Congenital Abdominal Wall Defects
Abdominal wall defects are divided into omphalocele and gastroschisis. While often considered together, they are distinct and separate entities. Differences between gastroschisis and omphalocele are illustrated in Figure 48-1 and summarized in Table 48-1.
TABLE 48-1
Differentiating Characteristics between Gastroschisis and Omphalocele
| Characteristic | Omphalocele | Gastroschisis |
| Herniated viscera | Bowel ± liver | Bowel only |
| Sac | Present | Absent |
| Associated anomalies | Common (50%) | Uncommon (<10%) |
| Location of defect | Umbilicus | Right of umbilicus |
| Mode of delivery | Vaginal/cesarean | Vaginal |
| Surgical management | Nonurgent | Urgent |
| Prognostic factors | Associated anomalies | Condition of bowel |
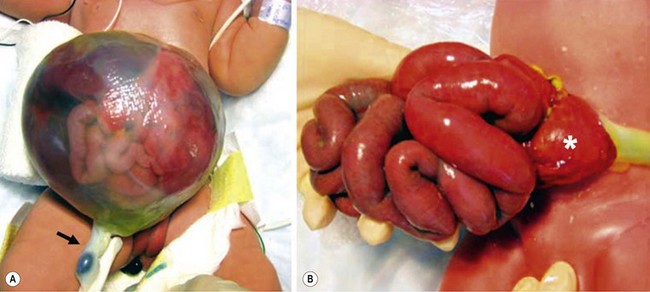
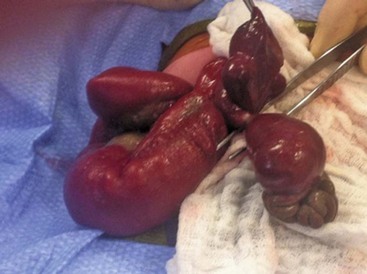
FIGURE 48-1 These two photographs nicely depict the differences between an omphalocele and gastroschisis. (A) In an omphalocele both the liver and bowel can be herniated. A sac is always present and the umbilical cord (arrow) inserts onto the sac. Moreover, this is always a midline defect. (B) With a gastroschisis, the liver is never herniated and a sac is absent. The location of the fascial defect is to the right of the umbilicus, and the umbilical cord is attached to the umbilicus. In addition to the large and small intestine, the stomach (asterisk) can sometimes be herniated as well.
Embryology and Etiology
The etiology for gastroschisis is less clear. One theory suggests that gastroschisis results from failure of the mesoderm to form in the anterior abdominal wall. Currently, the ventral body folds theory, which suggests failure of migration of the lateral folds (more frequent on the right side), is most widely accepted.1 This implies a gastroschisis develops early in gestation and prior to development of an omphalocele. Due to the increasing incidence of gastroschisis, there are a number of possible causative factors including tobacco, certain environmental exposures, lower maternal age and low socioeconomic status, all suggested by epidemiologic studies, but not proven.1–4
Gastroschisis
Prenatal Management and Diagnosis
Gastroschisis occurs in 1 in 4,000 live births.5 An increased incidence in mothers younger than 21 years of age has been widely documented.6 There has also been a significant worldwide increase in the incidence of gastroschisis in all age groups over the past two decades.7 Preterm delivery is more frequent in infants with gastroschisis, with an incidence of 28% compared with only 6% in babies without an abdominal wall defect.8
The majority of pregnancies complicated by gastroschisis are diagnosed sonographically by 20 weeks’ gestation.2,3 Often an ultrasound (US) evaluation is performed because of an abnormal maternal serum α-fetoprotein (AFP) level, which is universally elevated in the presence of gastroschisis.9,10 Detection of bowel loops freely floating in the amniotic fluid and a defect in the abdominal wall to the right of a normal umbilical cord are diagnostic of gastroschisis. Intrauterine growth retardation (IUGR) has been noted in a large number of these fetuses as well.11
Some authors advocate selective preterm delivery based on the finding of bowel distention and thickening on prenatal ultrasound.12 Dilated fetal bowel has been shown to correlate with a worse outcome, including fetal distress and demise in some series, but not in others.13 One problem with using bowel dilatation to predict outcome is the lack of a definition of ‘dilated,’ with ranges from 7 to 25 mm being considered abnormal.14,15 Moreover, there is also variability in the part of the intestine that is being measured. Studies in animal models have shown that the duration of amniotic fluid exposure is correlated with the degree of the inflammatory peel and intestinal dysmotility.16–19 Efforts to reduce this exposure by either amniotic fluid exchange or intrauterine furosemide treatment, which induces fetal diuresis, have shown to be beneficial in animals.19–22 These studies spurred early human trials with amniotic fluid exchange in Paris and Italy, but these trials have proven inconclusive.20,23
Concomitant bowel atresia is the most common associated anomaly in patients with gastroschisis, with rates ranging from 6.9–28% in several series (Table 48-2).24,25 A recent literature review noted associated anomalies in the cardiac, pulmonary, nervous, musculoskeletal, genitourinary systems, as well as chromosomal abnormalities in babies with gastroschisis.33
Perinatal Care
The optimal mode and timing of delivery for a fetus with gastroschisis has been debated for many years. Proponents of routine cesarean delivery (C-section) argue that the process of vaginal birth results in injury or increased risks for infection and sepsis.34 However, the literature suggests that both vaginal delivery and C-section are safe.35,36 A recent meta-analysis failed to demonstrate a difference in outcomes for infants delivered either vaginally or by C-section.37 Therefore, the delivery method should be at the discretion of the obstetrician and the mother, with C-section reserved for obstetric indications or fetal distress.
Preterm delivery of the fetus with gastroschisis has been advocated to limit exposure of the bowel to the amniotic fluid.12 Interleukin-6, interleukin-8, and ferritin are elevated in the amniotic fluid in fetuses with gastroschisis when compared with controls.18,38 Damage to the pacemaker cells and nerve plexi may contribute to the profound dysmotility and malabsorption seen in these infants.39 Early delivery may mitigate these effects, but the literature is mixed.12 A randomized trial from the UK found no benefit after induced early delivery with the only trends being an improvement in length of hospitalization and earlier initiation of feeding.40 Another study demonstrated that birth weight less than 2 kg was associated with increased morbidity.41 Currently available evidence does not support elective preterm delivery for gastroschisis.42
Neonatal Resuscitation and Management
Neonates with gastroschisis have significant evaporative water losses from the open abdominal cavity and exposed bowel. Appropriate intravenous access should be obtained and fluid resuscitation initiated after birth. Nasogastric (NG) decompression is important to prevent further gastric and intestinal distention. Routine endotracheal intubation is not necessary. The bowel should be wrapped in warm saline-soaked gauze and placed in a central position on the abdominal wall. The neonate should be positioned on the right side to prevent kinking of the mesentery with resultant bowel ischemia. The bowel should be wrapped with plastic wrap or the infant placed partially in a plastic bag to reduce evaporative losses and improve temperature homeostasis (Fig. 48-2). Although gastroschisis most often is an isolated anomaly, thorough examination of the neonate is important. In addition, the bowel must be carefully examined for intestinal atresia, necrosis, or perforation (Fig. 48-3). Recent evidence suggests that excess fluid resuscitation is detrimental and results in edema, an increase in time to closure, and an increased risk of abdominal compartment syndrome.43
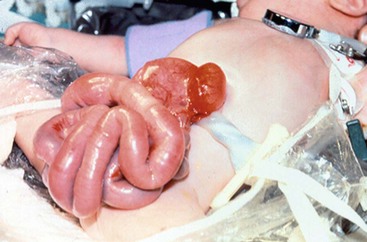
FIGURE 48-2 Photograph of a neonate with gastroschisis who has been transported wrapped with a bowel bag. The bag has been untied to allow inspection of the herniated bowel.
Surgical Management
The primary goal is to return the viscera to the abdominal cavity while minimizing the risk of damage due to trauma or increased intra-abdominal pressure. The two most commonly used treatment options are placement of a silo followed by serial reductions and delayed closure, or attempted primary closure.44 The timing and location of surgical intervention is also controversial.45 In all cases, inspection of the bowel for obstructing bands, perforation, or atresia must be undertaken. Bands crossing the bowel loops should be lysed before silo placement or primary abdominal closure to avoid subsequent intestinal obstruction.
Primary Closure
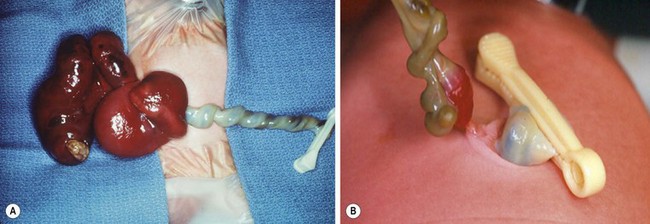
Historically, urgent primary closure of gastroschisis was advocated in all cases. This approach is still commonly practiced in neonates in whom reduction of the herniated viscera appears possible.46 Attempted primary closure has traditionally been performed in the operating room, but some authors have advocated primary closure at the bedside without general anesthesia.47–49 Some surgeons prefer to close the skin only and leave the fascia separated. Others have described the use of the umbilicus as an allograft.50,51 Prosthetic options for fascial closure include nonabsorbable mesh or bioprosthetic materials such as porcine small intestinal submucosa mesh.52 In the past, most surgeons have excised the umbilicus during closure. However, preservation of the umbilicus has been shown to lead to an excellent cosmetic result (Fig. 48-4).53,54
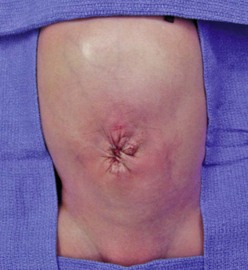
FIGURE 48-4 An excellent cosmetic result after gastroschisis repair in which the umbilicus was preserved.
Intra-abdominal pressure approximated from either the bladder pressure or stomach pressure can be used to guide the surgeon during reduction.55 Pressures higher than 10–15 mmHg are often associated with decreased renal and intestinal perfusion, and a silo or patch may be needed.56 Pressures higher than 20 mmHg can lead to renal failure and bowel ischemia.57 Similarly, an increase in central venous pressure greater than 4 mmHg has been correlated with the need for silo placement or patch closure.58 Splanchnic perfusion pressure, the difference between mean arterial pressure and intra-abdominal pressure, has also been used to guide the reduction. A splanchnic perfusion pressure less than 44 mmHg implies a decrease in intestinal blood flow.59
Staged Closure
In the mid-1990s, a prefabricated silo was developed with a circular spring that is positioned under the fascial opening, without the need for sutures or general anesthesia.60 This has made it possible to insert the silo in the delivery room or at the bedside (Fig. 48-5). After placement, the bowel is reduced daily into the abdominal cavity as the silo is shortened by sequential ligation. When the contents are entirely reduced, fascial and skin closure are performed. This process usually takes between one and 14 days with the majority being ready within a week, depending on the condition of the bowel and the infant.
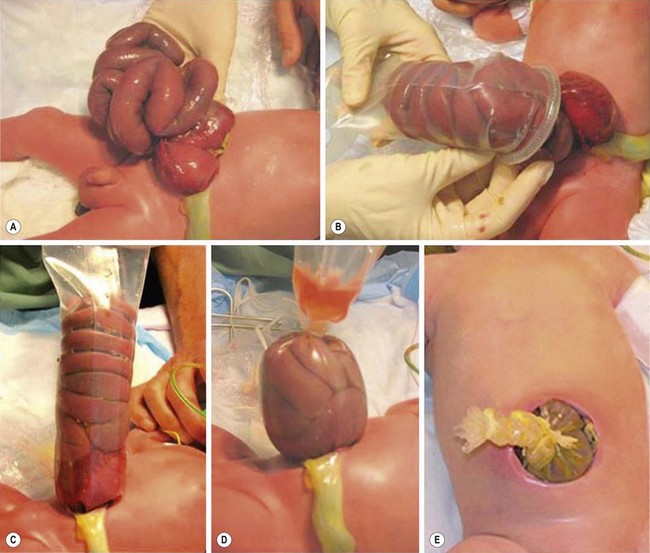
FIGURE 48-5 The use of a spring-loaded prefabricated silo is shown in these photographs. (A) The gastroschisis defect is seen. (B) An appropriate-sized spring-loaded silo is then placed over the eviscerated intestine. (C) The ring of the silo has been positioned under the fascial defect and attached to an overhead support to keep the bowel from torquing, which may result in intestinal ischemia. (D) Gradual reduction of the silo is performed. (E) Finally, the bowel has been completely returned to the abdominal cavity and the neonate is ready for transport to the operating room for closure of the fascia and skin.
Definitive closure in the operating room consists of raising small skin flaps around the fascial defect followed by fascial closure in a horizontal or vertical direction. Closure of the skin in a transverse direction creates a ‘keyhole’ appearance with a horizontal scar to the right of the umbilicus. This is why some surgeons advocate a vertical closure to allow for a central umbilicus. Also, a purse-string skin closure around the umbilicus can be performed to create a circular scar for improved cosmesis. Recently, the ‘plastic closure’ method has been described in which the umbilical cord is tailored to fill the gastroschisis defect and is then covered with an adhesive dressing.61,62 If the umbilical cord is not salvageable, the bowel can be covered with the dressing. Ingrowth of granulation tissue and epithelialization occurs over time. With this technique, an operation and general anesthesia can be avoided in many infants. Residual ventral hernia rates are reported to be 60–84%, the majority of which close spontaneously.54
The routine use of a preformed silo has increasingly come into favor, with the theory being that avoidance of high intra-abdominal pressure will avoid ischemic injury to the viscera and allow earlier extubation.60,63 One study reported fewer days on mechanical ventilation for patients undergoing silo reduction when compared to primary closure.47 However, there was no difference in time to full feeds or days on parenteral nutrition. Two reports noted a similar time to full enteral feedings but found that primary closure was associated with higher mean airway pressures, oxygen requirement, vasopressor requirement, and decreased urine output.64,65 However, other reviews have suggested that the trend to use a silo instead of immediate closure may have swung too far.66,67 Also, recent data from the Canadian Pediatric Surgeons Network (CAPSNet) database showed that infants who are able to undergo primary closure require less parenteral nutrition and hospitalization when compared with those who required staged reduction and repair.68 An attempt at a prospective randomized multicenter trial looking at silo vs. attempted primary closure that centered on ventilator days did not meet accrual targets and failed to show any significant difference.69 A similar trial has begun in the UK.70 Current data suggests that there is no significant difference in outcome with either approach (silo vs. immediate closure) for patients with uncomplicated gastroschisis.
Management of Associated Intestinal Atresia
Up to 10% of neonates with gastroschisis have an associated atresia, most commonly jejunal or ileal. In a database review of 4,344 infants with gastroschisis, a 5% incidence of small bowel atresia and a 2% incidence of large bowel atresia was noted.24 These atresias can be treated at the time of abdominal wall closure with resection and primary anastomosis in cases where there is minimal inflammatory peel. If the condition of the bowel makes primary anastomosis inadvisable, the bowel is reduced with the atresia intact and repair is undertaken four to six weeks after the initial abdominal wall closure.71 Some surgeons have chosen to create a stoma, particularly in the case of a distal atresia, to allow for enteral feeding while awaiting repair.72 If perforation is found, the perforated segment can be resected and a primary anastomosis performed if the inflammation is minimal (Fig. 48-6). Alternatively, an ostomy can be created followed by ostomy closure at a later date.73 There is no consensus about the optimal management for these complicated problems (see Table 48-2). Patients with an atresia are considered to be ‘complex’ to differentiate them from the patients without any such association (simple). Most authors note worse outcomes with complex cases.24,70,74
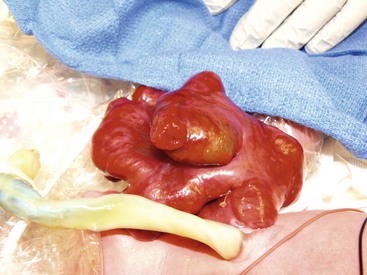
FIGURE 48-6 This newborn presented with gastroschisis and intestinal perforation. Note the two lumens in the exposed segment of intestine. Because there was not significant inflammation, this perforation was closed primarily and the bowel was placed in a silo. The baby recovered uneventfully.
An intestinal atresia should be differentiated from ‘vanishing bowel’ in infants with gastroschisis. This condition is usually associated with a very small abdominal wall defect and is characterized by necrosis and disappearance of some or all of the intestine (Fig. 48-7). Although this is a rare finding, it usually results in short bowel syndrome.75,76
Postoperative Course
Prokinetic medication may be helpful. In a rabbit model of gastroschisis, cisapride improved contractility of newborn intestine whereas erythromycin improved motility in control adult tissue only.77 However, a randomized controlled trial of erythromycin versus placebo found that enterally administered erythromycin did not improve time to achieve full enteral feedings.78 A similar randomized trial examining the use of cisapride in postoperative neonates, most of whom had gastroschisis, showed a beneficial effect, but this drug is not available in the USA.79
Long-Term Outcomes
Long-term outcomes for patients born with gastroschisis are generally excellent. The presence of complex disease is the most important prognostic determinant for a poor outcome.80,81 A number of authors have attempted to stratify patients with gastroschisis according to risk.82
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree