Amebiasis
Ibne Karim M. Ali and William A. Petri Jr.
Amebiasis denotes the disease caused solely by Entamoeba histolytica, although there are two other morphologically identical Entamoeba species that can also infect humans—Entamoeba dispar and Entamoeba moshkovskii. E dispar is about 10 times more prevalent than E histolytica in most endemic areas for amebiasis.1 Recent reports suggest that infection of E moshkovskii, which was once considered a free-living amoeba, is also common in some parts of the world such as Bangladesh, India, and Australia. Most E moshkovskii infections occur concomitantly with E dispar or E histolytica infections. Neither E dispar nor E moshkovskii infections are associated with disease.2 Free-living amoebic infections are discussed in Chapter 348.
 EPIDEMIOLOGY
EPIDEMIOLOGY
Amebiasis occurs worldwide but is much more common in developing nations. This is a result of contaminated water or food, leading to fecal-oral spread of the cyst. The World Health Organization estimates that E histolytica is second only to malaria as a protozoan cause of death. For example, the 1988 Mexican national sero-survey demonstrated serological evidence of E histolytica infection in 8.4% of the population.1 Nearly half of the children surveyed in a refugee camp in Dhaka, Bangladesh, had evidence of infection by age 5. Amebic dysentery is most common in grade-school children.3 Amebic liver abscess is 10 times more common in men than in women, being most common in men between the ages of 20 and 50 years.1,4 This male sex predominance is not observed in children. Steroid treatment and pregnancy appear to increase susceptibility to life-threatening infection. In the United States, most infections occur in immigrants or in those who travel to developing countries. Amebic liver abscess may present clinically with symptoms more than 6 months after travel to an endemic area. Residents of institutions for the mentally retarded and HIV-infected individuals are also at greater risk of E histolytica infection.
 PATHOPHYSIOLOGY
PATHOPHYSIOLOGY
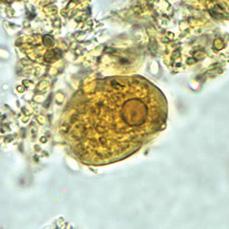
FIGURE 341-1. Trophozoite of E. histolytica/E. dispar in direct wet mount stained with iodine. (Courtesy of CDC: http://www.dpd.cdc.gov/dpdx/HTML/Amebiasis.htm; accessed August 14, 2010.)
E histolytica is a protozoan with an invasive, motile trophozoite and infectious cyst stages that is responsible for person to person transmission of infection. The trophozoite varies in diameter from approximately 10 to 60 μm, has a clear ectoplasm, and a single nucleus. The cyst averages 12 mm in diameter and has 1–4 nuclei (Figs. 341-1 and 341-2). Humans are the only reservoir for E. Histolytica. Cysts that are passed in the feces of infected individuals survive in a moist environment for months. Following their ingestion in contaminated food or water the cysts travel to the small intestine, the multinucleated metacystic amoeba is activated and emerges through a hole in the cyst wall, and immediately after excysting it undergoes division into eight uninucleate trophozoites. These organisms do not colonize the small intestine but are usually carried to the cecum where they become established.
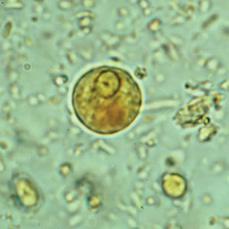
FIGURE 341-2. Cyst of E. histolytica/E. dispar in a wet mount stained with iodine. (Courtesy of CDC: http://www.dpd.cdc.gov/dpdx/HTML/Amebiasis.htm)
In 90% of patients, the trophozoites reencyst and produce asymptomatic infection which usually spontaneously resolves within 12 months. In 10% the parasite causes symptoms. Invasion of trophozoites then occurs, causing intestinal and hepatic abscesses.1
Trophozoites colonize the intestine by adhering to colonic mucin glycoproteins via a galactose and N-acetyl-D-galactosamine (Gal/GalNAc)–specific lectin. The remarkable tissue destruction for which the organism is named requires the parasite to contact host cells via the same Gal/GalNAc lectin. Parasites that have invaded humans resist destruction by the complement arm of the innate immune system via Gal/GalNAc lectin-mediated inhibition of assembly of the membrane attack complex.1,5
Human and parasite genetic differences are likely to play important roles in determining the pathogenecity of infection. Certain human HLA class II alleles appear to provide independent leptin receptor polymorphisms appear to provide protection against E histolytica infection. On the other hand, E histolytica genotypes have been shown to differ among those infected and presenting with no symptoms, diarrhea/dysentery or liver abscess.6 A cell-mediated immune response is likely to be important in clearing established infection by generating interferon-γ and tumor necrosis factor-α to activate macrophages and neutrophils to kill the trophozoite.3
Hepatic lesions illustrate lytic destruction of the hepatic parenchyma with abscess formation. The smallest lesions can measure a few millimeters in diameter, whereas others can extend to destroy most of the liver.
 CLINICAL MANIFESTATIONS
CLINICAL MANIFESTATIONS
Many infections with E histolytica, and all E dispar or E moshkovskii infections, are without symptoms.3 Asymptomatic individuals are referred to as carriers, or cyst passers. It is not unusual for a carrier of E histolytica to develop invasive amebiasis months later. Illness that is attributable to amebiasis can have an acute or gradual onset with mild to severe symptoms. Amebiasis occasionally can have a rapid, fulminant course. More often, there is a chronic course of a cyclical nature, consisting of mild symptoms, alternating with moderate to severe manifestations.
The clinical incubation period varies from approximately 4 days to possibly years but is usually a week to several months. Severe disease may be characterized by the sudden onset of frequent, copious diarrhea, usually containing mucus and blood; but more often, the symptoms develop gradually, with irregular bouts of diarrhea, abdominal pain, nausea, and loss of appetite. Weight loss is seen in half of patients. Erythrophagous trophozoites can be observed in the stool in as many as one third of cases of amebic colitis.3 Low-grade fever and leukocytosis are present in less than one half of patients. If the febrile reaction is marked, or if there is considerable polymorphonucleocytosis, an amebic liver abscess should be considered. In severe intestinal disease, palpation of the abdominal wall will reveal exquisite tenderness along the portion of the involved large bowel. Colonoscopy often reveals discrete ulcers that vary in size from a pinhead to large, coalesced lesions with overhanging necrotic edges but it is not unusual to see a diffusely inflamed mucosa resembling that of a nonspecific ulcerative colitis. Occasionally, the pathological process extends through the serosa and leads to perforation. Disease may be limited to the cecum. Barium enema examination may be normal.
Amebomas most frequently occur in the cecum, although they have been reported in all parts of the colon. The basic lesion consists of a granulomatous thickening of the colon that results from lytic necrosis followed by secondary pyogenic inflammation, leading to fibrosis, proliferative granulation tissue, and focal abscesses. The lesion may be well localized and can be mistaken for a tumor, or the colonic wall may be extensively involved.7
Liver Abscess
Liver abscess is the most frequent complication of amebiasis. About one third to almost one half of patients have no history of diarrhea.4 Abscesses usually are found in the right lobe of the liver, although this location is not helpful for distinguishing amebic from pyogenic (bacterial) abscesses. Clinically, examination reveals an enlarged liver and tenderness in the right upper quadrant.4 Polymorphonuclear leukocytosis is usually greater than 12,000/uL, and there is moderate anemia. The erythrocyte sedimentation rate is elevated, and chills with daily remitting fever of 39°C (102.2°F) to 40°C (104°F) are frequent. Abnormalities on routine chest radiography have been reported in 25% to 90%% of patients with amebic abscesses of the liver. Frequently, the right hemidiaphragm is elevated, which is of great diagnostic significance when present in the absence of a palpable hepatic mass. Furthermore, there may be consolidation at the base of the right lung or a right pleural effusion. At times, pain is referred to the right shoulder or the right lower quadrant of the abdomen.
Abscesses of the liver’s left lobe may present as an epigastric mass that frequently is mistaken for a neoplasm. These may rupture intraabdominally or into the pericardial sac with dire consequences. A right hepatic abscess may extend through the diaphragm into the right chest cavity or the pulmonary parenchyma, subsequently rupturing and draining through a bronchus. Jaundice is seen in approximately 10%% of amebic abscesses and usually is mild, but it can be severe with large abscesses. The serum alkaline phosphatase is moderately elevated in about two thirds of abscesses in adults. Such elevations in children may be difficult to interpret.
Primary Amebic Abscesses of the Lung and Brain
Primary amebic abscesses of the lung are rare. Lung involvement usually is secondary to hepatic abscess. Similarly, amebic brain abscesses are unusual and secondary to extraintestinal disease, especially hepatic, although several examples of direct hematogenous dissemination from the colon have been reported.
Amebiasis of the Skin
Amebiasis of the skin is usually secondary to perforation of the abdominal wall after an anterior amebic abscess ruptures. It also may occur when the rectum is perforated by a fistula or sinus tract that extends to the perineal skin, or it may occur as perianal extension of amebic colitis. These lesions may be extremely painful and are likely to become secondarily infected.
 DIAGNOSIS
DIAGNOSIS
Evaluation for amebic colitis should be considered in all patients in patients with colitis. Even in those patients with typical ulcers seen by colonoscopy, parasites may be found either in stool, or upon biopsy of lesions.
Identifying E histolytica in stool requires a specific antigen detection or PCR technique.8 Microscopy is an obsolete method that is unable to distinguish the more frequent nonpathogenic E dispar or E moshkovskii from E histolytica. In addition, microscopy misses up to two thirds of the infections detected by antigen tests or PCR. A stool antigen-detection test from TechLab (Blacksburg, VA) is the sole antigen-detection test commercially available for the specific diagnosis of E histolytica. It has comparable sensitivity and specificity to PCR but is much less cumbersome technically.8 The antigen-detection test takes 2 hours to perform in an EIA format and requires fresh (not formalin- or PVA-fixed) stool samples. Real-time PCR also has a high sensitivity and specificity for diagnosis of E histolytica, E dispar, and E moshkovskii; however, it requires sophisticated equipment and experienced technicians to perform, which are often absent in many poor countries where these parasites are endemic.
Serological tests are an important adjunct to antigen detection. Especially in the case of amebic liver abscess, most such patients do not have detectable parasites in stool.7 Tests for antiamebic antibodies are approximately 90% sensitive for amebic liver abscess and 70% sensitive for amebic colitis. The serological tests remain positive for years after an episode of amebiasis. As a result, a substantial number (between 10% and 35%) of residents of developing countries have antiamebic antibodies detected by current serological tests.7
Colonoscopy is preferable to sigmoidoscopy for the diagnosis of amebic colitis, because disease may be localized to the cecum or to the ascending colon. Wet preps of material scraped or aspirated from the base of ulcers should be examined for motile trophozoites. Biopsy specimens should be taken from the edge of the ulcers. Periodic acid-Schiff stains the parasites a magenta color and improves detection in biopsies.
Liver abscess usually is diagnosed by serological tests in combination with a radiological study (ultrasound, computer tomography, or magnetic resonance imaging) that demonstrates a defect in the liver (eFig. 341.1  ).7 Amebic liver abscesses on CT scans are usually rounded, well-defined, and low-attenuation lesions. The wall commonly enhances with contrast. None of these characteristics are sufficiently specific to differentiate a pyogenic from an amebic liver abscess. Until more specific diagnostic techniques are developed, the diagnosis of amebic liver abscess relies on detecting the risk factors for E histolytica infection, a lesion in the liver, and a positive serological test. Detecting E histolytica lectin antigen in sera and liver abscess pus samples of amebic liver abscess patients has been reported. However, prior antiamebic treatment in the patients with metronidazole significantly reduces the sensitivity of amebic antigen detection.7 Diagnostic aspiration under CT or ultrasonographic guidance may yield typical red-brown “anchovy paste” material, although the aspirate is more often yellow or gray-green. Typically, the aspirate is sterile (ie, no bacteria and no odor). This finding strongly suggests an amebic etiology for the abscess. Amoebae are infrequently seen by direct examination, but they often can be identified in the fluid by antigen detection or PCR.
).7 Amebic liver abscesses on CT scans are usually rounded, well-defined, and low-attenuation lesions. The wall commonly enhances with contrast. None of these characteristics are sufficiently specific to differentiate a pyogenic from an amebic liver abscess. Until more specific diagnostic techniques are developed, the diagnosis of amebic liver abscess relies on detecting the risk factors for E histolytica infection, a lesion in the liver, and a positive serological test. Detecting E histolytica lectin antigen in sera and liver abscess pus samples of amebic liver abscess patients has been reported. However, prior antiamebic treatment in the patients with metronidazole significantly reduces the sensitivity of amebic antigen detection.7 Diagnostic aspiration under CT or ultrasonographic guidance may yield typical red-brown “anchovy paste” material, although the aspirate is more often yellow or gray-green. Typically, the aspirate is sterile (ie, no bacteria and no odor). This finding strongly suggests an amebic etiology for the abscess. Amoebae are infrequently seen by direct examination, but they often can be identified in the fluid by antigen detection or PCR.
 TREATMENT
TREATMENT
Asymptomatic infection with E histolytica should be treated with a luminal agent alone; E dispar infection does not require treatment. Oral agents effective against luminal infection include diloxanide furoate (available only through the Centers for Disease Control and Prevention), paromomycin, and iodoquinol (Table 323-1). Invasive amebiasis (colitis, liver abscess, etc) should be treated with metro-nidazole or tinidazole for 10 days followed by a luminal agent; otherwise, patients are at risk of relapsing from residual infection in the intestine.1
Paromomycin and diloxanide furoate are associated with primarily gastrointestinal side effects, including diarrhea, nausea and vomiting, and flatulence. Diloxanide furoate is also associated with urticaria and pruritus. Metronidazole and tinidazole have gastrointestinal side effects, including anorexia, nausea, vomiting, diarrhea, and abdominal discomfort. They also have an unpleasant taste and trigger a disulfiram-like intolerance reaction to alcohol.
Fever remits after 3 to 4 days of treatment with metronidazole in the majority of patients with amebic liver abscess. For the rare patient that does not respond to metronidazole alone, the addition of chloroquine and/or percutaneous drainage of the liver abscess are useful.
No vaccine is available, but prototype subunit vaccines based on the Gal/GalNAc lectin and serine-rich protein are under study. Studies in Bangladesh have demonstrated that individuals with mucosal IgA antibodies to the surface Gal/GalNAc lectin of E histolytica show a reduced risk of acquiring a new infection with E histolytica, offering promise for vaccination.9
REFERENCES
See references on DVD.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


