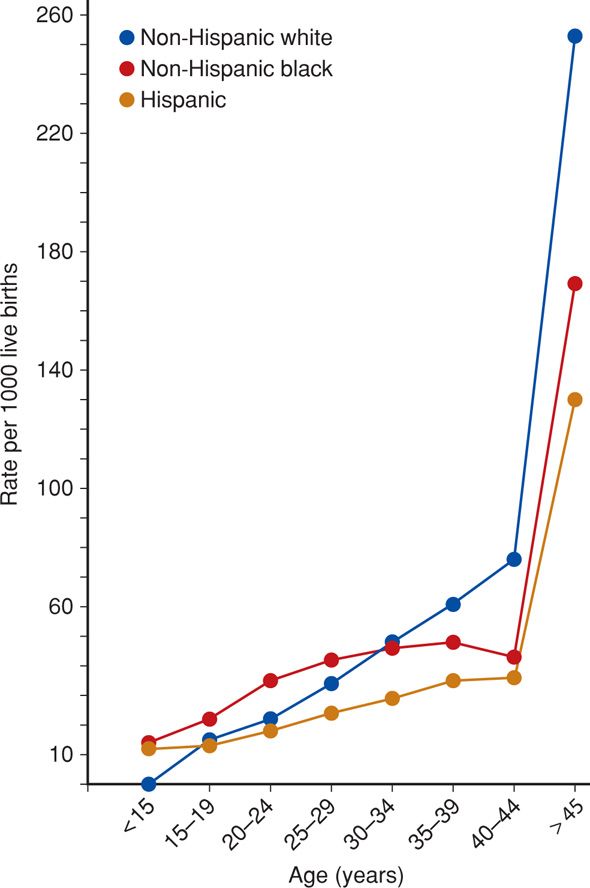
Race. The frequency of multifetal births varies significantly among different races and ethnic groups (Table 45-2). Abel and Kruger (2012) analyzed more than 8 million births in the United States between 2004 and 2008. These investigators found the highest rate of twinning in African American women (3.5 percent) and lower rates in white (3.0 percent) women. Hispanic, Asian, and Native American women had comparatively lower rates then white women. In one rural community in Nigeria, Knox and Morley (1960) found that twinning occurred once in every 20 births! These marked differences in twinning frequency may be the consequence of racial variations in levels of follicle-stimulating hormone—FSH (Nylander, 1973).
TABLE 45-2. Twinning Rates per 1000 Births by Zygosity
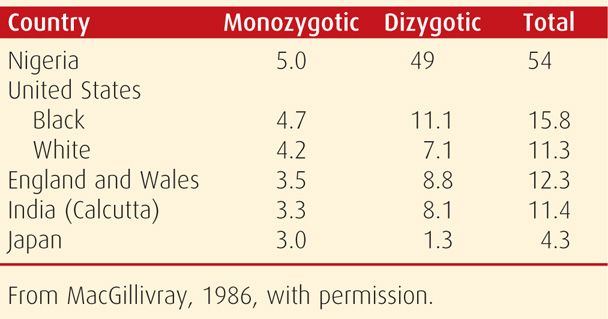
Maternal Age. As depicted in Figure 45-3, maternal age is another important risk factor for multiple births. Dizygotic twinning frequency increases almost fourfold between the ages of 15 and 37 years (Painter, 2010). It is in this age range that maximal FSH stimulation increases the rate of multiple follicles developing (Beemsterboer, 2006). The rate of twinning also increases dramatically with advancing maternal age because the use of ART is more likely in older women (Ananth, 2012). Although paternal age has been linked to frequency of twinning, its affect is felt to be small (Abel, 2012).
FIGURE 45-3 Multifetal birth rates in the United States according to maternal age and race, 2010. (Data from Martin, 2012.)
Parity. Increasing parity has been shown to independently increase the incidence of twinning in all populations studied. Antsaklis and coworkers (2013) noted a progressive increase in multiparity in twinning during a 30-year period, but cautioned that some of this increase is presumably due to ART. In a two-year study from Nigeria, where such technology is not commonly available, Olusanya (2012) calculated an eightfold increase in multiple gestations when parity was 4 or less and a 20-fold increase when parity was 5 or more compared with primiparas.
Heredity. As a determinant of twinning, the family history of the mother is more important than that of the father. In a study of 4000 genealogical records, White and Wyshak (1964) found that women who themselves were a dizygotic twin gave birth to twins at a rate of 1 set per 58 births. Women who were not a twin, but whose husbands were a dizygotic twin, gave birth to twins at a rate of 1 set per 116 pregnancies. Painter and associates (2010) performed genome-wide linkage analyses on more than 500 families of mothers of dizygotic twins and identified four potential linkage peaks. The highest peak was on the long arm of chromosome 6, with other suggestive peaks on chromosomes 7, 9, and 16. That said, the contribution of these variants to the overall incidence of twinning is likely small (Hoekstra, 2008).
Nutritional Factors. In animals, litter size increases in proportion to nutritional sufficiency. Evidence from various sources indicates that this occurs in humans as well. Nylander (1971) showed a definite increasing gradient in the twinning rate related to greater nutritional status as reflected by maternal size. Taller, heavier women had a twinning rate 25 to 30 percent greater than short, nutritionally deprived women. Evidence acquired during and after World War II showed that twinning correlated more with nutrition than body size. Widespread undernourishment in Europe during those years was associated with a marked fall in the dizygotic twinning rate (Bulmer, 1959). Several investigators have reported a 40-percent increase in the prevalence of twinning among women who have taken supplementary folic acid (Ericson, 2001; Haggarty, 2006; Hasbargen, 2000). Conversely, in a systematic review, Muggli and Halliday (2007) were unable to demonstrate a significant association. Analysis of twinning rate in Texas after folic acid fortification of cereal-grain products also did not demonstrate an independent increase in twinning between 1996 and 1998 (Waller, 2003).
Pituitary Gonadotropin. The common factor linking race, age, weight, and fertility to multifetal gestation may be FSH levels (Benirschke, 1973). This theory is supported by the fact that increased fecundity and a higher rate of dizygotic twinning have been reported in women who conceive within 1 month after stopping oral contraceptives, but not during subsequent months (Rothman, 1977). This may be due to the sudden release of pituitary gonadotropin in amounts greater than usual during the first spontaneous cycle after stopping hormonal contraception. Indeed, the paradox of declining fertility but increasing twinning with advancing maternal age can be explained by an exaggerated pituitary release of FSH in response to decreased negative feedback from impending ovarian failure (Beemsterboer, 2006).
Infertility Therapy. Ovulation induction with FSH plus chorionic gonadotropin or clomiphene citrate remarkably enhances the likelihood of multiple ovulations. A mainstay of current infertility therapy and common antecedent to IVF is ovarian stimulation followed by timed intrauterine insemination. In their review of this practice, McClamrock and coworkers (2012) reported rates of twin and higher-order multifetal pregnancies as high as 28.6 percent and 9.3 percent, respectively. Rates this high remain a major concern. There are currently two ongoing multicenter trials—Assessment of Multiple Gestations from Ovarian Stimulation (AMIGOS) and Pregnancy in Polycystic Ovary Syndrome II (PPCOSII)—that are designed to provide guidance on achieving maximum pregnancy rates while minimizing multifetal gestation rates (Diamond, 2011; Legro, 2012).
In general with IVF, the greater the number of embryos that are transferred, the greater the risk of twins and multiple fetuses. In 2005, 1 percent of infants born in the United States were conceived through ART, and these infants accounted for 17 percent of multifetal births (Wright, 2008). The American Society for Reproductive Medicine (2013) recently revised their age-related guidelines on numbers of cleavage-stage embryos or blastocysts to transfer in an effort to reduce the incidence of higher-order multifetal pregnancies. For example, women younger than 35 years with a favorable prognosis should have no more than two embryos transferred. These practices have effectively lowered rates (Kulkarni, 2013).
 Sex Ratios with Multiple Fetuses
Sex Ratios with Multiple Fetuses
In humans, as the number of fetuses per pregnancy increases, the percentage of male conceptuses decreases. Strandskov and coworkers (1946) found the percentage of males in 31 million singleton births in the United States was 51.6 percent. For twins, it was 50.9 percent; for triplets, 49.5 percent; and for quadruplets, 46.5 percent. Swedish birth data spanning 135 years reveals the number of males per 100 female infants born was 106 among singletons, 103 among twins, and 99 among triplets (Fellman, 2010). Females predominate even more in twins from late twinning events. For example, 68 percent of thoracopagus conjoined twins are female (Mutchinick, 2011). Two explanations have been offered. First, beginning in utero and extending throughout the life cycle, mortality rates are lower in females. Second, female zygotes have a greater tendency to divide.
 Determination of Zygosity
Determination of Zygosity
Twins of opposite sex are almost always dizygotic. In rare instances, due to somatic mutations or chromosome aberrations, the karyotype or phenotype of a monozygous twin gestation can be different. Most reported cases are related to postzygotic loss of the Y chromosome in one 46,XY twin. This results in a phenotypically female twin due to Turner syndrome (45,X). Zech and colleagues (2008) reported a rare case of a 47,XXY zygote that underwent postzygotic loss of the X chromosome in some cells and loss of the Y chromosome in other cells. The phenotype of the resultant twins was one male and one female. Karyotype analyses revealed both to be 46,XX/46,XY genetic mosaics.
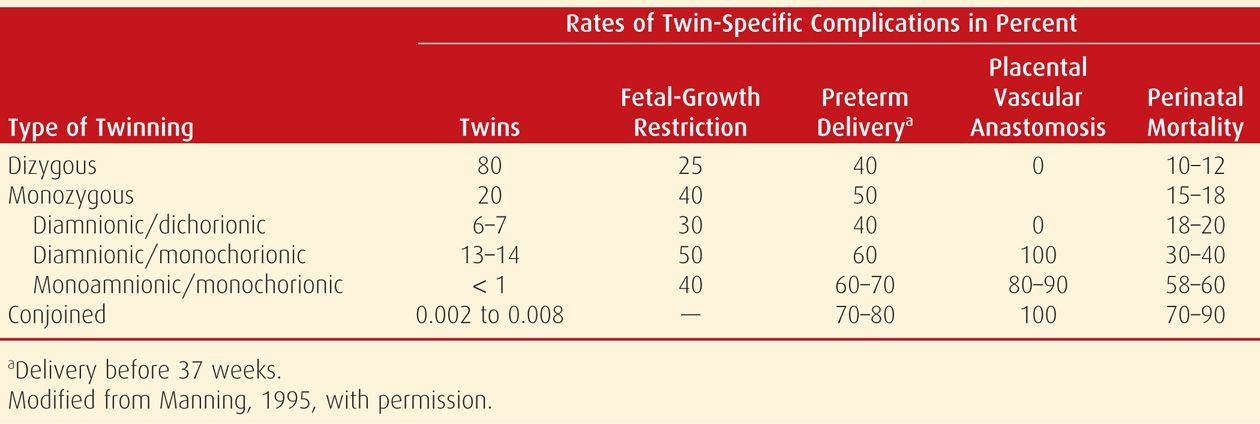
The risk for twin-specific complications varies in relation to zygosity as well as chorionicity—the number of chorions. As shown in Table 45-3, the latter is the more important determinant. Specifically, there are increased rates of perinatal mortality and neurological injury in monochorionic diamnionic twins compared with dichorionic pairs (Hack, 2008; Lee, 2008). In their retrospective analysis of more than 2000 twins, McPherson and associates (2012) reported that the risk of fetal demise in one or both monochorionic twins was twice that in dichorionic multifetal gestations.
TABLE 45-3. Overview of the Incidence of Twin Pregnancy Zygosity and Corresponding Twin-Specific Complications
Sonographic Determination of Chorionicity
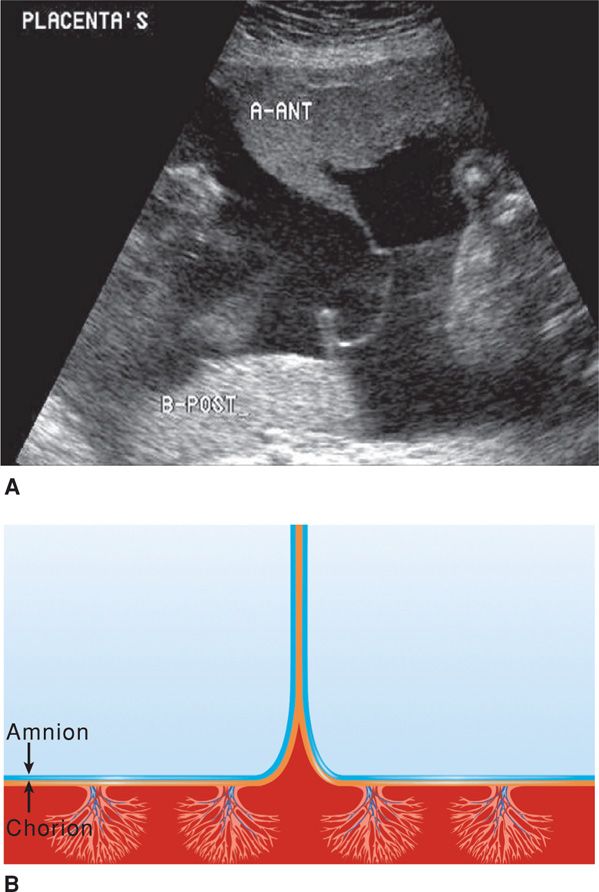
Chorionicity can sometimes be identified in the first trimester with sonography. Two separate placentas suggest dizygosity. In pregnancies with a single placental mass, it may be difficult to identify chorionicity. Identification of a thick dividing membrane—generally 2 mm or greater—supports a presumed diagnosis of dichorionicity. Also, the twin peak sign is seen by examining the point of origin of the dividing membrane on the placental surface. The peak appears as a triangular projection of placental tissue extending a short distance between the layers of the dividing membrane (Fig. 45-4).
FIGURE 45-4 A. Sonographic image of the “twin-peak” sign, also termed the “lambda sign,” in a 24-week gestation. At the top of this sonogram, tissue from the anterior placenta is seen extending downward between the amnion layers. This sign confirms dichorionic twinning. B. Schematic diagram of the “twin-peak” sign. A triangular portion of placenta is seen insinuating between the amniochorion layers.
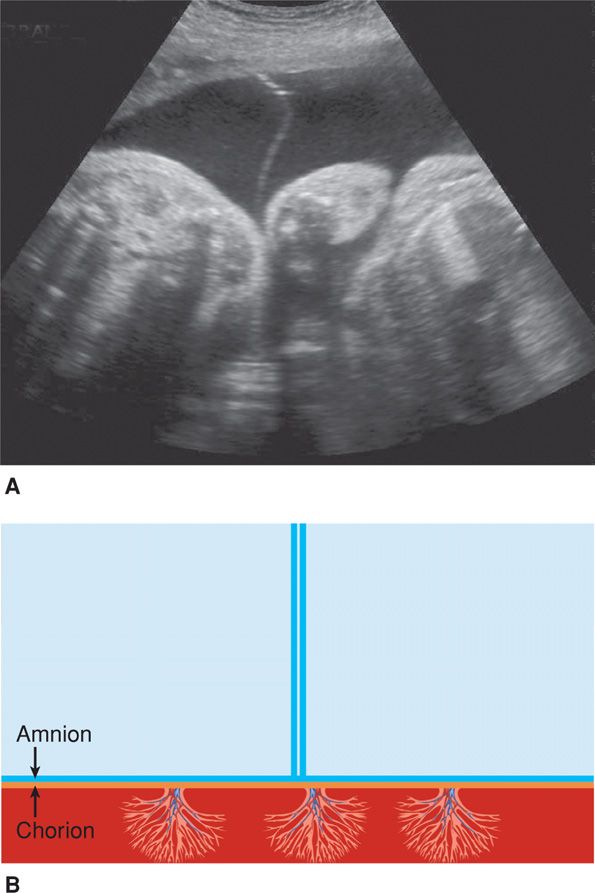
In contrast, monochorionic pregnancies have a dividing membrane that is so thin it may not be seen until the second trimester. The membrane is generally less than 2-mm thick, and magnification reveals only two layers. The right-angle relationship between the membranes and placenta and no apparent extension of placenta between the dividing membranes is called the T sign (Fig. 45-5). Evaluation of the dividing membrane can establish chorionicity in more than 99 percent of pregnancies in the first trimester (Miller, 2012).
FIGURE 45-5 A. Sonographic image of the “T” sign in a monochorionic diamnionic gestation at 30 weeks. B. Schematic diagram of the “T” sign. Twins are separated only by a membrane created by the juxtaposed amnion of each twin. A “T” is formed at the point at which amnions meet the placenta.
Placental Examination
A carefully performed visual examination of the placenta and membranes after delivery serves to establish zygosity and chorionicity promptly in approximately two thirds of cases. The following systematic examination is recommended. As the first neonate is delivered, one clamp is placed on a portion of its cord. Cord blood is generally not collected until after delivery of the other twin. As the second neonate is delivered, two clamps are placed on that cord. Three clamps are used to mark the cord of a third neonate, and so on as necessary. Until the delivery of the last fetus, each cord segment must remain clamped to prevent fetal hypovolemia and anemia caused by blood leaving the placenta via anastomoses and then through an unclamped cord.
The placenta should be carefully delivered to preserve the attachment of the amnion and chorion. With one common amnionic sac, or with juxtaposed amnions not separated by chorion arising between the fetuses, the fetuses are monozygotic. If adjacent amnions are separated by chorion, then the fetuses could be either dizygotic or monozygotic, but dizygosity is more common (see Figs. 45-1 and 45-6). If the neonates are of the same sex, blood typing of cord blood samples may be helpful. Different blood types confirm dizygosity, although demonstrating the same blood type in each fetus does not confirm monozygosity. For definitive diagnosis, more complicated techniques such as DNA fingerprinting can be used, but these tests are generally not performed at birth unless there is a pressing medical indication.
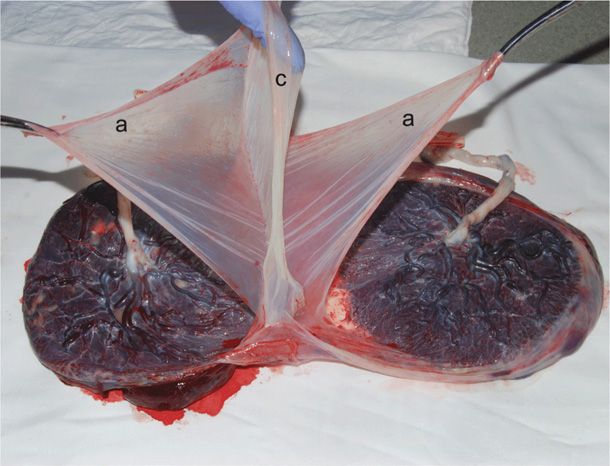
FIGURE 45-6 Dichorionic diamnionic twin placenta. The membrane partition that separated twin fetuses is elevated and consists of chorion (c) between two amnions (a).
DIAGNOSIS OF MULTIPLE FETUSES
 Clinical Evaluation
Clinical Evaluation
During examination, accurate fundal height measurement, described in Chapter 9 (p. 176), is essential. With multiple fetuses, uterine size is typically larger during the second trimester than expected. Rouse and associates (1993) reported fundal heights in 336 well-dated twin pregnancies. Between 20 and 30 weeks, fundal heights averaged approximately 5 cm greater than expected for singletons of the same fetal age.
In general, it is difficult to diagnose twins by palpation of fetal parts before the third trimester. Even late in pregnancy, it may be difficult to identify twins by abdominal palpation, especially if one twin overlies the other, if the woman is obese, or if there is hydramnios. Palpating two fetal heads, often in different uterine quadrants, strongly supports a twin diagnosis.
Late in the first trimester, fetal heart action may be detected with Doppler ultrasonic equipment. Thereafter, it becomes possible to identify two fetal heartbeats if their rates are clearly distinct from each other and from that of the mother. Careful examination with an aural fetal stethoscope can identify fetal heart sounds in twins as early as 18 to 20 weeks.
 Sonography
Sonography
By careful sonographic examination, separate gestational sacs can be identified early in twin pregnancy (Fig. 45-7). Subsequently, each fetal head should be seen in two perpendicular planes so as not to mistake a cross section of the fetal trunk for a second fetal head. Ideally, two fetal heads or two abdomens should be seen in the same image plane, to avoid scanning the same fetus twice and interpreting it as twins. Sonographic examination should detect practically all sets of twins. Given the increased frequency of sonographic examinations during the first trimester, early detection of a twin pregnancy is common. Indeed, one argument in favor of routine first-trimester sonographic screening is earlier detection of multiple fetuses.
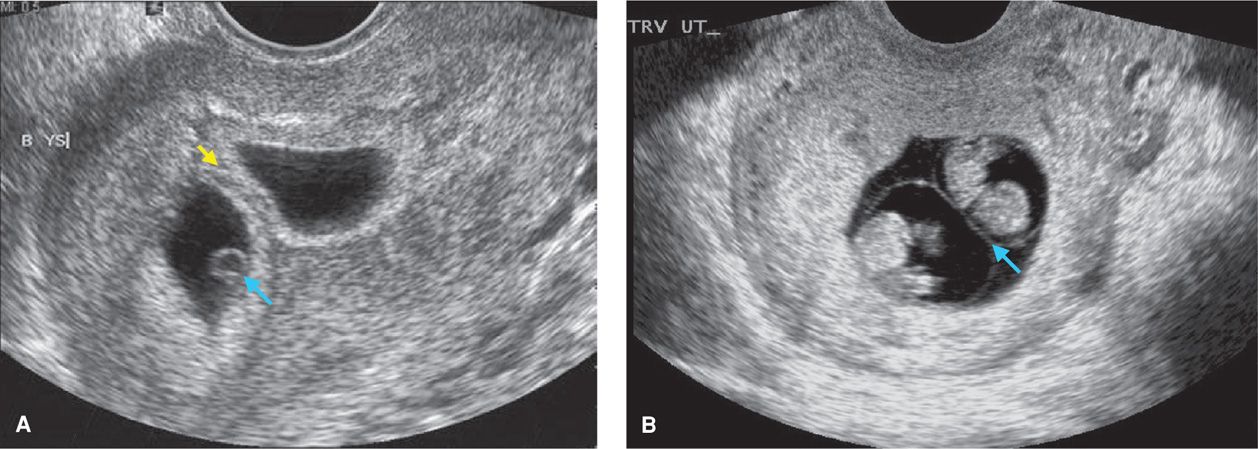
FIGURE 45-7 Sonograms of first-trimester twins. A. Dichorionic diamnionic twin pregnancy at 6 weeks’ gestation. Note the thick dividing chorion (yellow arrow). One of the yolk sacs is indicated (blue arrow). B. Monochorionic diamnionic twin pregnancy at 8 weeks’ gestation. Note the thin amnion encircling each embryo, resulting in a thin dividing membrane (blue arrow).
Higher-order multifetal gestations are more difficult to evaluate. Even in the first trimester, it can be difficult to identify the actual number of fetuses and their position. This determination is especially important if pregnancy reduction or selective termination is considered (p. 919).
 Other Diagnostic Aids
Other Diagnostic Aids
Radiography and Magnetic Resonance Imaging
Abdominal radiography can be used if fetal number in a higher-order multifetal gestation is uncertain. However, radiographs generally have limited utility and may lead to an incorrect diagnosis if there is hydramnios, obesity, fetal movement during the exposure, or inappropriate exposure time. Additionally, fetal skeletons before 18 weeks’ gestation are insufficiently radiopaque and may be poorly seen.
Although not typically used to diagnose multifetal pregnancy, magnetic resonance (MR) imaging may help delineate complications in monochorionic twins (Hu, 2006). Bekiesinska-Figatowska and colleagues (2013) reviewed their experience with 17 complicated twin gestations evaluated by both sonographic and MR imaging. They concluded that MR imaging provides a more detailed assessment of pathology in twins and is particularly helpful in cases of conjoined twins.
Biochemical Tests
There is no biochemical test that reliably identifies multiple fetuses. Serum and urine levels of β-hCG and maternal serum alpha-fetoprotein (MSAFP) are generally higher with twins compared with singletons. However, levels may vary considerably and overlap with those of singletons.
MATERNAL ADAPTATION TO MULTIFETAL PREGNANCY
The various physiological burdens of pregnancy and the likelihood of serious maternal complications are typically greater with multiple fetuses than with a singleton. This should be considered, especially when counseling a woman whose health is compromised and in whom multifetal gestation is recognized early. Similar consideration is given to a woman who is not pregnant but is considering infertility treatment.
Beginning in the first trimester, and temporarily associated with higher serum β-hCG levels, women with multifetal gestation often have nausea and vomiting in excess of women with a singleton pregnancy. In women with multiple fetuses, blood volume expansion is greater and averages 50 to 60 percent compared with 40 to 50 percent in those with a singleton (Pritchard, 1965). Although the red cell mass also increases, it does so proportionately less in twin pregnancies. Combined with increased iron and folate requirements, this predisposes to anemia. This augmented hypervolemia teleologically offsets blood loss with vaginal delivery of twins, which is twice that with a single fetus.
Women carrying twins also have a typical pattern of arterial blood pressure change. MacDonald-Wallis and coworkers (2012) analyzed serial blood pressures in more than 13,000 singleton and twin pregnancies. As early as 8 weeks’ gestation, the diastolic blood pressure in women with twins was lower than that with singleton pregnancies but generally increased to a greater degree at term. In an earlier study, Campbell (1986) demonstrated that this increase was at least 15 mm Hg in 95 percent of women with twins compared with only 54 percent of women with a singleton.
Hypervolemia along with decreased vascular resistance has an impressive affect on cardiac function. Kametas and associates (2003) assessed function in 119 women with a twin pregnancy. In these women, cardiac output was increased another 20 percent above that in women with a singleton pregnancy. Kuleva and colleagues (2011) compared serial echocardiography results in 20 women with uncomplicated twin pregnancies with those of 10 women with singletons. Similarly, they also demonstrated a greater increase in cardiac output in those with twins. Both studies found the cardiac output rise was predominantly due to greater stroke volume and to a much lesser degree due to increased heart rate. Vascular resistance was significantly lower in twin gestations throughout pregnancy compared with singleton gestations (Kuleva, 2011).
Uterine growth in a multifetal gestation is substantively greater than in a singleton pregnancy. The uterus and its nonfetal contents may achieve a volume of 10 L or more and weigh in excess of 20 pounds. Especially with monozygotic twins, excessive amounts of amnionic fluid may rapidly accumulate. In these circumstances, maternal abdominal viscera and lungs can be appreciably compressed and displaced by the expanding uterus. As a result, the size and weight of the large uterus may preclude more than a sedentary existence for these women.
If hydramnios develops, maternal renal function can become seriously impaired, most likely as the consequence of obstructive uropathy (Quigley, 1977). With severe hydramnios, therapeutic amniocentesis may provide relief for the mother, may improve obstructive uropathy, and possibly may lower the preterm delivery risk that follows preterm labor or prematurely ruptured membranes (Chap. 11, p. 236). Unfortunately, hydramnios is often characterized by acute onset remote from term and by rapid reaccumulation following amniocentesis.
PREGNANCY COMPLICATIONS
 Spontaneous Abortion
Spontaneous Abortion
Miscarriage is more likely with multiple fetuses. During a 16-year study, Joo and colleagues (2012) demonstrated that the spontaneous abortion rate per live birth in singleton pregnancies was 0.9 percent compared with 7.3 percent in multiple pregnancies. Furthermore, they found that monochorial placentation was more common in multiple gestations ending in miscarriage than in those resulting in a livebirth. Twin pregnancies achieved through ART are at increased risk for abortion compared with those conceived spontaneously (Szymusik, 2012).
 Congenital Malformations
Congenital Malformations
The incidence of congenital malformations is appreciably increased in multifetal gestations compared with singletons. Glinianaia and associates (2008) reported that the rate of congenital malformations was 406 per 10,000 twins versus 238 per 10,000 singletons. In this survey-based study, the malformation rate in monochorionic twins was almost twice that of dichorionic twin gestations. This increase has been attributed to the high incidence of structural defects in monozygotic twins. But from a 30-year European registry of multifetal births, Boyle and coworkers (2013) found a steady increase in structural anomalies from 1987 (2.16 percent) to 2007 (3.26 percent). During this time, the proportion of dizygotic twins increased by 30 percent, while the proportion of monozygotic twins remained stable. This higher risk of congenital malformations in dizygotic twins over time correlates with increased availability of ART. An increase in birth defects related to ART has been reported repeatedly (Talauliker, 2012).
 Low Birthweight
Low Birthweight
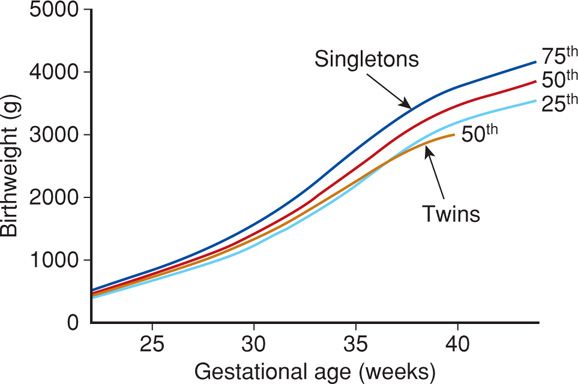
Multifetal gestations are more likely to be low birthweight than singleton pregnancies due to restricted fetal growth and preterm delivery. From 1988 to 2012 at Parkland Hospital, data were collected from 357,205 singleton neonates without malformations and from 3714 normal twins who were both liveborn. Birthweights in twin infants closely paralleled those of singletons until 28 to 30 weeks’ gestation. Thereafter, twin birthweights progressively lagged (Fig. 45-8). Beginning at 35 to 36 weeks, twin birthweights clearly diverge from those of singletons.
FIGURE 45-8 Birthweight percentiles (25th to 75th) for 357,205 singleton neonates compared with the 50th birthweight percentile for 3714 twins, Parkland Hospital 1988—2012. Infants with major malformations, pregnancies complicated by stillbirth, and twin gestations with > 25 percent discordance were excluded. (Data courtesy of Dr. Don McIntire.)
In general, the degree of growth restriction increases with fetal number. The caveat is that this assessment is based on growth curves established for singletons. Several authorities argue that fetal growth in twins is different from that of singleton pregnancies, and thus abnormal growth should be diagnosed only when fetal size is less than expected for multifetal gestation. Accordingly, twin and triplet growth curves have been developed (Kim, 2010; Odibo, 2013; Vora, 2006).
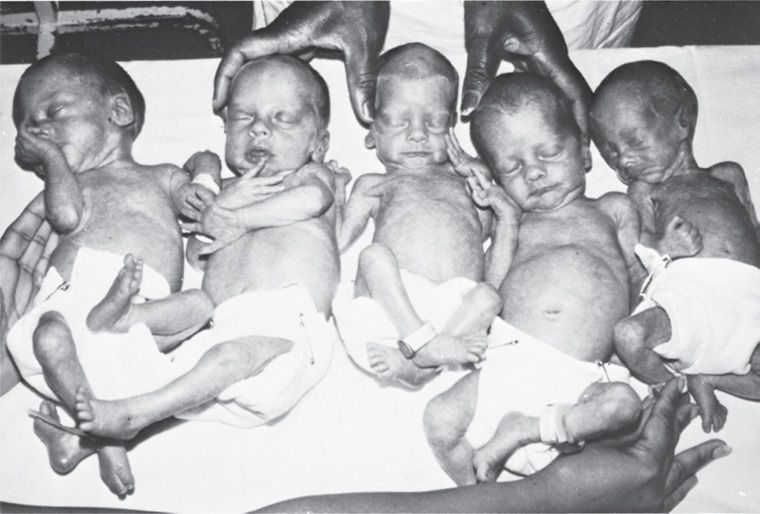
The degree of growth restriction in monozygotic twins is likely to be greater than that in dizygotic pairs (Fig. 45-9). With monochorionic embryos, allocation of blastomeres may not be equal, vascular anastomoses within the placenta may cause unequal distribution of nutrients and oxygen, and discordant structural anomalies resulting from the twinning event itself may affect growth. For example, the quintuplets shown in Figure 45-10 represent three dizygotic and two monozygotic fetuses. When delivered at 31 weeks, the three neonates from separate ova weighed 1420, 1530, and 1440 g, whereas the two derived from the same ovum weighed 990 and 860 g.
FIGURE 45-9 Marked growth discordance in monochorionic twins. (Photograph contributed by Dr. Laura Greer.)
FIGURE 45-10 Davis quintuplets at 3 weeks following delivery. The first, second, and fourth newborns from the left each arose from separate ova, whereas the third and fifth neonates are from the same ovum.
In the third trimester, the larger fetal mass leads to accelerated placental maturation and relative placental insufficiency. In dizygotic pregnancies, marked size discordancy usually results from unequal placentation, with one placental site receiving more perfusion than the other. Size differences may also reflect different genetic fetal-growth potentials. Discordancy can also result from fetal malformations, genetic syndromes, infection, or umbilical cord abnormalities such as velamentous insertion, marginal insertion, or vasa previa (Chap. 6, p. 122).
 Hypertension
Hypertension
Hypertensive disorders due to pregnancy are more likely to develop with multiple fetuses. The exact incidence attributable to twin gestation is difficult to determine because twin pregnancies are more likely to deliver preterm before preeclampsia can develop and because women with twin pregnancies are often older and multiparous. The incidence of pregnancy-related hypertension in women with twins is 20 percent at Parkland Hospital. In their analysis of 513 twin pregnancies > 20 weeks’ gestation, Fox and coworkers (2014) also identified 20 percent of parturients with either gestational hypertension or preeclampsia. Case-control analyses suggest that prepregnancy body mass index (BMI) ≥ 30 kg/m2 and egg donation are additional independent risk factors for preeclampsia. Gonzalez and colleagues (2012) compared 257 women with twins and gestational diabetes with 277 nondiabetic women carrying twins. These researchers found a twofold increased risk of preeclampsia in women diagnosed with gestational diabetes. Finally, in the Matched Multiple Birth Dataset for the National Center for Health Statistics, Luke and associates (2008) analyzed 316,696 twin, 12,193 triplet, and 778 quadruple pregnancies. These investigators found that the risk for pregnancy-associated hypertension was significantly increased for triplets and quadruplets (11 and 12 percent, respectively) compared with that for twins (8 percent).
These data suggest that fetal number and placental mass are involved in preeclampsia pathogenesis. Women with twin pregnancies have levels of antiangiogenic soluble fms-like tyrosine kinase-1 (sFlt-1) that are twice that of singletons. Levels are seemingly related to increased placental mass rather than primary placental pathology (Bdolah, 2008; Maynard, 2008). Rana and coworkers (2012) measured antiangiogenic sFlt-1 and proangiogenic placental growth factor (PlGF) in 79 women with twins referred for evaluation of preeclampsia. In the 58 women identified with either gestational hypertension or preeclampsia, there was a stepwise increase in sFlt-1 concentrations, decrease in PlGF levels, and increase in sFlt-1/PlGF ratios compared with normotensive twin pregnancies. With multifetal gestation, hypertension not only develops more often but also tends to develop earlier and be more severe. In the analysis of angiogenic factors mentioned above, more than one half presented before 34 weeks, and in those who did, the sFlt-1/PlGF ratio rise was more striking (Rana, 2012). This relationship is discussed in Chapter 40 (p. 735).
 Preterm Birth
Preterm Birth
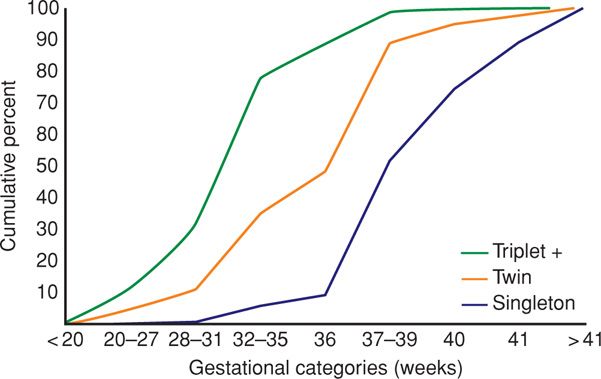
The duration of gestation decreases with increasing fetal number (Fig. 45-11). According to Martin and colleagues (2012), more than five of every 10 twins and nine of 10 triplets born in the United States in 2010 were delivered preterm. Delivery before term is a major reason for increased neonatal morbidity and mortality rates in multifetal pregnancy. Prematurity is increased sixfold and tenfold in twins and triplets, respectively (Giuffre, 2012). In their review, Chauhan and associates (2010) reported that, similar to singleton pregnancies, approximately 60 percent of preterm births in twins are indicated, about a third result from spontaneous labor, and 10 percent follow prematurely ruptured membranes. In their analysis of almost 300,000 live births in Ohio, Pakrashi and DeFranco (2013) found that the proportion of preterm birth associated with premature membrane rupture increased with gestational plurality from 13 percent with singletons to 20 percent with triplets or more.
FIGURE 45-11 Cumulative percent of singleton, twin, and triplet or higher-order multifetal births according to gestational age at delivery in the United States during 1990. (From Luke, 1994, with permission.)
The preterm birth rate among multifetal gestations has increased during the past two decades. In an analysis of nearly 350,000 twin births, Kogan and coworkers (2000) showed that during the 16-year period ending in 1997, the term birth rate among twins declined by 22 percent. Joseph and colleagues (2001) attributed this decline to an increased rate of indicated preterm deliveries. This trend is not necessarily negative, as it was associated with decreased perinatal morbidity and mortality rates among twins that reached 34 weeks. Although the causes of preterm delivery in twins and singletons may be different, neonatal outcome is generally the same at similar gestational ages (Gardner, 1995; Kilpatrick, 1996; Ray, 2009). Importantly, outcomes for preterm twins who are markedly discordant may not be comparable with those for singletons because whatever caused the discordance may have long-lasting effects.
 Prolonged Pregnancy
Prolonged Pregnancy
More than 40 years ago, Bennett and Dunn (1969) suggested that a twin pregnancy of 40 weeks or more should be considered postterm. Twin stillborn neonates delivered at 40 weeks or beyond commonly had features similar to those of postmature singletons (Chap. 43, p. 864). From an analysis of almost 300,000 twin births, Kahn and coworkers (2003) calculated that at and beyond 39 weeks, the risk of subsequent stillbirth was greater than the risk of neonatal mortality. At Parkland Hospital, twin gestations have empirically been considered to be prolonged at 40 weeks.
 Long-Term Infant Development
Long-Term Infant Development
Historically, twins have been considered cognitively delayed compared with singletons (Record, 1970; Ronalds, 2005). However, in cohort studies evaluating normal-birthweight term infants, cognitive outcomes between twins and singletons are similar (Lorenz, 2012). Christensen and associates (2006) found similar national standardized test scores in ninth grade in 3411 twins and 7796 singletons born between 1986 and 1988. In contrast, among normal-birthweight infants, the cerebral palsy risk is higher among twins and higher-order multiples. For example, the cerebral palsy rate has been reported to be 2.3 per 1000 in singletons, 12.6 per 1000 in twins, and 44.8 per 1000 in triplets (Giuffre, 2012). Much of this excess risk is thought to be related to an increased risk of fetal-growth restriction, congenital anomalies, twin-twin transfusion syndrome, and fetal demise of a cotwin (Lorenz, 2012).
UNIQUE FETAL COMPLICATIONS
Several unique and fascinating complications arise in multifetal pregnancies. These have been best described in twins but can be found in higher-order multifetal gestations. Most fetal complications due to the twinning process itself are seen with monozygotic twins. Their pathogenesis is best understood after reviewing the possibilities shown in Figure 45-1.
 Monoamnionic Twins
Monoamnionic Twins
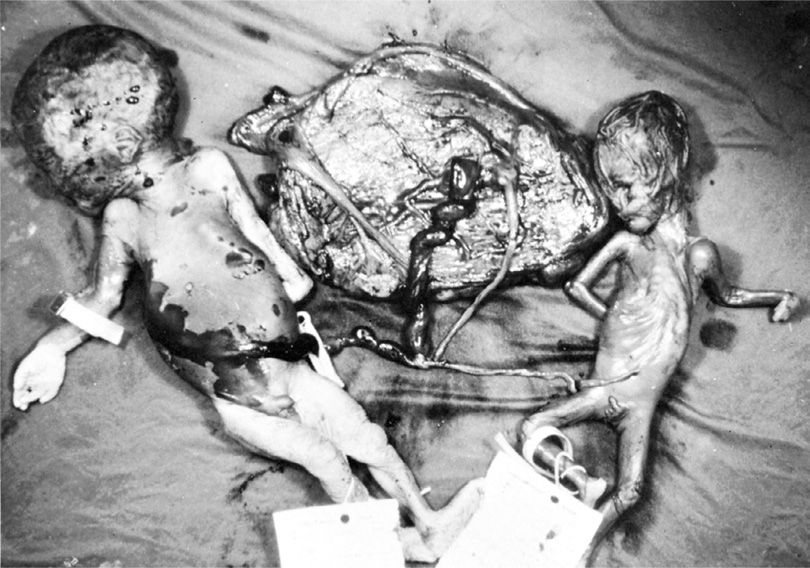
Only about 1 percent of all monozygotic twins will share an amnionic sac (Hall, 2003). Said another way, approximately 1 in 20 monochorionic twins are monoamnionic (Lewi, 2013). This configuration is associated with a high fetal death rate from cord entanglement, congenital anomalies, preterm birth, or twin-twin-transfusion syndrome, which is described subsequently. In a comprehensive review, Allen and associates (2001) reported that monoamnionic twins diagnosed antenatally and alive at 20 weeks have approximately a 10-percent risk of subsequent fetal demise. In a Dutch report of 98 monoamnionic twin pregnancies, the perinatal mortality rate was 17 percent (Hack, 2009). Umbilical cord entanglement, a frequent cause of death, is estimated to complicate at least half of cases (Fig. 45-12). Diamnionic twins can become monoamnionic if the dividing membrane ruptures and therefore have similar associated morbidity and mortality rates.
FIGURE 45-12 Monozygotic twins in a single amnionic sac. The smaller fetus apparently died first, and the second subsequently succumbed when umbilical cords entwined.
Unfortunately, there are no management methods that guarantee good outcomes for either or both twins. This is because of the unpredictability of fetal death from cord entanglement and the lack of an effective means of monitoring for it. Quinn and colleagues (2011) retrospectively evaluated the feasibility of inpatient continuous fetal heart monitoring in 17 sets of monoamnionic twins. After review of more than 10,000 hours of fetal tracing, these investigators concluded that this was possible in only 50 percent of cases. Importantly, an abnormal fetal heart rate tracing prompted delivery in only six cases. Morbid cord entanglement appears to occur early, and monoamnionic pregnancies that have successfully reached 30 to 32 weeks are at reduced risk. In the Dutch series described above, the incidence of intrauterine demise dropped from 15 percent after 20 weeks to 4 percent when gestational age exceeded 32 weeks (Hack, 2009).
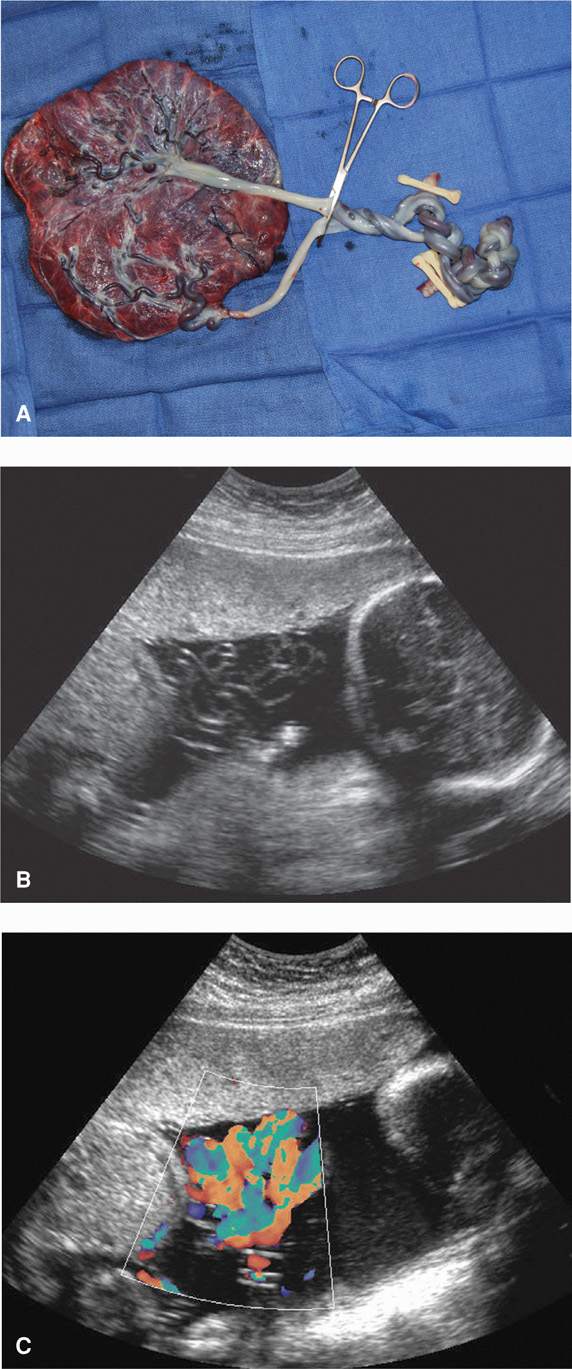
Although umbilical cords frequently entangle, factors that lead to pathological umbilical vessel constriction are unknown. Color-flow Doppler sonography can be used to diagnose entanglement (Fig. 45-13). However, once identified, evidence to guide management is observational, retrospective, and subject to biased reporting. One proposed management scheme is based on a study by Heyborne and coworkers (2005), who reported no stillbirths in 43 twin pregnancies of women admitted at 26 to 27 weeks’ gestation for daily fetal surveillance. Conversely, there were 13 stillbirths in 44 women who were managed as outpatients and admitted only for an obstetrical indication. Because of this report, women with monoamnionic twins are recommended to undergo 1 hour of daily fetal heart rate monitoring, either as outpatients or as inpatients, beginning at 26 to 28 weeks. With initial testing, a course of betamethasone is given to promote pulmonary maturation (Chap. 42, p. 850). If fetal testing remains reassuring, cesarean delivery is performed at 34 weeks and after a second course of betamethasone. This management scheme is used at Parkland Hospital and resulted in the successful 34-week delivery of the twins depicted in Figure 45-13.
FIGURE 45-13 Monochorionic monoamnionic cord entanglement. A. Despite marked knotting of the cords, vigorous twins were delivered by cesarean. B. Preoperative sonogram of this pregnancy shows entwined cords. C. This finding is accentuated with application of color Doppler. (Photographs contributed by Dr. Julie Lo.)
 Aberrant Twinning Mechanisms
Aberrant Twinning Mechanisms
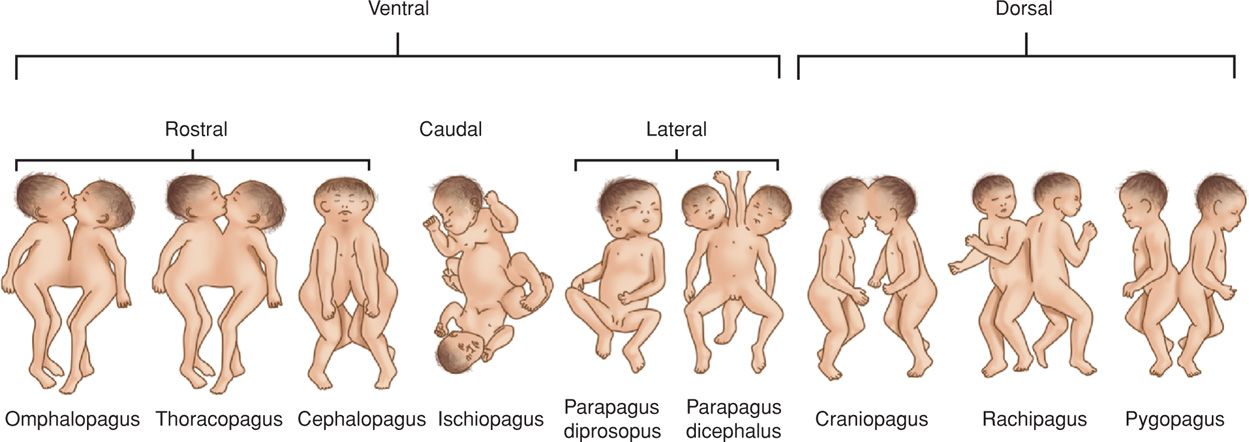
Several aberrations in the twinning process result in a spectrum of fetal malformations. These are traditionally ascribed to incomplete splitting of an embryo into two separate twins. However, it is possible that they may result from early secondary fusion of two separate embryos. These separated embryos are either symmetrical or asymmetrical, and the spectrum of anomalies is shown in Figure 45-14.
FIGURE 45-14 Possible outcomes of monozygotic twinning. The asymmetrical category contains twinning types in which one twin complement is substantially smaller and incompletely formed.
Conjoined Twins
In the United States, united or conjoined twins have been referred to as Siamese twins–after Chang and Eng Bunker of Siam (Thailand), who were displayed worldwide by P. T. Barnum. Joining of the twins may begin at either pole and may produce characteristic forms depending on which body parts are joined or shared (Fig. 45-15). Of these, thoracopagus is the most common (Mutchinick, 2011). The frequency of conjoined twins is not well established. At Kandang Kerbau Hospital in Singapore, Tan and coworkers (1971) identified seven cases of conjoined twins among more than 400,000 deliveries–an incidence of 1 in 60,000.
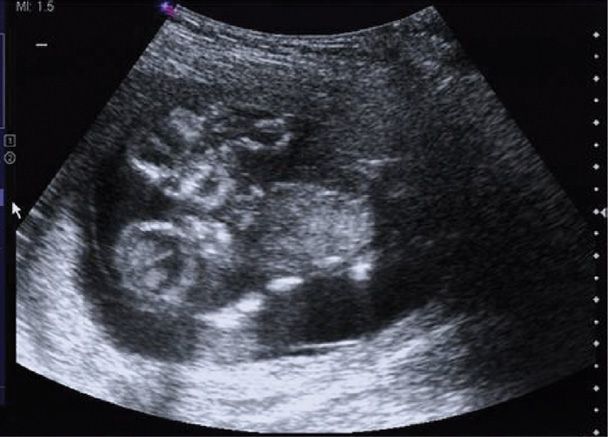
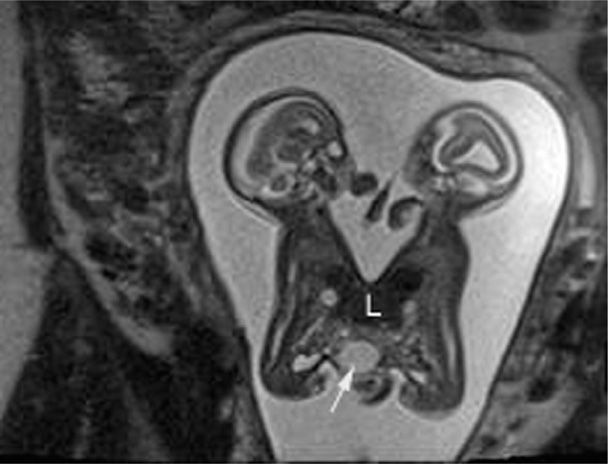
As reviewed by McHugh and associates (2006), conjoined twins can frequently be identified using sonography at midpregnancy. This provides an opportunity for parents to decide whether to continue the pregnancy. As shown in Figure 45-16, identification of cases during the first trimester is also possible. A targeted examination, including a careful evaluation of the connection and the organs involved, is necessary before counseling can be provided. As shown in Figure 45-17, MR imaging can play an important adjunctive role in clarifying shared organs. Compared with sonography, MR imaging may provide superior views, especially in later pregnancy when amnionic fluid is diminished and fetal crowding is increased (Hibbeln, 2012).
FIGURE 45-16 Sonogram of a conjoined twin pregnancy at 13 weeks’ gestation. These thoracoomphalopagus twins have two heads but a shared chest and abdomen.
FIGURE 45-17 Magnetic resonance imaging of conjoined twins. This T2-weighted HASTE sagittal image demonstrates fusion from the level of the xiphoid process to just below the level of the umbilicus, that is, omphalopagus twins. Below the fused liver (L), there is a midline cystic mass (arrow) within the tissue connecting the twins. An omphalomesenteric cyst was favored given the location within the shared tissue. (Image contributed by Dr. April Bailey.)
Surgical separation of an almost completely joined twin pair may be successful if essential organs are not shared (Spitz, 2003; Tannuri, 2013). Consultation with a pediatric surgeon often assists parental decision making. Conjoined twins may have discordant structural anomalies that further complicate decisions about whether to continue the pregnancy.
Viable conjoined twins should be delivered by cesarean. For the purpose of pregnancy termination, however, vaginal delivery is possible because the union is most often pliable (Fig. 45-18). Still, dystocia is common, and if the fetuses are mature, vaginal delivery may be traumatic to the uterus or cervix.
FIGURE 45-18 Conjoined twins aborted at 17 weeks’ gestation. (Photograph contributed by Dr. Jonathan Willms.)
External Parasitic Twins
This is a grossly defective fetus or merely fetal parts, attached externally to a relatively normal twin. A parasitic twin usually consists of externally attached supernumerary limbs, often with some viscera. Classically, however, a functional heart or brain is absent. Attachment mirrors those sites described earlier for conjoined twins (see Fig. 45-14). Parasites are believed to result from demise of the defective twin, with its surviving tissues attached to and vascularized by its normal twin (Spencer, 2001). In a worldwide collaborative epidemiological study, parasitic twins were found to account for 3.9 percent of all conjoined twins and to occur more frequently in male fetuses (Mutchinick, 2011).
Fetus-in-Fetu
Early in development, one embryo may be enfolded within its twin. Normal development of this rare parasitic twin usually arrests in the first trimester. As a result, normal spatial arrangement of and presence of many organs is lost. Classically, vertebral or axial bones are found in these fetiform masses, whereas heart and brain are lacking. These masses are typically supported by their host by a few large parasitic vessels (Spencer, 2000). Malignant degeneration is rare (Kaufman, 2007).
 Monochorionic Twins and Vascular Anastomoses
Monochorionic Twins and Vascular Anastomoses
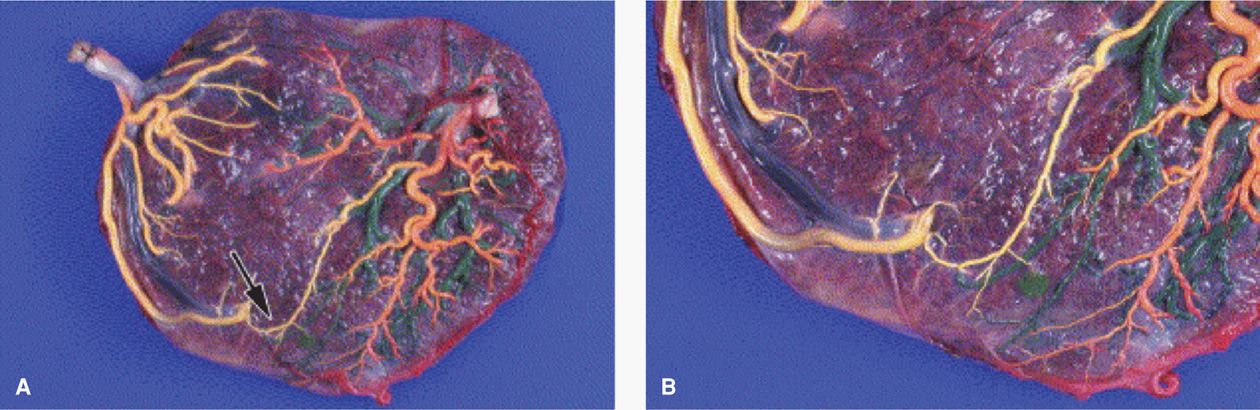
Another group of fascinating fetal syndromes can arise when monozygotic twinning results in two amnionic sacs and a common surrounding chorion. This leads to anatomical sharing of the two fetal circulations through anastomoses of placental arteries and veins. All monochorionic placentas likely share some anastomotic connections. However, there are marked variations in the number, size, and direction of these seemingly haphazard connections (Fig. 45-19). In their analyses of more than 200 monochorionic placentas, Zhao and colleagues (2013) found the median number of anastomoses to be 8 with an interquartile range of 4 to 14. With rare exceptions, anastomoses between twins are unique to monochorionic twin placentas.
FIGURE 45-19 Shared placenta from pregnancy complicated by twin-twin transfusion syndrome. The following color code was applied for injection. Left twin: yellow = artery, blue = vein; right twin: red = artery, green = vein. A. Part of the arterial network of the right twin is filled with yellow dye, due to the presence of a small artery-to-artery anastomosis (arrow). B. Close-up of the lower portion of the placenta displays the yellow dye-filled anastomosis. (From De Paepe, 2005, with permission.)
Artery-to-artery anastomoses are most common and are identified on the chorionic surface of the placenta in up to 75 percent of monochorionic twin placentas. Vein-to-vein and artery-to-vein communications are each found in approximately half. One vessel may have several connections, sometimes to both arteries and veins. In contrast to these superficial vascular connections on the surface of the chorion, deep artery-to-vein communications can extend through the capillary bed of a given villus (Fig. 45-20). These deep arteriovenous anastomoses create a common villous compartment or third circulation that has been identified in approximately half of monochorionic twin placentas.
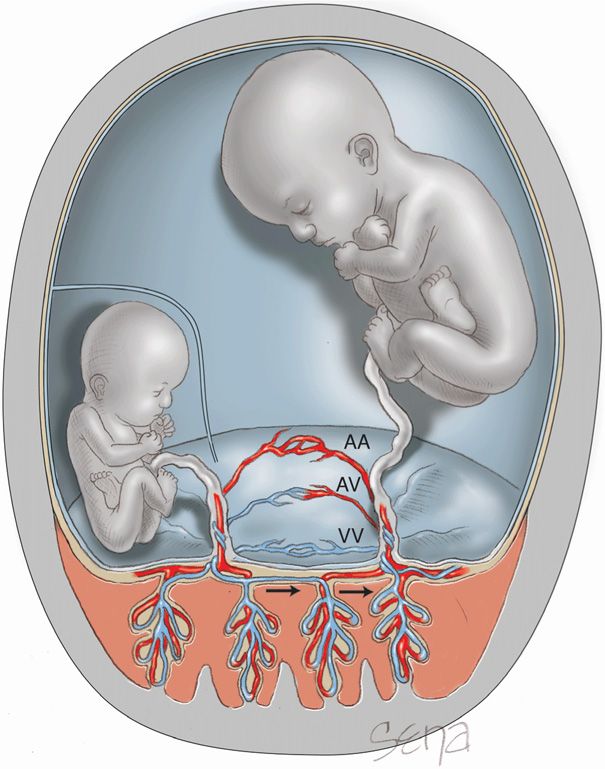
FIGURE 45-20 Anastomoses between twins may be artery-to-vein (AV), artery-to-artery (AA), or vein-to-vein (VV). Schematic representation of an AV anastomosis in twin-twin transfusion syndrome that forms a “common villous district” or “third circulation” deep within the villous tissue. Blood from a donor twin may be transferred to a recipient twin through this shared circulation. This transfer leads to a growth-restricted discordant donor twin with markedly reduced amnionic fluid, causing it to be “stuck.”
Whether these anastomoses are dangerous to either twin depends on the degree to which they are hemodynamically balanced. In those with significant pressure or flow gradients, a shunt will develop between fetuses. This chronic fetofetal transfusion may result in several clinical syndromes that include twin-twin transfusion syndrome (TTTS), twin anemia polycythemia sequence (TAPS), and acardiac twinning.
Twin-Twin Transfusion Syndrome (TTTS)
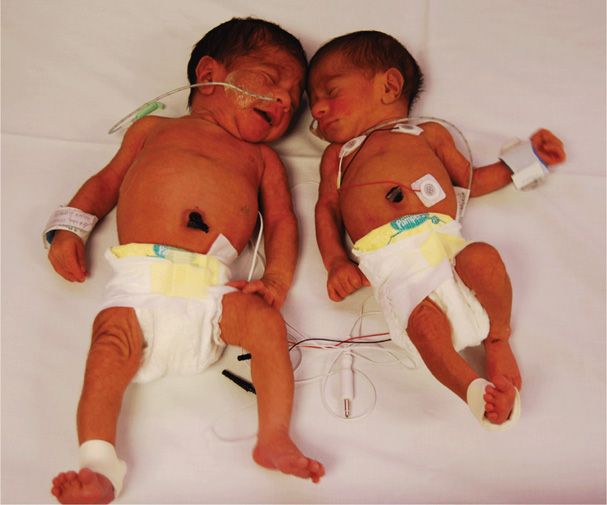
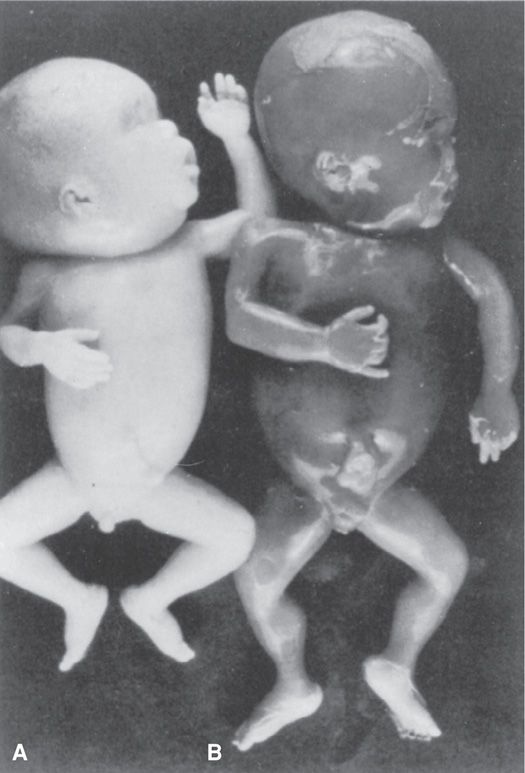
The prevalence of this condition is approximately 1 to 3 per 10,000 births (Simpson, 2013). In this syndrome, blood is transfused from a donor twin to its recipient sibling such that the donor may eventually become anemic and its growth may be restricted. In contrast, the recipient becomes polycythemic and may develop circulatory overload manifest as hydrops. The donor twin is pale, and its recipient sibling is plethoric (Fig. 45-21). Similarly, one portion of the placenta often appears pale compared with the remainder.
FIGURE 45-21 Twin-twin transfusion syndrome at 23 weeks. A. Pale donor twin (690 g) also had oligohydramnios. B. The plethoric recipient twin (730 g) had hydramnios. (From Mahone, 1993, with permission.)
The recipient neonate may have circulatory overload from heart failure and severe hypervolemia and hyperviscosity. Occlusive thrombosis is another concern. Finally, polycythemia in the recipient twin may lead to severe hyperbilirubinemia and kernicterus (Chap. 33, p. 644).
Pathophysiology. Any of the different types of vascular anastomoses discussed before may be found with monochorionic placentas. Classically, chronic TTTS results from unidirectional flow through arteriovenous anastomoses. Deoxygenated blood from a donor placental artery is pumped into a cotyledon shared by the recipient (see Fig. 45-20). Once oxygen exchange is completed in the chorionic villus, the oxygenated blood leaves the cotyledon via a placental vein of the recipient twin. Unless compensated—typically through arterioarterial anastomoses—this unidirectional flow leads to an imbalance in blood volumes (Lewi, 2013).
Clinically important twin-twin transfusion syndrome frequently is chronic and results from significant vascular volume differences between the twins. Even so, the pathogenesis is more complex than a net transfer of red blood cells from one twin to another. Indeed, in most monochorionic twin pregnancies complicated by the syndrome, there is no difference in hemoglobin concentrations between the donor and recipient twin (Lewi, 2013).
The syndrome typically presents in midpregnancy when the donor fetus becomes oliguric from decreased renal perfusion (Simpson, 2013). This fetus develops oligohydramnios, and the recipient fetus develops severe hydramnios, presumably due to increased urine production. Virtual absence of amnionic fluid in the donor sac prevents fetal motion, giving rise to the descriptive term stuck twin or polyhydramnios-oligohydramnios–syndrome—“poly-oli.” This amnionic fluid imbalance is associated with growth restriction, contractures, and pulmonary hypoplasia in the donor twin, and premature rupture of the membranes and heart failure in the recipient.
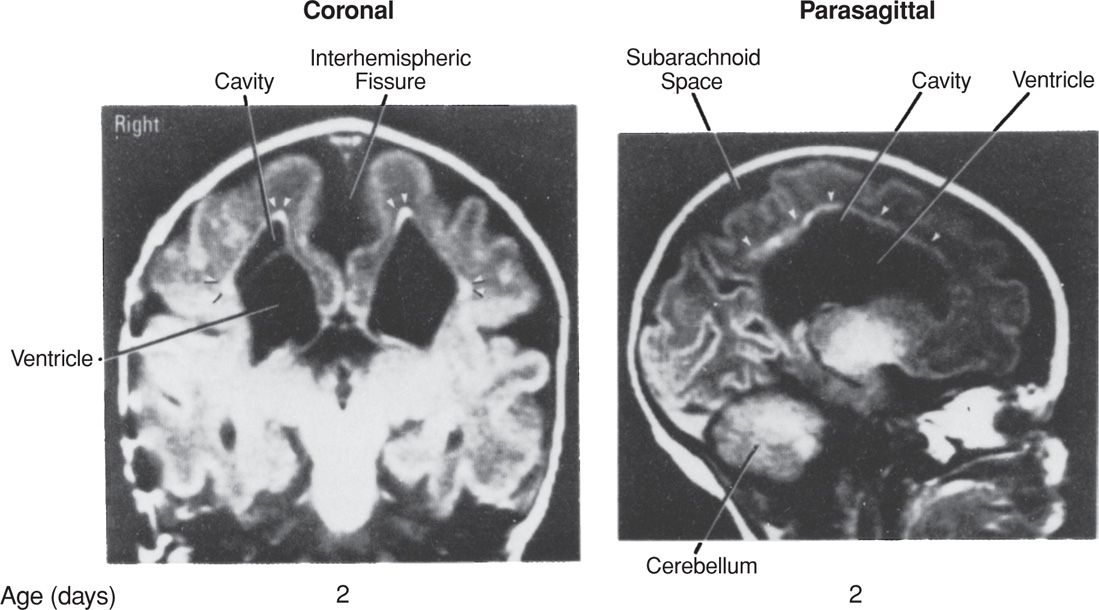
Fetal Brain Damage. Cerebral palsy, microcephaly, porencephaly, and multicystic encephalomalacia are serious complications associated with placental vascular anastomoses in multifetal gestation. The exact pathogenesis of neurological damage is not fully understood but is likely caused by ischemic necrosis leading to cavitary brain lesions (Fig. 45-22). In the donor twin, ischemia results from hypotension, anemia, or both. In the recipient, ischemia develops from blood pressure instability and episodes of severe hypotension (Lopriore, 2011). Cerebral lesions may also be due to postnatal injury associated with preterm delivery. Quarello and associates (2007) reviewed data from 315 liveborn fetuses from pregnancies with twin-twin-transfusion syndrome and reported cerebral abnormalities in 8 percent.
FIGURE 45-22 Cranial magnetic resonance imaging study of a diamnionic–monochorionic twin performed on day 2 of life. The subarachnoid space and lateral ventricles are markedly enlarged. There are large cavitary lesions in the white matter adjacent to the ventricles. The bright signals (arrowheads) in the periphery of the cavitary lesions most probably correspond to gliosis. (From Bejar, 1990, with permission.)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree