Infections of the Central Nervous System
Kiran P. Maski and Nicole J. Ullrich
Infections of the central nervous system (CNS) are less frequent than infections of other organ systems, because the brain is protected by both the anatomic barrier of the bony skull and blood-brain barrier. Nevertheless, CNS infections remain a significant cause of morbidity and mortality in children. Classic signs and symptoms of CNS infections are often not present or more subtle in young children, thereby making accurate diagnosis a challenge for the general pediatrician (Table 555-1). The outcome of CNS infection predominantly depends on the site of infection and etiologic organism in addition to host immune status. For full description of bacterial and viral CNS infections, see Chapters 231 and 232. In this chapter, we focus on the neurologic presentation of CNS infections and discuss the unique CNS involvement of purulent CNS infections, latent viral infections, and prion diseases.
SIGNS AND SYMPTOMS OF CNS INFECTIONS
 MENINGITIS/MENINGOENCEPHALITIS
MENINGITIS/MENINGOENCEPHALITIS
The most common presentation of meningitis includes nonspecific signs and symptoms such as headache and nuchal rigidity. Meningismus refers to a condition in which the neck and back muscles tense as a reflex to avoid painful extension of the inflamed meninges. The physical findings of Kernig sign (passive extension of the knee from flexed thigh position elicits pain in the back) and Brudzinski sign (passive flexion of the neck elicits spontaneous flexion the lower extremities) are thought to reflect both the inflammation of the meninges and increased intracranial pressure. Nuchal rigidity is rare in neonates and uncommon in children ages 12 to 18 months. Other symptoms of meningitis vary depending on the etiology and location of the infection. The associated symptoms of fever, vomiting, lethargy, irritability, photophobia, anorexia, and dehydration all support the diagnosis.1 In neonates, a full fontanel may be present in addition to failure to thrive, irritability, apnea, seizures, opisthotonus, poor feeding, temperature instability, jaundice, and grey appearance.2
The anatomic boundaries between the meninges and brain parenchyma are indistinct. Many patients experience concurrent infections of the meninges and parenchyma, known as meningoencephalitis, often accompanied by signs of headache, generalized seizures, and mental status changes (delirium, agitation, somnolence, lethargy). Within the brain parenchyma, the inflammatory response is characterized by neutrophilic infiltrations and increased vascular permeability via alteration of the blood-brain barrier. Focal neurologic symptoms/signs may also be present including unilateral headache, paresis, cranial nerve palsy, or partial seizures. These findings may suggest a more localized involvement of brain parenchyma, or even focal infection, such as those caused by abscess or tuberculoma. Slowed blood flow secondary to infection within the parenchyma or meninges can result in acute vascular events such as venous infarction that then are also likely to produce more localizable neurologic signs. Patients who experience severely depressed consciousness, coma, vomiting, bulging fontanel, diastasis of sutures, or third or sixth cranial nerve palsy may have concurrent increased intracranial pressure secondary to inflammation from infection. These signs may also result from subdural effusions which develops in 10% to 30% cases of meningitis, particularly in infants.3Progression to decorticate/decerebrate posturing; papilledema; hyperventilation; and the triad of hypertension, bradycardia, and apnea suggests the development of dangerously high intracranial pressure and impending herniation. It is imperative to start antibiotic treatment first and then to consider neuroimaging if focal signs/symptoms and/or suspicion of increased intracranial pressure are present.
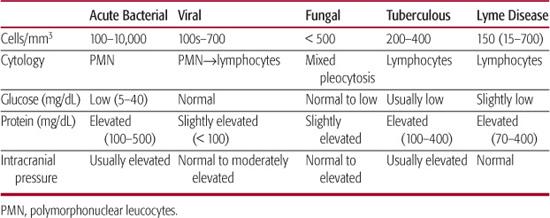
Meningitis is associated with seizure in up to 23% of children.4 In the younger child, it may be difficult to distinguish between a classic febrile seizure and a symptomatic seizure associated with fever resulting from underlying meningitis/encephalitis.  Meningitis should be suspected if seizures occur 3 or more days after the onset of fever. Meningeal signs may be absent in children younger than 18 months of age who present with fever and seizure and there should be a low threshold to perform cerebrospinal fluid analysis. The American Academy of Pediatrics strongly urges physicians to consider lumbar puncture in infant less than 12 months who present with febrile seizure.7 Careful attention should be made to changes in serum glucose, calcium and sodium (secondary to syndrome of inappropriate antidiuretic hormone [SIADH]) concentrations as seizure precipitants. Lumbar puncture results may also help to distinguish between viral, bacterial, parasitic, and fungal processes so that therapy can be tailored appropriately. Table 555-2 shows a comparison of cerebrospinal fluid (CSF) profiles associated with various CNS infectious agents. Specific agents causing bacterial and viral meningitis are listed in Tables 231-1 and 232-1.
Meningitis should be suspected if seizures occur 3 or more days after the onset of fever. Meningeal signs may be absent in children younger than 18 months of age who present with fever and seizure and there should be a low threshold to perform cerebrospinal fluid analysis. The American Academy of Pediatrics strongly urges physicians to consider lumbar puncture in infant less than 12 months who present with febrile seizure.7 Careful attention should be made to changes in serum glucose, calcium and sodium (secondary to syndrome of inappropriate antidiuretic hormone [SIADH]) concentrations as seizure precipitants. Lumbar puncture results may also help to distinguish between viral, bacterial, parasitic, and fungal processes so that therapy can be tailored appropriately. Table 555-2 shows a comparison of cerebrospinal fluid (CSF) profiles associated with various CNS infectious agents. Specific agents causing bacterial and viral meningitis are listed in Tables 231-1 and 232-1.
Table 555-1. Signs and Symptoms of CNS Infection
 CEREBELLITIS/BRAINSTEM ENCEPHALITIS
CEREBELLITIS/BRAINSTEM ENCEPHALITIS
Localized infections of the cerebellum can produce an acute cerebellar ataxia. Acute onset of gait difficulties, incoordination and nystagmus, slurred speech, and vomiting suggests either infectious or postinfectious cerebellitis. Fever is noted in nearly 75% of cases and latency of illness to onset of ataxia averages 9 days.8 The differential diagnosis of fever with cerebellar signs includes cerebellar abscess and acute labrynthitis. Rhomboencephalitis or brainstem encephalitis results from infection of the brainstem, specifically the pons and medulla. Symptoms/signs include meningismus, fever, and headache in addition to cerebellar dysfunction, hemi/quadriparesis, cranial nerve abnormalities (facial palsy, diplopia, dysphagia, dysautonomia), and opsoclonus. Cerebrospinal fluid (CSF) pleocytosis with increased protein and decreased glucose concentrations, and magnetic resonance imaging (MRI) that demonstrates increased signal in the pons/medulla, suggest the underlying diagnosis. In the pediatric population, Listeria,9 West Nile virus,10 enterovirus 71,11 and HHV612 infections have all been reported as etiologic agents of rhomboencephalitis.
Table 555-2. Cerebrospinal Fluid Profile in CNS Infections
 MYELITIS
MYELITIS
Infection of the spinal cord or infectious myelitis can also be categorized into focal and diffuse pathology. Certain viruses, such as poliovirus, Epstein-Barr virus, enterovirus, echovirus, and coxsackie virus exhibit tropism for the anterior horn cell.13 Infection of anterior horn cells produces asymmetric motor weakness with or without fever, encephalopathy, and nausea/vomiting. Headache and mental status changes suggest a more widespread meningoencephalitis. Weakness that extends to the face and bulbar nuclei can result in autonomic, respiratory, and circulatory compromise. If the infection is limited to the anterior horn cell, sensory examination may remain normal. Acute transverse myelitis (ATM) (see Chapter 556) is defined as inflammation of gray and white matter in one or more adjacent spinal cord segments. Acute transverse myelitis is thought to result either from direct infection or postinfectious immune mediated processes, leading to diffuse involvement in the spinal cord and subsequent sensory and motor dysfunction.14 Early signs of spinal cord dysfunction include paresthesias, back pain in segmental distribution, bilateral weakness, bowel/bladder dysfunction, and sensory level with dysesthesia or hyperalgesia. Autonomic dysfunction may or may not be present. Symptoms typically develop over 4 hours to 21 days ultimately with progression to flaccid paralysis and then spasticity over weeks. In the setting of ATM, magnetic resonance imaging (MRI) of the spine typically demonstrates T2 hyperintense lesions that extend more than 2 vertebral body lengths, often with cord expansion and variable contrast enhancement.15 Cerebrospinal fluid analysis demonstrates increased protein content, suggestive of inflammation. The constellation of symptoms including fever, back pain, point tenderness of the spinous processes, motor weakness, and/or sphincter compromise may suggest spinal epidural abscess (EDA). There are no clear-cut clinical diagnostic criteria for EDA, and signs and symptoms commonly overlap with ATM. Immediate MRI with and without gadolinium and intravenous (IV) antibiotics are indicated if concern for epidural abscess exists.
DIAGNOSIS OF CNS INFECTION
Neurologic signs and symptoms, laboratory investigations and neuroimaging studies can all help facilitate the localization of disease processes and provide guidance for management. After history and physical examination are complete, the decision to perform lumbar puncture (LP) or neuroimaging should be made. A concern is that if intracranial pressure (ICP) is elevated LP may precipitate herniation of the hindbrain through the foramen. Raised ICP is common in children with uncomplicated acute bacterial meningitis. Careful measurement of opening pressure with legs extended can help identify elevated ICP. In one study, 33 of 35 infants and children with pyogenic meningitis had a significant elevation in CSF opening pressure at the time of evaluation.18 The vast majority of children presenting with suspected bacterial meningitis, however, have no clinical evidence of herniation; therefore routine computerized tomography (CT) scan of the brain is not indicated.19 A diagnostic LP should be done expeditiously and appropriate antibiotic therapy should be promptly initiated.
A computerized tomography (CT) scan of the brain should be considered prior to lumbar puncture if patient presents with symptoms or signs of ICP such as depressed mental status, posturing, papilledema, increase in blood pressure with slow pulse rate, or focal neurologic symptoms or signs. 
Risk of herniation with lumbar puncture still exists even with normal head CT, and there is considerable variation in normal ventricular and cisternal size, making interpretation of a single scan unreliable.20 Cerebral edema may produce CT abnormalities including slit-like lateral and third ventricles, generalized low attenuation of the white matter, and obliteration of the basilar and suprachiasmatic cisterns.21 If concern for herniation exists based on clinical judgment, the decision to perform an LP may need to be deferred. When the child’s condition has stabilized, the need for CSF evaluation can be reconsidered.
 CEREBROSPINAL FLUID (CSF) INTERPRETATION
CEREBROSPINAL FLUID (CSF) INTERPRETATION
Evaluation of cerebrospinal fluid (CSF) can help determine the diagnosis of bacterial or viral meningitis or encephalitis (Table 555-2). Although CSF is considered an acellular space, 5 white blood cell (WBC) count and 5 red blood cell (RBC) count per μL are considered normal when CSF is sampled by lumbar puncture (LP); newborns, in contrast, may have up to 20 WBC/μL in the CSF.22 Pleocytosis is the rule in bacterial meningitis with CSF WBC counts in the range of 100 to 10,000/μL. Early in the disease, WBC in CSF may be normal. Polymorphonuclear cells predominate and usually account for more than 90% of the total. Very high WBC counts (> 50,000/μL) raise the possibility of intracranial abscess. CSF glucose is usually less than 50% of simultaneous serum glucose concentration, but low CSF glucose is also found in tuberculosis, fungal meningitis, subarachnoid hemorrhage, and carcinomatous meningitis. CSF protein concentration is usually elevated to 100 to 500 mg/dL in bacterial meningitis. The alteration in protein concentration reflects disruption of blood-brain barrier but is not in itself indicative of bacterial meningitis. Protein concentrations may be significantly elevated in patients with meningitis caused by Mycobacterium tuberculosis. Gram stain is positive in more than 90% of untreated meningitis and culture positive in 70% to 90% of patients with untreated meningitis. 
NEUROLOGIC OUTCOME OF CNS INFECTION
The range of neurologic sequelae from central nervous system (CNS) infection largely depends on the etiologic agent, and duration and location of infection. CNS infections also vary in severity. Organisms with tropism to the brain can take years to demonstrate clinical disease, as with infections caused by transformed prions and slow viruses, or may produce rapid inflammation and neuronal destruction, as in some cases of meningococcal bacterial or herpes simplex virus (HSV) meningitis.
The neurologic complications after bacterial meningitis include bilateral severe or profound deafness, mental retardation, seizures, and spasticity and/or paresis.24 By contrast, most children with viral meningitis recover spontaneously, though persistent fatigue, sleep disturbances, behavior and concentration difficulties, and incoordination have been noted.25 Diseases attributed to chronic latent viruses and prion diseases are considered neurodegenerative diseases with uniformly poor outcome. Focal purulent collections in the brain and spine have shown dramatic improvement in mortality and reduction in neurologic sequelae as a result of advances in neuromaging and improved medical and neuro-surgical techniques. Future investigations into the role of selective immunomodulation and neuroprotective strategies may lead to significant advances in management and outcome of CNS infections.
Table 555-1 briefly reviews the clinical presentation and management of specific CNS infections; many of the bacterial and viral infections are discussed in Section 17. This chapter focus on CNS abscesses, chronic latent infections and prion diseases.
INTRACRANIAL AND SPINAL ABSCESSES
A brain or spinal abscess is a focal, intraparenchymal infection that begins as a localized cerebritis and becomes encapsulated.26 Brain and spinal abscesses are relatively uncommon in pediatric patients, but are potentially neurologically devastating and life threatening. In children, the most common sources of infection are related to underlying cyanotic congenital heart disease, meningitis, chronic otitis media, mastoiditis, sinusitis, cellulitis of scalp, face, orbit, dental infection, penetrating head injury, immunodeficient states, and intracranial foreign bodies. Early diagnosis and treatment of brain abscess can be life saving. The diagnosis and treatment of both spinal and brain abscesses are discussed in detail in Chapter 231. 
SUBDURAL EMPYEMA
Subdural empyema (SDE) refers to a collection of infected cerebrospinal fluid (CSF) or pus between the dura and arachnoid spaces and accounts for 15% to 20% of all localized intracranial infections.51 Routine immunization for Haemophilus influenza has decreased the overall incidence because SDE was a common complication of H influenza meningitis.52 Now, the most common predisposing features are otorhinologic infections, especially paranasal sinus infections in children. Pathogens spread to the subdural space via valveless emissary veins in association with thrombophlebitis from infection of sinuses, mastoid, or middle ear or extension of osteomyelitis of the skull.53 Once infection reaches subdural space, it can easily spread over the convexities of the brain, extending to both hemispheres. Mastoiditis, untreated middle ear infection, skull trauma, neurosurgery, instrumentation of the ears or nose, and infection of preexisting subdural hematoma are known risk factors. In infants, SDE can result from poorly treated meningitis with infected subdural effusion, typically with the same hasorganism (Fig. 555-1).54 The causative agents of SDE represent those found in sinus and otic infections; aerobic and anaerobic streptococci are isolated in 44% to 60% of cases.55, 56 Postoperative and posttraumatic infections are often due to staphylococci and gram negative aerobic bacilli.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


