Pathogenesis and Transmission
The causative agent for syphilis is Treponema pallidum. Minute abrasions on the vaginal mucosa provide an entry portal for the spirochete, and cervical eversion, hyperemia, and friability increase the transmission risk. Spirochetes replicate and then disseminate through lymphatic channels within hours to days. The incubation period averages 3 weeks—3 to 90 days—depending on host factors and inoculum size. The early stages of syphilis include primary, secondary, and early latent syphilis. These are associated with the highest spirochete loads and transmission rates of up to 30 to 50 percent. In late-stage disease, transmission rates are much lower because of smaller inoculum sizes.
The fetus acquires syphilis by several routes. Spirochetes readily cross the placenta to cause congenital infection. Because of immune incompetence prior to midpregnancy, the fetus generally does not manifest the immunological inflammatory response characteristic of clinical disease before this time (Silverstein, 1962). Although transplacental transmission is the most common route, neonatal infection may follow after contact with spirochetes through lesions at delivery or across the placental membranes. Increased maternal syphilis rates have been linked to substance abuse, especially crack cocaine; inadequate prenatal care and screening; and treatment failures and reinfection (Johnson, 2007; Lago, 2004; Trepka, 2006; Warner, 2001; Wilson, 2007). A report from Maricopa County, Arizona, also cited minority race or ethnicity as a risk factor (Kirkcaldy, 2011).
 Clinical Manifestations
Clinical Manifestations
Maternal syphilis can cause preterm labor, fetal death, fetal-growth restriction, and neonatal infection (Krakauer, 2012; Saloojee, 2004). Any stage of maternal syphilis may result in fetal infection, but risk is directly related to maternal spirochete load (Fiumara, 1952; Golden, 2003). Maternal syphilis is staged according to clinical features and disease duration:
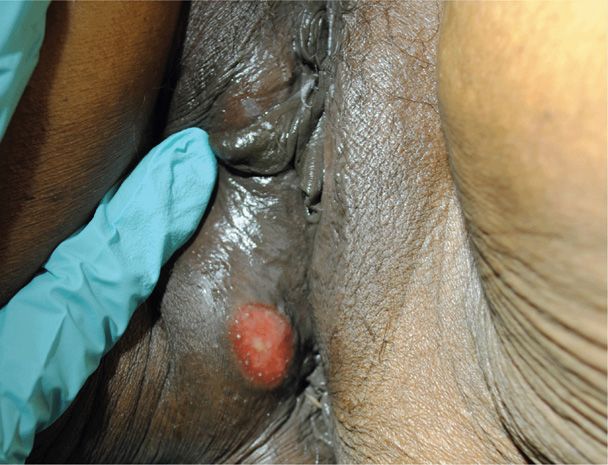
1. Primary syphilis is diagnosed by the characteristic chancre, which develops at the inoculation site. It is usually painless, with a raised, red, firm border and a smooth base (Fig. 65-1). Nonsuppurative lymphadenopathy may develop. A chancre will usually resolve spontaneously in 2 to 8 weeks, even if untreated. Multiple lesions may be found, predominantly in HIV-1 co-infected women.
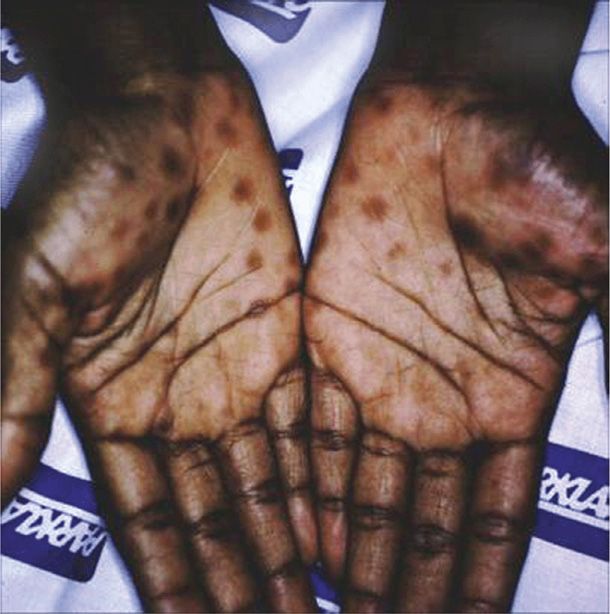
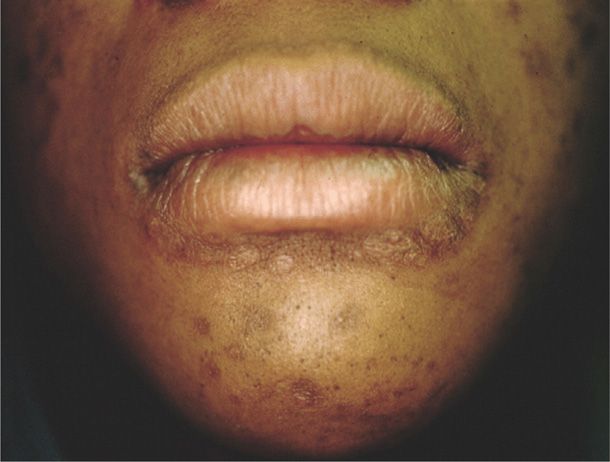
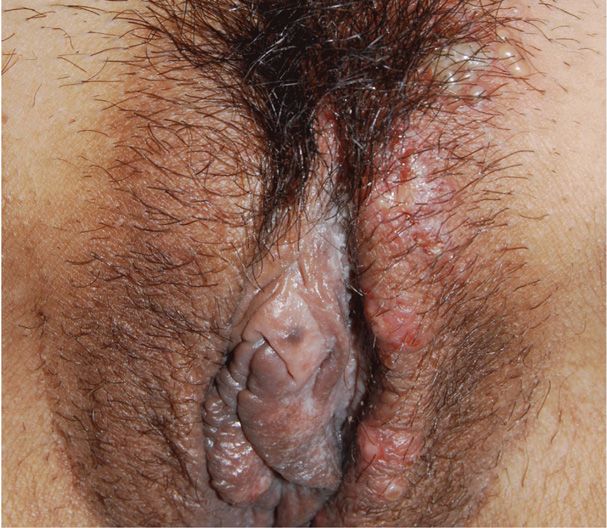
2. Secondary syphilis is diagnosed when the spirochete is disseminated and affects multiple organ systems. Manifestations develop 4 to 10 weeks after the chancre appears and include dermatological abnormalities in up to 90 percent of women. A diffuse macular rash, plantar and palmar targetlike lesions, patchy alopecia, and mucous patches may be seen (Figs. 65-2 and 65-3). Condylomata lata are flesh-colored papules and nodules found on the perineum and perianal area. They are teeming with spirochetes and are highly infectious. Most women with secondary syphilis will also express constitutional symptoms such as fever, malaise, anorexia, headache, myalgias, and arthralgias. Up to 40 percent will have cerebrospinal fluid abnormalities, although only 1 to 2 percent will develop clinically apparent aseptic meningitis. Hepatitis, nephropathy, ocular changes, anterior uveitis, and periostitis may also develop.
3. Latent syphilis develops when primary or secondary syphilis is not treated. It is characterized by reactive serological testing, but resolved clinical manifestations. Early latent syphilis is latent disease acquired within the preceding 12 months. Disease diagnosed beyond 12 months is either late latent syphilis or latent syphilis of unknown duration.
4. Tertiary or late syphilis is a slowly progressive disease affecting any organ system but is rarely seen in reproductive-aged women.
FIGURE 65-1 Primary syphilis. Photograph of a chancre with a raised, firm border and smooth, red base.
As noted, congenital infection is uncommon before 18 weeks. Once fetal syphilis develops, however, it manifests as a continuum of involvement. Fetal hepatic abnormalities are followed by anemia and thrombocytopenia, then ascites and hydrops (Hollier, 2001). Stillbirth remains a major complication (Di Mario, 2007; Hawkes, 2011; Rac, 2014b). The newborn may have jaundice with petechiae or purpuric skin lesions, lymphadenopathy, rhinitis, pneumonia, myocarditis, nephrosis, or long-bone involvement.
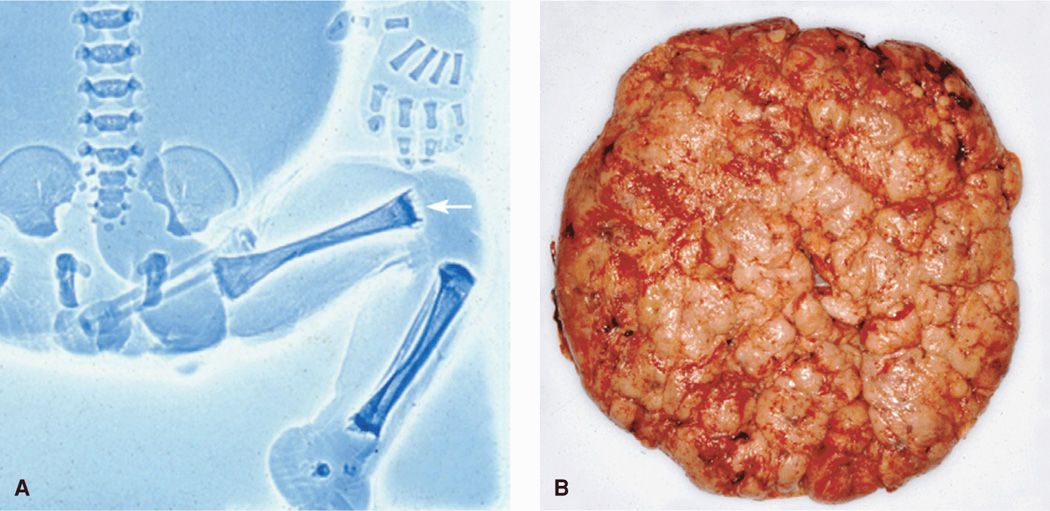
With syphilitic infection, the placenta becomes large and pale (Fig. 65-4). Microscopically, villi lose their characteristic arborization and become thicker and clubbed (Kapur, 2004). Sheffield and colleagues (2002c) described such large villi in more than 60 percent of syphilitic placentas. Blood vessels markedly diminish in number, and in advanced cases, they almost entirely disappear as a result of endarteritis and stromal cell proliferation. Likely related, Lucas and coworkers (1991) demonstrated increased vascular resistance in uterine and umbilical arteries of infected pregnancies. In a study of 25 untreated women, Schwartz and associates (1995) reported that necrotizing funisitis was present in a third. Spirochetes were detected in almost 90 percent using silver and immunofluorescent staining.
FIGURE 65-4 Congenital syphilis. A. Fetogram of a stillborn infant infected with syphilis showing the “moth-eaten” appearance of the femurs (arrow). B. Enlarged hydropic placenta of a syphilis-infected neonate.
 Diagnosis
Diagnosis
The United States Preventative Services Task Force has reaffirmed its recommendation that clinicians screen all pregnant women for syphilis to treat and prevent congenital infection (Wolff, 2009). Testing is now required by law in all states and should be performed at the first prenatal visit. In populations in which the prevalence of syphilis is high, serological testing should be performed in the third trimester and again at delivery (Centers for Disease Control and Prevention, 2010b).
Treponema pallidum cannot be cultured from clinical specimens. Definitive diagnosis of early-stage lesions is made using dark-field examination and direct immunofluorescent antibody staining of lesion exudates (Centers for Disease Control and Prevention, 2010b). In asymptomatic patients or for screening purposes, serological testing is used. There are two types. The first type is nontreponemal testing that includes the Venereal Disease Research Laboratory (VDRL) and the rapid plasma reagin (RPR). These tests are quantified and expressed as titers. Because titers reflect disease activity, they increase during early syphilis and often exceed levels of 1:32 in secondary syphilis. Following treatment of primary and secondary syphilis, serological testing at 3 to 6 months usually confirms a fourfold drop in VDRL or RPR titers (Rac, 2014a). Those with treatment failure or reinfection may lack this decline. Because VDRL titers do not correspond directly to RPR titers, consistent use of the same test for surveillance is recommended.
The other type of testing is treponemal-specific and includes the fluorescent treponemal-antibody absorption tests (FTA-ABS), the microhemagglutination assay for antibodies to T pallidum (MHA-TP), or the Treponema pallidum passive particle agglutination (TP-PA) test. These treponemal-specific tests generally remain positive throughout life.
Each of the serological tests has limitations including false-positive results. Traditionally, nontreponemal tests have been used for screening in the United States, and results are then confirmed by a specific treponemal test. Several laboratories have recently implemented a reverse screening algorithm, that is, screening with a treponemal test (Binnicker, 2012; Park, 2011). Both approaches are effective if appropriate screening, follow-up, and treatment are implemented. Patel and colleagues (2012) described missed opportunities for preventing and treating congenital syphilis in New York City. They found that 63 percent of mothers with newborns diagnosed with congenital syphilis had a missed opportunity for prevention. Most of these did not receive serological testing.
Rapid syphilis testing for “point of care” diagnosis is currently being developed and may be useful in settings with limited prenatal care (Centers for Disease Control and Prevention, 2010b; Greer, 2008). Treponemal immunochromatographic strip (ICS) tests and enzyme immunoassays (EIAs) are increasingly being used for screening and confirmatory testing (Henrich, 2011; Lukehart, 2012).
The prenatal diagnosis of congenital syphilis is difficult because an infected fetus often has a normal sonographic examination. Some findings, however, may be suggestive or even diagnostic—hydrops fetalis, ascites, hepatomegaly, placental thickening, elevated middle cerebral artery Doppler velocimetry measurements, and hydramnios all suggest infection (Rac, 2014b). Polymerase chain reaction (PCR) is specific for detection of T pallidum in amnionic fluid, and treponemal DNA has been found in 40 percent of pregnancies infected before 20 weeks (Nathan, 1997; Wendel, 1991). Fetal syphilis has also been verified by amnionic fluid dark-field examination or rabbit infectivity testing in 64 percent of a cohort of women with untreated syphilis (Hollier, 2001). Although prenatal diagnosis can be made by funipuncture or amniocentesis, its clinical utility is not clear.
 Treatment
Treatment
Syphilis therapy during pregnancy is given to eradicate maternal infection and to prevent or treat congenital syphilis. Parenteral penicillin G remains the preferred treatment for all stages of syphilis during pregnancy. Currently recommended treatment guidelines are shown in Table 65-1 and are the same as for nonpregnant adults. For pregnant women, authorities recommend that a second dose of benzathine penicillin G be given 1 week after the initial dose.
TABLE 65-1. Recommended Treatment for Pregnant Women with Syphilis

In retrospective analyses, benzathine penicillin G has been shown to be highly effective for early maternal infection. In a study of 340 pregnant women so treated, Alexander and associates (1999) reported six cases—1.8 percent—of congenital syphilis. Four of these six neonates were from a group of 75 women with secondary syphilis. The other two were identified in those delivered from a group of 102 women with early latent syphilis. Congenital syphilis was generally confined to neonates of women treated after 26 weeks and is likely related to the duration and severity of fetal infection. Sheffield and coworkers (2002b) reported that high maternal serological titers, preterm delivery, and delivery shortly after antepartum therapy are all risks for failure of maternal treatment to prevent neonatal infection. There are no proven alternatives to penicillin therapy during pregnancy. Erythromycin and azithromycin may be curative for the mother, but because of limited transplacental passage, these drugs do not prevent all congenital disease (Berman, 2004; Centers for Disease Control and Prevention, 2010b; Wendel, 1988; Zhou, 2007). In several countries, macrolide resistant strains of T pallidum are now prevalent (Stamm, 2010). Cephalosporins may prove useful, but data are limited (Augenbraun, 1999, 2002; Zhou, 2007). Tetracyclines, including doxycycline, are effective for treatment of syphilis in the nonpregnant woman. These are generally not recommended during pregnancy, however, because of the risk of yellow-brown discoloration of fetal deciduous teeth (Chap. 12, p. 248).
Penicillin Allergy
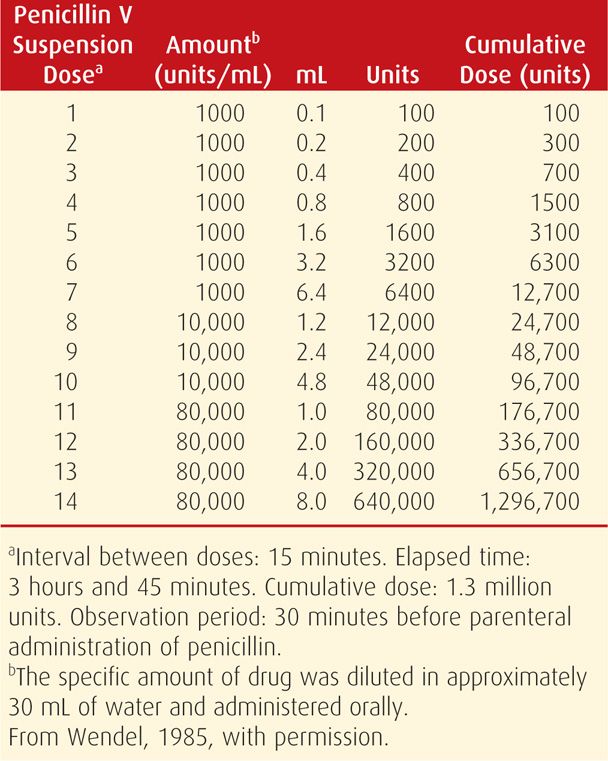
Women with a history of penicillin allergy should have either a oral stepwise penicillin dose challenge or skin testing performed to confirm the risk of immunoglobulin E (IgE)-mediated anaphylaxis. If confirmed, penicillin desensitization is recommended, as shown in Table 65-2, and then followed by benzathine penicillin G treatment (Chisholm, 1997; Wendel, 1985).
TABLE 65-2. Penicillin Allergy—Oral Desensitization Protocol for Patients with a Positive Skin Test
In most women with primary syphilis and approximately half with secondary infection, penicillin treatment causes a Jarisch-Herxheimer reaction. Uterine contractions frequently develop with this reaction and may be accompanied by decreased fetal movement and late fetal heart rate decelerations (Klein, 1990). In a study of 50 pregnant women who received benzathine penicillin for syphilis, Myles and colleagues (1998) reported a 40-percent incidence of a Jarisch-Herxheimer reaction. Of the 31 women monitored electronically, 42 percent developed regular uterine contractions with a median onset of 10 hours, and 39 percent developed variable decelerations with a median onset of 8 hours. All contractions resolved within 24 hours of therapy. Lucas and associates (1991) used Doppler velocimetry and demonstrated acutely increased vascular resistance during this time. Beta-lactam antibiotics for group B streptococcus intrapartum prophylaxis can also trigger this reaction in a woman with untreated syphilis (Rac, 2010).
All women with syphilis should be offered counseling and testing for HIV (Koumans, 2000). For women with concomitant HIV infection, the Centers for Disease Control and Prevention (2010b) recommend the same treatment as for HIV-negative persons. Clinical and serological surveillance to detect treatment failures is also recommended at 3, 6, 9, 12, and 24 months in HIV-positive patients.
GONORRHEA
Gonorrhea remains the second most commonly reported notifiable disease in the United States. The incidence of gonorrhea in the United States for 2012 was 107.5 cases per 100,000 persons, which is an increase of 4 percent since 2011 (Centers for Disease Control and Prevention, 2013b). The highest rates in women of any ethnicity were in the groups aged 20 to 24 years (578.5) and 15 to 19 years (521.2). Its prevalence in prenatal clinics in 2011 among women aged 15 to 24 years was 0.8 percent, although inner-city STI clinics have reported a prenatal prevalence of 5 to 10 percent (Berggren, 2011; Johnson, 2007). Risk factors include single marital status, adolescence, poverty, drug abuse, prostitution, other STIs, and lack of prenatal care (Berggren, 2011; Gorgos, 2011). Gonococcal infection is also a marker for concomitant chlamydial infection in up to 40 percent of infected women (Christmas, 1989; Miller, 2004). In most pregnant women, gonococcal infection is limited to the lower genital tract—the cervix, urethra, and periurethral and vestibular glands. Acute salpingitis is rare in pregnancy, but pregnant women account for a disproportionate number of disseminated gonococcal infections (Bleich, 2012; Ross, 1996; Yip, 1993).
Gonococcal infection may have deleterious effects in any trimester. There is an association between untreated gonococcal cervicitis and septic abortion as well as infection after voluntary abortion (Burkman, 1976). Preterm delivery, prematurely ruptured membranes, chorioamnionitis, and postpartum infection are reported to be more common in women infected with Neisseria gonorrhoeae (Alger, 1988; Johnson, 2011). Bleich and coworkers (2012) reviewed outcomes of 32 pregnant women admitted to Parkland Hospital for disseminated gonococcal infection. Although all the women promptly responded to appropriate antimicrobial therapy, one stillbirth was attributed to gonococcal sepsis.
 Screening and Treatment
Screening and Treatment
The U.S. Preventative Services Task Force recommends gonorrhea screening for all sexually active women, including pregnant women, if they are at increased risk (Meyers, 2008). Risk factors include age < 25 years, prior gonococcal infection, other STIs, prostitution, new or multiple sexual partners, drug use, and inconsistent condom use. For women who test positive, screening for syphilis, Chlamydia trachomatis, and HIV should precede treatment, if possible. If chlamydial testing is unavailable, presumptive therapy is given. Screening for gonorrhea in women is by culture or nucleic acid amplification tests (NAATs). Rapid tests for gonorrhea, although available, do not yet reach the sensitivity or specificity of culture or NAAT (Greer, 2008).
Gonorrhea treatment has evolved during the past decade due to the ability of N gonorrhoeae to rapidly develop antimicrobial resistance (Centers for Disease Control and Prevention, 2011b,c; Dionne-Odom, 2011; Ram, 2012; Unemo, 2011). Rapid development of fluoroquinolone resistance caused the CDC to remove that therapeutic class from its treatment guidelines in 2007. The Gonococcal Isolate Surveillance Project, a national sentinel surveillance system established to monitor trends in gonococcal antimicrobial resistance, has reported decreasing susceptibility to cephalosporins. This is the one remaining class of antimicrobial agents currently recommended for gonorrhea treatment (Bolan, 2012; Centers for Disease Control and Prevention, 2010b, 2012). This global public health threat has led the CDC in 2012 to change the gonorrhea treatment recommendations.
Listed in Table 65-3 are the updated recommendations for treatment of uncomplicated gonococcal infection during pregnancy. The increased ceftriaxone dose of 250 mg should be given along with 1 gram of azithromycin. The latter provides another drug with a different mechanism of action against N gonorrhoeae and treats chlamydial co-infections. Cefixime tablets should be reserved for situations that preclude ceftriaxone treatment. If they are used, a test-of-cure should be performed 1 week after treatment. Azithromycin, 2 grams orally as a single dose, can be used in cephalosporin-allergic women. However, this treatment should be limited due to emerging macrolide resistance (Centers for Disease Control and Prevention, 2011c). Treatment is also recommended for sexual contacts. A test-of-cure is unnecessary if symptoms resolve, but because gonococcal reinfection is common, a second screening in late pregnancy should be considered for women treated earlier (Blatt, 2012).
TABLE 65-3. Treatment of Uncomplicated Gonococcal Infections During Pregnancy
Ceftriaxone, 250 mg intramuscularly as a single dose
plus
Azithromycin, 1 gram orally as a single dose
 Disseminated Gonococcal Infections (DGI)
Disseminated Gonococcal Infections (DGI)
Gonococcal bacteremia may cause disseminated infections that manifest as petechial or pustular skin lesions, arthralgias, septic arthritis, or tenosynovitis. For treatment, the Centers for Disease Control and Prevention (2010b) has recommended ceftriaxone, 1000 mg intramuscularly or intravenously (IV) every 24 hours. Treatment should be continued for 24 to 48 hours after improvement, and therapy is then changed to an oral agent to complete 1 week of therapy. Prompt recognition and antimicrobial treatment will usually result in favorable outcomes in pregnancy (Bleich, 2012). Meningitis and endocarditis rarely complicate pregnancy, but they may be fatal (Bataskov, 1991; Burgis, 2006; Ram, 2012). For gonococcal endocarditis, ceftriaxone 1000 to 2000 mg IV every 12 hours should be continued for at least 4 weeks, and for meningitis, 10 to 14 days (Centers for Disease Control and Prevention, 2010b).
CHLAMYDIAL INFECTIONS
Chlamydia trachomatis is an obligate intracellular bacterium that has several serotypes, including those that cause lymphogranuloma venereum. The most commonly encountered strains are those that attach only to columnar or transitional cell epithelium and cause cervical infection. It is the most commonly reported infectious disease in the United States, with 1.4 million cases reported in 2012. It is estimated, however, that there are approximately 2.8 million new cases annually, although most are undiagnosed (Centers for Disease Control and Prevention, 2008). In 2012, the incidence of chlamydial infection among women was 2Δ times greater than among men. Selective prenatal screening clinics in 2011 reported a median chlamydial infection rate of 7.7 percent (Centers for Disease Control and Prevention, 2013b).
Although most pregnant women have asymptomatic infection, a third have urethral syndrome, urethritis, or Bartholin gland infection (Peipert, 2003). Mucopurulent cervicitis may be due to chlamydial or gonococcal infection or both. It may also represent normal, hormonally stimulated endocervical glands with abundant mucus production. Other chlamydial infections not usually seen in pregnancy are endometritis, salpingitis, peritonitis, reactive arthritis, and Reiter syndrome.
The role of chlamydial infection in pregnancy complications remains controversial. A few studies have reported a direct association between C trachomatis and miscarriage, whereas most show no correlation (Baud, 2011; Coste, 1991; Paukku, 1999; Sugiura-Ogasawara, 2005). It is disputed whether untreated cervical infection increases the risk of preterm delivery, preterm ruptured membranes, and perinatal mortality (Andrews, 2000, 2006; Baud, 2008; Blas, 2007; Silva, 2011). Johnson and colleagues (2011) reported a twofold risk for low-birthweight infants.
Chlamydial infection has not been associated with an increased risk of chorioamnionitis or with pelvic infection after cesarean delivery (Blanco, 1985; Gibbs, 1987). Conversely, delayed postpartum uterine infection has been described by Hoyme and associates (1986). The syndrome, which develops 2 to 3 weeks postpartum, is distinct from early postpartum metritis. It is characterized by vaginal bleeding or discharge, low-grade fever, lower abdominal pain, and uterine tenderness.
There is vertical transmission to 30 to 50 percent of neonates delivered vaginally from infected women. Perinatal transmission to newborns can cause pneumonia. Moreover, C trachomatis is the most commonly identifiable infectious cause of ophthalmia neonatorum (Chap. 32, p. 631).
 Screening and Treatment
Screening and Treatment
Prenatal screening for C trachomatis is a complex issue, although there is little evidence for its effectiveness in asymptomatic women who are not in high-risk groups (Kohl, 2003; Meyers, 2007; Peipert, 2003). Identification and treatment of asymptomatic infected women may prevent neonatal infections, but evidence of adverse pregnancy outcome prevention is lacking. Currently, the U.S. Preventive Services Task Force (2007) and the CDC recommend prenatal screening at the first prenatal visit for women at increased risk for chlamydial infection, and again during the third trimester if high-risk behavior continues. In a systematic review of repeat chlamydial infection among women, Hosenfeld and coworkers (2009) reported a reinfection rate of 14 percent, and most recurred within the first 8 to 10 months. Interestingly, in another study, Sheffield and colleagues (2005) found that almost half of pregnant women with asymptomatic cervical chlamydia underwent spontaneous resolution of infection.
Diagnosis is made predominantly by culture or NAAT. Cultures are more expensive and less accurate than newer NAATs, including PCR (Greer, 2008). Roberts and associates (2011) evaluated nucleic acid amplification testing of urine compared with cervical secretions in more than 2000 pregnant women and found them to be equivalent.
Currently recommended treatment regimens for chlamydial infections are shown in Table 65-4. Azithromycin is first-line treatment and has been found to be safe and effective in pregnancy. The fluoroquinolones and doxycycline are avoided in pregnancy, as is erythromycin estolate because of drug-related hepatotoxicity. Repeat chlamydial testing 3 to 4 weeks after completion of therapy is recommended.
TABLE 65-4. Treatment of Chlamydia trachomatis Infections During Pregnancy

 Lymphogranuloma Venereum
Lymphogranuloma Venereum
L1, L2, and L3 serovars of C trachomatis cause lymphogranuloma venereum (LGV). The primary genital infection is transient and seldom recognized. Inguinal adenitis may develop and at times lead to suppuration. It may be confused with chancroid. Ultimately, the lymphatics of the lower genital tract and perirectal tissues may be involved. Here, sclerosis and fibrosis can cause vulvar elephantiasis and severe rectal stricture. Fistula formation involving the rectum, perineum, and vulva also may evolve.
For treatment during pregnancy, erythromycin base, 500 mg orally four times daily, is given for 21 days (Centers for Disease Control and Prevention, 2010b). Although data regarding efficacy are scarce, some authorities recommend azithromycin, 1000 mg orally weekly for 3 weeks.
HERPES SIMPLEX VIRUS
Genital herpes simplex virus infection is one of the most common sexually transmitted diseases according to the Centers for Disease Control and Prevention (2010a,b). An estimated 50 million adolescents and adults are currently affected. In 2012, there were 228,000 initial office visits for genital herpes (Centers for Disease Control and Prevention, 2013b). Although most women are unaware of their infection, approximately one in six has serological evidence for HSV-2 infection. This incidence is as high as one in two for non-Hispanic black pregnant women (Corey, 2012). As most cases of HSV are transmitted by persons who are asymptomatic or unaware of their disease, herpes infections have become a major public health concern. It is estimated that 0.5 to 2 percent of pregnant women acquire HSV-1 or -2 during pregnancy (Brown, 1997).
 Pathogenesis and Transmission
Pathogenesis and Transmission
Two types of HSV have been distinguished based on immunological and clinical differences. Type 1 is responsible for most nongenital infections. However, more than half of new cases of genital herpes in adolescents and young adults are caused by HSV-1 infection. This is thought to be due to an increase in oral-genital sexual practices (Mertz, 2003; Pena, 2010; Roberts, 2003). Type 2 HSV is recovered almost exclusively from the genital tract and is usually transmitted by sexual contact. Most recurrences—greater than 90 percent—are secondary to HSV-2. There is a large amount of DNA sequence homology between the two viruses, and prior infection with one type attenuates a primary infection with the other type.
Neonatal transmission is by three routes: (1) intrauterine in 5 percent, (2) peripartum in 85 percent, or (3) postnatal in 10 percent (Kimberlin, 2004b). The fetus becomes infected by virus shed from the cervix or lower genital tract. It either invades the uterus following membrane rupture or is transmitted by contact with the fetus at delivery. The overall transmission rate is 1 in 3200 to 1 in 30,000 births depending on the population studied (Corey, 2012; Mahnert, 2007; Whitley, 2007). Neonatal herpes is caused by both HSV-1 and HSV-2, although HSV-2 infection predominates. Most infected infants are born to mothers with no reported history of HSV infection (Gardella, 2010).
The risk of neonatal infection correlates with the presence of HSV in the genital tract, the HSV type, invasive obstetrical procedures, and stage of maternal infection (Brown, 2005, 2007). Infants born to women who acquire genital HSV near the time of delivery have a 30- to 50-percent risk of infection. This is attributed to higher viral loads and the lack of transplacental protective antibodies (Brown, 1997; Brown, 2000). Women with recurrent HSV have less than a 1-percent risk of neonatal infection (Pasternak, 2010; Prober, 1987).
 Clinical Manifestations
Clinical Manifestations
Once transmitted by genital-genital or oral-genital contact, HSV-1 or -2 replicates at the entry site. Following mucocutaneous infection, the virus moves retrograde along sensory nerves. It then remains latent in cranial nerves or dorsal spinal ganglia. HSV infections may be categorized into three groups:
1. First episode primary infection describes cases in which HSV-1 or -2 is isolated from genital secretions in the absence of HSV-1 or -2 antibodies. Only a third of newly acquired HSV-2 genital infections are symptomatic (Langenberg, 1999). The typical incubation period of 6 to 8 days (range 1 to 26 days) may be followed by a “classic presentation.” This is characterized by a papular eruption with itching or tingling, which then becomes painful and vesicular. Multiple vulvar and perineal lesions may coalesce, and inguinal adenopathy may be severe (Fig. 65-5). Transient systemic influenza-like symptoms are common and are presumably caused by viremia. Hepatitis, encephalitis, or pneumonia may develop, although disseminated disease is rare. Cervical involvement is common, but it may be inapparent clinically. Some cases are severe enough to require hospitalization. In 2 to 4 weeks, all signs and symptoms of infection disappear. Many women do not present with the typical lesions—instead, a pruritic or painful abraded area or knife-slit may be present. The incidence of asymptomatic primary infections may be as high as 80 percent (Centers for Disease Control and Prevention, 2010a,b).
2. First episode nonprimary infection is diagnosed when HSV is isolated in women who have only the other serum HSV-type antibody present. For example, HSV-2 is isolated from genital secretions in women already expressing serum HSV-1 antibodies. In general, these infections are characterized by fewer lesions, fewer systemic manifestations, less pain, and briefer duration of lesions and viral shedding. This is likely because of some immunity from cross-reacting antibodies, for example, from childhood-acquired HSV-1 infection. In many cases, it may be impossible to differentiate clinically between the two types of first infection. Thus, serological confirmation may be beneficial.
3. Reactivation disease is characterized by isolation of HSV-1 or -2 from the genital tract in women with the same serotype antibodies. During the latency period, in which viral particles reside in nerve ganglia, reactivation is common and mediated through variable but poorly understood stimuli. Reactivation is termed recurrent infection and results in herpes-virus shedding. Most recurrent genital herpes is caused by type 2 virus (Centers for Disease Control and Prevention, 2010a,b; Corey, 2012). These lesions generally are fewer in number, are less tender, and shed virus for shorter periods—2 to 5 days—than those of primary infection. Typically, they recur at the same sites. Recurrences are most common in the first year after initial infection, and rates slowly decline over several years.
FIGURE 65-5 First-episode primary genital herpes simplex virus infection. On the left labium majora and mons, both vesicles and ulcers are seen.
Asymptomatic viral shedding is defined by HSV as detected by culture or PCR in the absence of clinical findings. Most infected women shed virus intermittently, and most HSV transmission to a partner occurs during periods of asymptomatic viral shedding. Gardella and coworkers (2005) reported an HSV culture-positive rate of 0.5 percent and a PCR-positive rate of 2.7 percent in asymptomatic women presenting for delivery. More data are needed to determine the effect of asymptomatic shedding on neonatal transmission.
Most primary and first-episode infections in early pregnancy are probably not associated with an increased rate of spontaneous abortion or stillbirth (Eskild, 2002). In their review, Fagnant and Monif (1989) found only 15 cases of congenital herpetic infection that were acquired during early pregnancy. Brown and Baker (1989) reported that late-pregnancy primary infection may be associated with preterm labor and fetal-growth restriction.
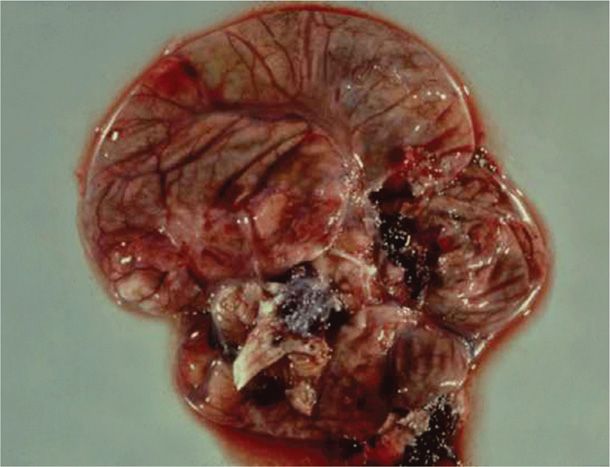
Newborn infection may manifest in a number of ways. Infection may be localized to the skin, eye, or mouth—SEM disease—in about 40 percent of cases. Central nervous system disease with encephalitis is seen in 30 percent of cases. Disseminated disease as shown in Figure 65-6 with involvement of multiple major organs is found in 32 percent. Localized infection is usually associated with a good outcome. Conversely, even with acyclovir treatment, disseminated infection has a mortality rate of nearly 30 percent (Corey, 2009; Kimberlin, 2004a,b, 2011). Importantly, serious developmental and central nervous system morbidity is seen in 20 to 50 percent of survivors with disseminated or cerebral infection.
FIGURE 65-6 Cross-section showing necrotic brain tissue from a newborn who died from disseminated herpesvirus infection.
 Diagnosis
Diagnosis
According to the Centers for Disease Control and Prevention (2010a), clinical diagnosis of genital herpes is both insensitive and nonspecific and should be confirmed by laboratory testing. HSV tests available are either virological or type-specific serological tests.
Virological tests are performed on a specimen from a mucocutaneous lesion. Cell culture and PCR are the preferred tests. The sensitivity of HSV isolation is relatively low for viral culture as vesicular lesions ulcerate and then crust. Viral isolation results sometimes are not available for 1 to 2 weeks. PCR assays are more sensitive, the results generally are available in 1 to 2 days, and specimen handling is easier. The major limitation of PCR is cost and laboratory availability. The PCR assay is the preferred test for HSV detection in spinal fluid. Regardless of the test performed, HSV viral type should be differentiated because HSV type influences counseling and long-term prognosis. A negative culture or PCR result does not exclude infection. False-positive results are rare.
Several serological assay systems are available to detect antibody to HSV glycoproteins G1 and G2 (Anzivino, 2009; Centers for Disease Control and Prevention, 2010a). These proteins evoke type-specific antibody responses to HSV-1 and HSV-2 infection, respectively, and they reliably differentiate the two. This permits confirmation of clinical infection and identification of asymptomatic carriers. The Food and Drug Administration (FDA) has approved type-specific tests that are enzyme-linked immunosorbent assay (ELISA) or blot-style tests. Providers should request type-specific glycoprotein G-based assays when serology is being performed. Sensitivity approaches 90 to 100 percent and specificity 99 to 100 percent (Wald, 2002). IgG antibodies usually are detected 1 to 2 weeks after a primary infection. IgM antibody detection is not a useful test.
Point-of-care testing is being increasingly requested by practitioners to help guide management intrapartum or near delivery. There are type-specific serological tests available that use capillary blood or serum. Sensitivity varies from 80 to 98 percent with specificity ≥ 96 percent. False-positive and false-negative results may occur. Real-time quantitative PCR of genital tract secretions has recently been developed by Gardella and colleagues (2010). They reported sensitivity of 99.6 percent and specificity of 96.7 percent with a turnaround time of 2 hours. These features would prove valuable to guide clinical decision making at delivery.
Routine serological screening for herpes in pregnancy is not recommended by the American College of Obstetricians and Gynecologists (2012) due to cost and limited data on efficacy in reducing the incidence of neonatal herpes. Cost-decision analysis for prenatal type-specific antibody screening and suppressive therapy for partners has been found to be unacceptably expensive (Barnabas, 2002; Rouse, 2000; Thung, 2005). In contrast, it was found to be potentially cost-effective by Baker and associates (2004). Cleary and coworkers (2005) calculated that universal prenatal screening would reduce rates of perinatal death and severe sequelae from neonatal HSV. But they also found that it would require treatment of 3849 women to prevent one case of neonatal death or disease with severe sequelae. Universal screening would identify previously undiagnosed women who could then be counseled regarding safe practices and antepartum antiviral suppression, but a concrete benefit remains unproven.
 Management
Management
Antiviral therapy with acyclovir, famciclovir, or valacyclovir has been used for treatment of first-episode genital herpes in nonpregnant patients. Oral or parenteral preparations attenuate clinical infection and the viral shedding duration. Suppressive therapy has also been given to limit recurrent infections and to reduce heterosexual transmission (Corey, 2004, 2012). For intense discomfort, oral analgesics and topical anesthetics may provide some relief, and urinary retention is treated with an indwelling bladder catheter. Acyclovir appears to be safe for use in pregnant women (Briggs, 2011; Stone, 2004). The manufacturers of acyclovir and valacyclovir, in cooperation with the Centers for Disease Control and Prevention, maintained a registry of outcomes following exposure to these drugs during pregnancy through 1999. More than 700 neonates exposed during the first trimester were evaluated, and no increased adverse effects were found (Stone, 2004). At this time, there are insufficient data with famciclovir exposure, although a pregnancy registry is being maintained (1-888-669-6682).
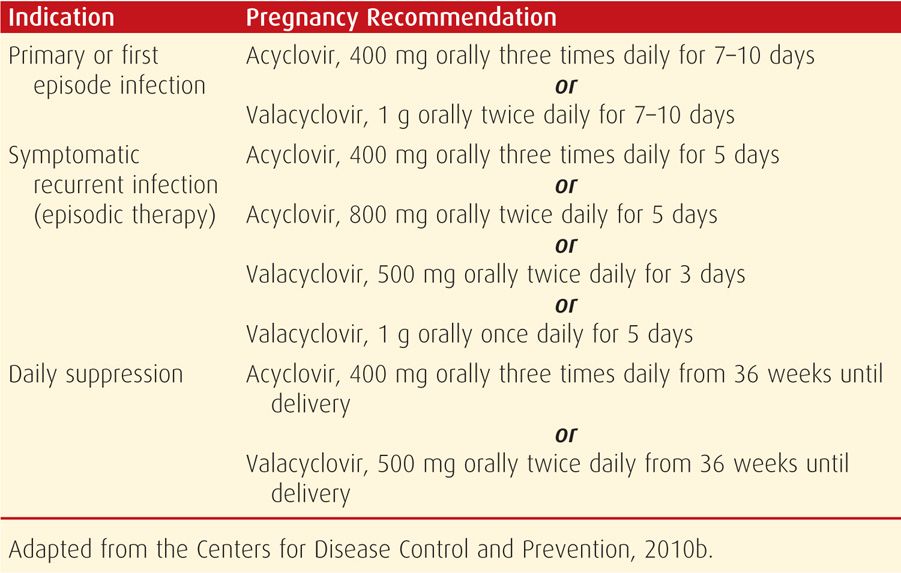
Women with a primary outbreak during pregnancy may be given antiviral therapy to attenuate and decrease the duration of symptoms and viral shedding (Table 65-5). Women with HIV co-infection may require a longer duration of treatment. Those with severe or disseminated HSV are given IV acyclovir, 5 to 10 mg/kg, every 8 hours for 2 to 7 days until clinically improved. This is followed by oral antiviral therapy to complete at least 10 days of total therapy (Centers for Disease Control and Prevention, 2010b). Recurrent HSV infections during pregnancy are treated for symptomatic relief only (see Table 65-5). Acyclovir resistance has been reported, predominantly with HSV-2 and in immunocompromised patients (Corey, 2012).
TABLE 65-5. Antiviral Medications for Herpesvirus Infection in Pregnancy
 Peripartum Shedding Prophylaxis
Peripartum Shedding Prophylaxis
Several studies have shown that acyclovir or valacyclovir suppression initiated at 36 weeks’ gestation will decrease the number of HSV outbreaks at term, thus decreasing the need for cesarean delivery (Hollier, 2008). Such suppressive therapy will also decrease viral shedding defined by both culture and PCR techniques (Scott, 2002; Sheffield, 2006; Watts, 2003). A systematic review of studies of acyclovir prophylaxis given from 36 weeks to delivery was reported by Sheffield and colleagues (2003). They found that delivery suppressive therapy was associated with significantly decreased rates of clinical HSV recurrence, cesarean deliveries for HSV recurrences, total HSV detection, and asymptomatic shedding. Subsequent studies using valacyclovir suppression have shown similar results (Andrews, 2006; Sheffield, 2006). Because of these studies, the American College of Obstetricians and Gynecologists (2012) recommends viral therapy at or beyond 36 weeks for women who have any recurrence during pregnancy. It is unclear whether suppression is needed for women with outbreaks before but not during pregnancy. There have been several case reports of atypical neonatal herpes disease following maternal antiviral suppression. Thus, suppression does not prevent all cases of neonatal disease (Pinninti, 2012).
On presentation for delivery, a woman with a history of HSV should be questioned regarding prodromal symptoms such as vulvar burning or itching. A careful examination of the vulva, vagina, and cervix should be performed, and suspicious lesions should be cultured or PCR tested. Currently, there is no approved point-of-care test to guide management. Cesarean delivery is indicated for women with active genital lesions or prodromal symptoms (American College of Obstetricians and Gynecologists, 2012). Of note, 10 to 15 percent of neonates with HSV are born to women undergoing cesarean delivery. Cesarean delivery is not recommended for women with a history of HSV infection but no active genital disease at the time of delivery. Moreover, an active lesion in a nongenital area is not an indication for cesarean delivery. Instead, an occlusive dressing is placed, and vaginal delivery is allowed.
There is no evidence that external lesions cause ascending fetal infection with preterm ruptured membranes. Major and associates (2003) described expectant management of preterm premature membrane rupture in 29 women < 31 weeks. There were no cases of neonatal HSV, and the maximum infection risk was calculated to be 10 percent. Antiviral treatment use in this setting is reasonable, but of unproven efficacy. For women with a clinical recurrence at delivery, there is not an absolute duration of membrane rupture beyond which the fetus would not benefit from cesarean delivery (American College of Obstetricians and Gynecologists, 2013). Women with active HSV may breast feed if there are no active HSV breast lesions. Strict hand washing is advised. Valacyclovir and acyclovir may be used during breast feeding, as drug concentrations in breast milk are low. One study found the acyclovir concentration to be only 2 percent of that used for therapeutic dosing of the neonate (Sheffield, 2002a).
Other antiviral agents are under development. One is pritelivir, a viral helicase-primase complex that reduces HSV-2 shedding (Wald, 2014). Also, HSV-2 vaccine development is currently being aggressively pursued. Two glycoprotein D-based subunit vaccine trials have shown benefit in discordant couples by decreasing HSV-2 acquisition (Stanberry, 2002). Interestingly, a recent randomized controlled trial of a newer glycoprotein D-based HSV-2 vaccine showed it to be effective in women in preventing HSV-1, but not HSV-2, genital disease (Belshe, 2012). Further research is needed to better understand these challenging and contradictory results (Johnson, 2011).
CHANCROID
Haemophilus ducreyi can cause painful, nonindurated genital ulcers termed soft chancres that at times are accompanied by painful suppurative inguinal lymphadenopathy. Although common in some developing countries, it had become rare in the United States by the 1970s. There was an increase in the late 1980s, but only fifteen cases were reported in the United States in 2012. This marked decrease likely represents a true decline as well as diagnostic difficulties that led to underdiagnosis (Centers for Disease Control and Prevention, 2013b). Importantly, the ulcerative lesion is a high-risk cofactor for HIV transmission.
Diagnosis by culture is difficult because appropriate media are not widely available. Instead, clinical diagnosis is made when typical painful genital ulcer(s) are dark-field negative for spirochetes and herpes-virus test results are negative. No FDA-cleared PCR test is yet available. Recommended treatment in pregnancy is azithromycin, 1 g orally as a single dose; erythromycin base, 500 mg orally three times daily for 7 days; or ceftriaxone, 250 mg in a single intramuscular dose (Centers for Disease Control and Prevention, 2010b).
HUMAN PAPILLOMAVIRUS
Human papillomavirus (HPV) is one of the most common sexually transmitted infections, and more than 40 types infect the genital tract. Most reproductive-aged women become infected within a few years of becoming sexually active, although most infections are asymptomatic and transient. Oncogenic or high-risk HPV types 16 and 18 are associated with dysplasia and are discussed in Chapter 63 (p. 1222). Mucocutaneous external genital warts termed condyloma acuminata are usually caused by HPV types 6 and 11 but may also be caused by intermediate-and high-oncogenic-risk HPV.
The National Health and Nutrition Examination Survey (NHANES) 2003 to 2006 reported an overall HPV prevalence of 43 percent in females aged 14 to 59 years (Hariri, 2011). A 27-percent seroprevalence was also reported for subjects 18 through 25 years in the National Longitudinal Study of Adolescent Health (Manhart, 2006). Similar rates of HPV positivity have been reported for pregnant women (Aydin, 2010; Gajewska, 2005; Hernandez-Giron, 2005; Rama, 2010). Prevalence is highest in younger age groups.
 External Genital Warts—Condyloma Acuminata
External Genital Warts—Condyloma Acuminata
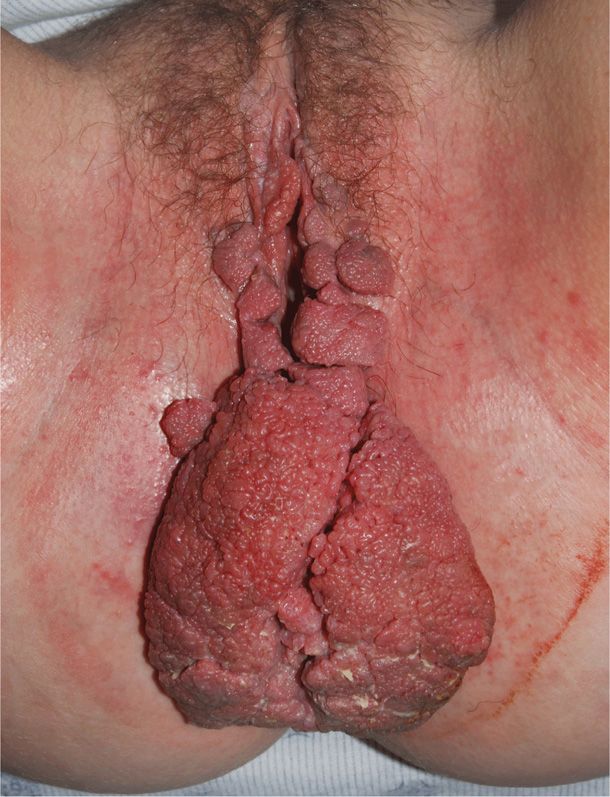
For unknown reasons, genital warts frequently increase in number and size during pregnancy. Acceleration of viral replication by the physiological changes of pregnancy might explain perineal lesion growth and progression of some to cervical neoplasm (Fife, 1999; Rando, 1989). These lesions may sometimes grow to fill the vagina or cover the perineum, thus making vaginal delivery or episiotomy difficult (Fig. 65-7). Because HPV infection may be subclinical and multifocal, most women with vulvar lesions also have cervical infection, and vice versa (Ault, 2003; Kroupis, 2011; Reichman, 2012).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree