Lung Growth in Infancy and Childhood
Richard L. Auten
For the pediatrician, it is important to understand lung growth and development in order to properly diagnose and treat children for whom both may have gone wrong. It is crucial to the development of new diagnostic and treatment choices. This chapter will highlight the components of the developing lung, the factors that regulate their interaction, and the effects on pulmonary function in children.
Growth of the lung postnatally during infancy and childhood has typically been described in terms of the subdivision of alveoli, which accompanies the lengthening and widening of conducting airways, along with the vascular supplies for the growing bronchi and lung parenchyma.1 Investigations using animal models of mammalian lung development and disease increasingly reveal a dynamic process of lung cell turnover, repair, and regeneration with particular windows of vulnerability which will be important to pediatricians. Disrupted lung growth in early childhood has lifelong effects on lung function.2
CHILDHOOD EVENTS THAT PROGRAM ADULT LUNG FUNCTION
Students and trainees of recent vintage will doubtless have been introduced to the “Barker hypothesis,” which describes a relationship between early life events and the susceptibility to acquired disease states in adulthood. Cohort studies from England describing study subjects born 70 to 80 years ago linked the diagnosis of pneumonia before age 2 years with substantial decrements in lung function (forced expiratory volume in 1 second, abbreviated FEV1) adjusted for age and height.3 More recent reports from a similarly designed larger cohort study from England failed to demonstrate a significant association between childhood “chest diseases” such as asthma, pneumonia, and whooping cough, and an accelerated rate of decline in lung function, so the role of childhood lung infection and lung growth on later adult pulmonary function is not yet clear.4
On the other hand, premature birth, arguably a disruption of lung development at an early vulnerable period, is associated with impaired lung function in later childhood and the adult years, with significant effects on forced expiratory flows and wheezing. Structural changes in lung parenchyma observed in chest-computed tomography scans of adults who were born prematurely are common, but their functional significance is unclear.5 Disrupted alveolar development or altered capacities of repair that occur in early childhood may lower the threshold at which chronic age-related pulmonary diseases are manifested  . Improved understanding of the interactions of normal lung growth and environmental exposures will be indispensable to pediatricians now and in the future who seek to protect and preserve the function of the lung with its vulnerable interface with the “outside world.”
. Improved understanding of the interactions of normal lung growth and environmental exposures will be indispensable to pediatricians now and in the future who seek to protect and preserve the function of the lung with its vulnerable interface with the “outside world.”
PHASES OF LUNG DEVELOPMENT
Lung development (see Fig. 59-1) begins in embryonic life with outpouching of the tracheal primordium from foregut endoderm. Reciprocal signaling between epithelial cells lining the respiratory tract and underlying mesenchymal cells controls branching morphogenesis of the conducting airways. Toward the end of normal pregnancy, terminal respiratory saccules become lined with more flattened epithelium that will become type I pneumocytes that perform gas exchange in concert with pulmonary capillaries. Adjacent cuboidal epithelial cells in the primitive alveoli will become type II alveolar pneumocytes. Normal development of the lung during postnatal development in infancy and childhood is dominated by formation and differentiation of alveoli during the first years.
ALVEOLAR DEVELOPMENT
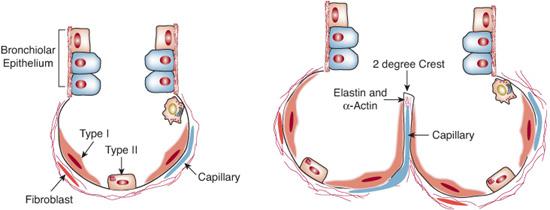
It has been proposed that alveolar homeostasis should be conceptualized as a functional “syncytium,” comprised of interdependent reciprocal signaling among pulmonary fibroblasts, pulmonary capillary endothelial cells, alveolar epithelial cells, and, after birth, resident alveolar macrophages (Fig. 502-1).6
Type I alveolar epithelium constitutes most of the alveolar surface area, and conducts most of the lung water trafficking through aqueporin channels necessary to keep alveoli from flooding. These epithelial cells overlie a matrix of mesenchyme-containing pulmonary fibroblasts within collagen, actin, and elastin fibers. Together with pulmonary capillaries, they form the alveolar septum, which attenuates and flattens during infancy, increasing surface area for gas exchange. Type II alveolar epithelial cells are metabolically active, synthesizing, secreting, and metabolizing pulmonary surfactant that allows alveoli to remain gas filled at the end of exhalation. These cells also appear to serve as a progenitor pool from which other type II cells and type I cells derive during development, repair, and regeneration.7 They secrete cytokines such as vascular endothelial growth factor (VEGF), which induce proliferation in neighboring capillary endothelial cells to allow extension of capillary tubes into alveolar septa during alveologenesis. Along with alveolar macrophages and bronchiolar epithelium, type II cells secrete proinflammatory cytokines and leukocyte chemokines in response to inflammatory stimuli.
The increases in lung surface area and relative reductions in alveolar size with lung growth are driven by flattening of the alveolar type I pneumocytes overlying elastin and collagen fibers in alveolar septa (Fig. 502-2). The actual number of alveoli and their rate of growth in infancy and early childhood are not precisely known, but the best available evidence suggests that additional alveoli develop at the extreme periphery of the growing lung, and that this process continues through age 2 years, with significant slowing thereafter. The specific contributions of paracrine factors from the cellular denizens of the alveolus during this stage of lung development have not been thoroughly studied.8
Alveolar repair and regeneration is believed to recapitulate alveolar growth in certain aspects. Lessons from animal models suggest that alveolar number is closely matched with metabolic demands and the size of the individual.9 Surgical removal of enough lung results in compensatory increases in lung volume in the remaining lung, with generation of more alveoli, if the loss of lung occurs early in postnatal development in animal models.10 There are very limited data in humans regarding the plasticity of alveolar number during infancy and childhood. Post-transplant increases in total alveolar volume estimated by measuring functional residual capacity suggest that near normal lung growth occurs in the transplanted lung in children less than age 3 years11; this appears to depend on the age of the transplanted lung, with immature lung demonstrating greater plasticity.12
Defects or disturbances in alveolar growth are most commonly found in children prematurely born and subjected to oxidative stress and intensive care aimed at treating respiratory insufficiency. The most common syndrome is bronchopulmonary dysplasia (BPD), a term used to describe the morphologic changes seen in conducting airways at autopsy, as a sequela of respiratory distress syndrome, before the development of surfactant replacement therapy. See Chapters 59 and 513.
Modern neonatal intensive care supports survival of less mature babies, with oxidative and mechanical insults taking place at earlier stages of lung development. Modern-day bronchopulmonary dysplasia (BPD) is believed to result in fewer, larger alveoli as demonstrated in a premature baboon model designed specifically to mimic modern neonatal intensive care.13 The lifelong growth potential of alveoli in children with BPD is unclear, but some evidence suggests that individuals born prematurely are at risk for airflow limitations, as well as emphysema in adulthood.14
Intervention with antenatal glucocorticoids just before preterm delivery is an example of a medical intervention at a specific stage of lung development that has been clearly shown to “accelerate” lung development and function, reducing the incidence and severity of respiratory distress syndrome (surfactant deficiency). Glucocorticoid administration just before preterm delivery augments surfactant synthesis and secretion and interstitial elastin deposition, which mark lung maturation and support alveolar structure, and are among many pathways beneficially affected.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


