Secondary brachial plexus surgery
Nonoperative management
Indications
Contraindications
Minimal functional deficits
Motivated patient with available motors for transfer
Stiff joints
Unavailable motors for transfer
Secondary brachial plexus surgery |
|---|
Physical/occupational therapy recommendations |
Maintain supple range of motion |
Alleviate contracture |
Maximize independence |
Participate in the decision-making process |
Elbow
Elbow flexion is motored by the biceps, brachialis, and brachioradialis muscles. Innervation is primarily from the C5 and C6 nerve roots. Elbow extension is driven by the triceps and anconeus muscles. Innervation is mainly C7, although the lower trunk can contribute to triceps’ innervation (a.k.a. lower trunk triceps) (Kozin 2007a).
Elbow Flexion
Since the most common brachial plexus injury affects C5 and C6 (a.k.a. Erb’s palsy), lack of elbow flexion is the principal problem. Failure to regain elbow flexion via spontaneous recovery of nerve reconstruction (nerve graft or nerve transfer) requires treatment. The loss of elbow flexion yields substantial impairment and difficulties with numerous activities of daily living, such as feeding and grooming.
Tendon transfer is the primary method to restore elbow flexion. There are a variety of potential donors, including the flexor-pronator mass (Steindler), triceps, latissimus dorsi, pectoralis major, and triceps muscle (Kozin 2002; Chuang et al. 1993). There are advantages and disadvantages to each of these transfers. The flexor-pronator transfer is relatively straightforward; however, this transfer provides limited and weak elbow flexion to about 90°. In addition, the patients must flex the fingers to bend the elbow (Steindler effect). This limits the usability of the transfer. The triceps to biceps transfer is effective in restoring elbow flexion, but the donor deficit is substantial. The loss of elbow extension leads to a decreased available workspace and elbow flexion contracture over time. The pectoralis major transfer can be unipolar or bipolar. The unipolar transfer detaches the insertion and transfers the tendon to the biceps using an intervening graft. This transfer if often ineffective and induces a concomitant internal rotation force at the shoulder. The bipolar transfer detaches both the origin and insertion and rotates the entire muscle into the arm. This transfer is effective, but the scarring and disfigurement are considerable. The latissimus dorsi muscle is our preferred donor for tendon transfer (Zancolli and Mitre 1973; Kozin 2002). The latissimus dorsi innervation is from the thoracodorsal nerve with a large C7 component. Therefore, the muscle is often available in C5 and C6 injuries. Furthermore, the loss of the latissimus dorsi has negligible effect on function, especially if the teres major is working.
Technique: Bipolar Latissimus Dorsi Transfer
Bipolar latissimus dorsi transfer |
|---|
Preoperative planning |
OR table: regular |
Position/positioning aids: lateral decubitus with bean bag |
Fluoroscopy location: none |
Equipment: standard |
Tourniquet: none |
Bipolar latissimus dorsi transfer is performed with the patient in the lateral decubitus position. The arm and torso are prepped and draped to the midline for adequate exposure. The ipsilateral thigh is also prepared to allow for harvesting fascia lata autograft. For allograft, an Achilles tendon or posterior tibialis tendon is procured. A long linear incision is performed along the posterior axillary line extending from the insertion of the latissimus dorsi muscle along the muscle belly toward the iliac crest. A large skin flap is elevated over the posterior aspect of the latissimus all the way to the midline. The lateral border of the muscle is identified and separated from the underlying serratus anterior muscle. The muscle with the thoracodorsal fascia is harvested from distal to proximal. The thoracodorsal neurovascular pedicle is isolated between the proximal 1/3 and distal 2/3 s on the undersurface of the latissimus. Meticulous hemostasis is attained throughout this dissection using electrocautery. The entire latissimus dorsi muscle is harvested on the thoracodorsal neurovascular pedicle (Fig. 1). Mobilization of the vascular pedicle to the subscapular artery is performed, with ligation of the arterial branch (lateral thoracic artery) to the serratus muscle.
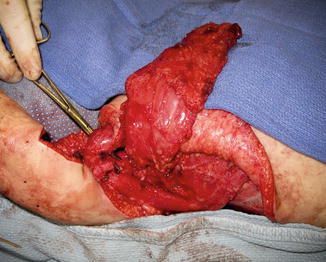

Fig. 1
An eighteen-month-old with infraclavicular brachial plexus injury, absent elbow flexion, and insufficient soft tissue along the anterior arm. The latissimus dorsi muscle harvested on the thoracodorsal neurovascular pedicle prior to transfer beneath the pectoralis major muscle (Courtesy of Shriners Hospital for Children, Philadelphia)
The tendinous insertion of the latissimus tendon into the humerus is divided. A separated anterior deltopectoral approach is then performed, and the latissimus dorsi muscle and pedicle are passed from posterior to anterior into this interval (Fig. 2). The thoracodorsal vessels must avoid torsion during passage. A large subcutaneous passage is tunneled from the deltopectoral interval to the antecubital fossa and the biceps tendon is isolated. The origin of the latissimus dorsi is attached to the biceps tendon utilizing a weave technique. Augmentation with fascia lata autograft or allograft strengthens the repair. The antecubital incision is closed before setting final tension at the anterior shoulder level. The latissimus tendon is attached to the coracoid or acromion, which should yield a 30° tenodesis effect at the elbow. Suction drains are placed into the back wound. The arm is immobilized to the chest wall for 6 weeks with the elbow at 90°. Protective extension block splinting of the elbow is continued until 3 months after surgery (Fig. 3).
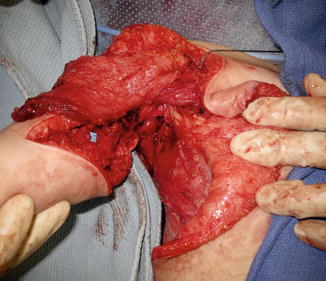


Fig. 2
The latissimus dorsi muscle passed under pectoralis major and into arm for restoration of elbow flexion (Courtesy of Shriners Hospital for Children, Philadelphia)

Fig. 3
Postoperative right elbow flexion against gravity with well-healed posterior axillary incision (Courtesy of Shriners Hospital for Children, Philadelphia)
Bipolar latissimus dorsi transfer |
|---|
Surgical steps |
Long linear incision is performed along the posterior axillary line |
Identify lateral border of the latissimus |
Separate from the underlying serratus anterior muscle |
Isolate thoracodorsal neurovascular pedicle |
Mobilization of the vascular pedicle to the subscapular artery and ligate lateral thoracic artery |
Perform deltopectoral approach and antecubital approach |
Isolate the coracoid, acromion, and biceps tendon |
Create large subcutaneous tunnel between deltopectoral and antecubital incisions |
Release origin and insertion of the latissimus muscle |
Pass the muscle from back to front without kinking the neurovascular pedicle |
Attach origin of the latissimus dorsi to the biceps tendon. Augment with fascia lata autograft or allograft |
Attach the latissimus tendon to the coracoid or acromion |
Bipolar latissimus dorsi transfer |
|---|
Postoperative protocol |
Suction drains are placed into the back wound |
Arm immobilized to the chest wall for 6 weeks with the elbow at 90° |
Tendon transfer firing and reeducation instituted |
Protective extension block splinting of the elbow is continued until 3 months after surgery |
Bipolar latissimus dorsi transfer | |
|---|---|
Potential pitfalls and preventions | |
Potential pitfall | Pearls for prevention |
Pitfall #1 Difficultly isolating the neurovascular pedicle | Separate the serratus anterior from the latissimus dorsi muscle |
Pitfall #2 Inadequate pedicle length | Dissect proximal to the subscapular artery; ligate the lateral thoracic artery |
Pitfall #3 Suboptimum attachment to the biceps tendon | Augmentation with autograft or allograft |
Pitfall #4 Postoperative seroma in the back | Avoid removing drains too early; wait until patient is ambulating |
Pitfall #5 Difficulty with firing transfer | Enlist an experienced therapist; try alternative methods such as biofeedback |
Free-Tissue Transfer
Free-tissue transfer is considered when there are no available local donors (Chuang et al. 1993, 1996; Manktelow 1986). The primarily transferrable muscle is the gracilis based upon the obturator neurovascular supply (Manktelow 1986). The motor nerve to empower the free-muscle transfer is either the spinal accessory or intercostal nerves. This surgical procedure requires adequate training and skill in microvascular techniques.
Technique: Free Gracilis Muscle Transfer
Free gracilis muscle transfer |
|---|
Preoperative planning |
OR table: regular |
Position/positioning aids: standard, prep the contralateral donor thigh and leg along with the recipient arm and hemithorax |
Fluoroscopy location: none |
Equipment: intraoperative and postoperative Doppler (Cook-Swartz Doppler), nerve stimulator (checkpoint stimulator), microscope, microsurgical instrumentation, fibrin glue |
Tourniquet: none |
The surgery can be performed with one or two surgical teams. The gracilis muscle is harvested from the contralateral thigh as the pedicle will orient better than an ipsilateral harvest (Fig. 4). The gracilis is isolated posterior to the adductor magnus and longus muscles via a long medial incision from the pubis to the distal aspect of the thigh. The obturator neurovascular pedicle is isolated in the proximal 1/3 of the muscle and carefully traced in a proximal direction to increase the length of the pedicle (Fig. 5). The dominant arterial supply is provided by a branch from the profunda femoris. There are consistently two veins, a single artery, and a single nerve. The tendinous origin is isolated proximal to the neurovascular pedicle. The distal dissection is straightforward and the gracilis is isolated as distal as possible. The tendinous insertion is released from the pes anserinus. Frequently, a skin paddle is harvested with the muscle to enhance donor site coverage and facilitate flap monitoring after surgery.
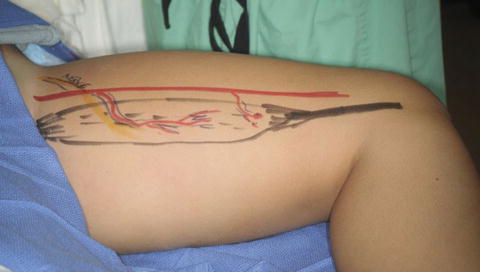
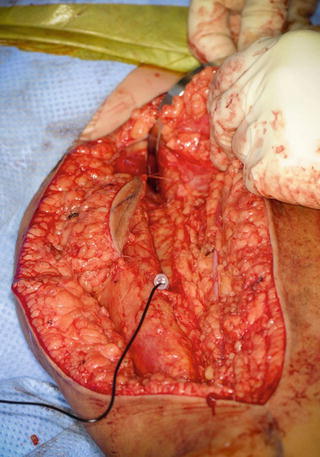

Fig. 4
An eight-year-old child with brachial plexus birth palsy status post exploration and grafting as an infant with persistent lack of right elbow flexion. Left gracilis harvest incision diagrammed (Courtesy of Shriners Hospital for Children, Philadelphia)

Fig. 5
Left gracilis muscle isolated and pedicle extended in a proximal direction (Courtesy of Shriners Hospital for Children, Philadelphia)
The upper limb is prepared by isolating the nerve(s) to power the gracilis muscle and finding an appropriate artery for inflow and veins for outflow. The spinal accessory nerve is isolated via a supraclavicular brachial plexus approach. The most facile technique requires isolation of the trapezius muscle at its attachment into the acromion. Dissection proceeds across the muscle better in a slow and deliberate fashion. The spinal accessory nerve and its adjacent artery are encountered coursing along the trapezius muscle. Electrical stimulation with a biphasic stimulator (Checkpoint device, Checkpoint Surgical LLC, Cleveland, Ohio, USA) is helpful. There are branches of the cervical plexus in the vicinity that will not stimulate muscle contraction and should not be confused with the spinal accessory nerve.
The intercostal nerves are harvested via an inframammary incision that extends from the costochondral junction to the posterior axillary fold (Kozin 2007a; Krakauer and Wood 1994; Malessy and Thomeer 1998). The skin along with the pectoralis major and pectoralis minor is elevated in a cephalad direction to expose the underlying ribs. Three intercostal nerves are preferred to power the gracilis muscle (Fig. 6). The nerves are harvested by reflecting the intercostal muscle in an inferior direction. A two-pronged hook is carefully placed on the rib and retracted in cephalad direction. The underlying intercostal nerve is isolated and a vessel loop placed around the nerve. The nerve is then painstakingly dissected from costochondral junction to the posterior axillary fold. Meticulous technique is necessary to avoid entering the lung pleura. This process is repeated three times.
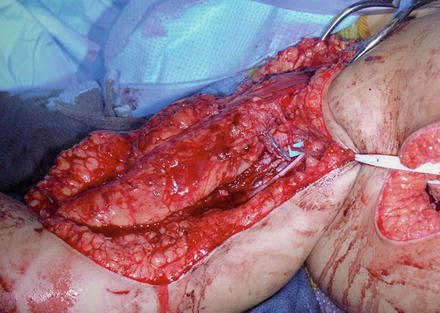

Fig. 6
Gracilis muscle innervated via intercostal nerve transfers (blue background) (Courtesy of Shriners Hospital for Children, Philadelphia)
The donor artery and veins must also be identified and prepared. The choice varies with the size of the patient, the diameter of the vessels, the presence or absence of previous surgery, and the preference of the surgeon. A frequent selection is end to side anastomosis to the brachial artery and end to end anastomosis to cephalic vein branches (Fig. 7). Microsurgical expertise is a perquisite, especially in children with small caliper vessels.
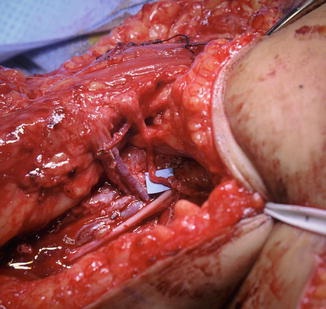

Fig. 7
Gracilis muscle anastomosis via end to side to brachial artery and vein (Courtesy of Shriners Hospital for Children, Philadelphia)
The arm is further prepared by isolation of the attachment site for the gracilis origin and insertion. The origin is either the clavicle or the acromion. The insertion site is usually any remaining biceps tendon or directly into the coronoid if the biceps is completely absent.
Once the donor and recipient sites are prepared, the gracilis pedicle is ligated and the muscle transported into the arm. The origin is secured and the tendon passed to the elbow to provide some stability to muscle. Full attention is then directed to microsurgical arterial and venous anastomosis. The arterial repair is completed first to minimize the ischemia time. Subsequently, the two venous anastomosis repairs are completed. After successful anastomoses, attention is directed to nerve coaptation using suture and/or fibrin glue (Tisseel, Baxter Pharmaceuticals, Deerfield, Illinois, USA). Doppler cuffs (Cook Catheters, Cook Medical Inc., Bloomington, Indiana, USA) are placed around the artery and a vein for postoperative monitoring. Lastly, the gracilis tendon is woven into the biceps tendon or secured through a drill hole in the coronoid. Tension is set such that there is no tension across the neurovascular repair and the elbow rests in about 30° of flexion.
The wounds are all closed with suction drains placed into the thigh. The arm is immobilized with the arm at the side and elbow in 90° of flexion. The patient is transferred to the intensive care unit for flap monitoring via the skin paddle and Doppler catheters. Routine anticoagulation other than a daily aspirin is not recommended. The Doppler cuffs are removed 5 days after surgery. The wires can be cut rather than pulled, which may result in vascular disruption.
The arm is immobilized for 6 weeks and gentle passive range of motion started. The terminal 30° of elbow extension is not allowed until signs of reinnervation and recruitment become evident. Appropriate therapy is mandatory to achieve a successful outcome.
Free gracilis muscle transfer |
|---|
Surgical steps |
Long linear incision along the medial aspect of the donor thigh |
Identify the gracilis from the pubis to the distal aspect of the thigh |
Isolate the obturator neurovascular pedicle in the proximal 1/3 of the muscle |
Trace neurovascular pedicle in proximal direction to increase the length of the pedicle |
Release the tendinous insertion from the pes anserinus |
Prepare the recipient upper limb |
Select and isolate the donor nerve(s), artery, and veins. In a similar fashion, isolate the eventual origin and insertion sites |
Ligate the obturator neurovascular pedicle and transport the muscle into the arm |
Stabilize the gracilis muscle in the arm by passing the tendon to the elbow |
Complete arterial repair to minimize the ischemia time |
Complete two venous anastomoses to allow for venous egress |
Coapt obturator nerve to donor nerve(s) |
Secure gracilis origin to clavicle or coracoid and complete distal weave to the biceps tendon |
Apply Doppler cuffs and close |
Free gracilis muscle transfer |
|---|
Postoperative protocol |
Suction drains are placed into the thigh wound |
Arm immobilized to the chest wall for 6 weeks with the elbow at 90° |
Protective extension block splinting of the elbow is continued until reinnervation |
Tendon transfer firing and reeducation instituted once reinnervation is evident. If the intercostal nerves are utilized, coughing often activates the muscle |
Free gracilis muscle transfer | |
|---|---|
Potential pitfalls and preventions | |
Potential pitfall | Pearls for prevention |
Pitfall #1 Neurovascular pedicle too short | Trace pedicle in proximal direction to ensure adequate length |
Pitfall #2 Difficulty with anastomosis | Critical step in “setup.” Make sure the recipient vessels are adequately prepared |
Pitfall #3 Failure of anastomosis | Be prepared to try again and have emergency OR team available |
Pitfall #4 Donor difficulty – identification of spinal accessory | Resides directly on top of the trapezius muscle; stimulator is helpful |
Pitfall #5 Donor difficulty – intercostal nerve harvest can result in pneumothorax | Small rents in the parietal pleura do not require chest tube placement; large rents require chest tube |
Double Free-Tissue Transfer
In extremely desperate cases with flail limbs, double free-muscle transfers are a valiant attempt to restore marginal function (Doi et al. 2000). The “double Doi” is performed in two stages. In the first operation, finger extension and elbow flexion are restored by a single free-muscle transfer innervated by the spinal accessory nerve. Finger flexion is restored by a second free muscle innervated by the fifth and sixth intercostal nerves. The third and fourth intercostal nerves are coapted to the motor branch of the triceps muscle to restore elbow extension and to stabilize the elbow against the flexion moment generated by the first muscle transfer.
Elbow Extension
Deficient elbow extension removes an antagonist to elbow flexion and inhibits certain activities of daily living. The lack of an antagonist to elbow flexion results in an elbow contracture due to the unimpeded forces in the elbow flexors. Certain activities of daily living can be accomplished by gravity-assisted elbow extension. In contrast, elbow extension against gravity is required for overhead activities and pushing up from a chair. In patients that are upper extremity weight bearers (e.g., paraplegia or crutch walkers), the loss of strong active elbow extension is especially problematic. There are a limited number of donors for elbow extension. Options include the posterior deltoid, latissimus dorsi, and biceps (Kozin 2007b). Free-muscle transfer is another possibility. The choice of donor is confounded by trying to avoid those muscles that have been reinnervated. Reinnervated muscle is often weaker and loses some of its physiologic properties (excursion and cross-sectional area). In addition, mass reinnervation can result in the inability to selectively fire an individual muscle. A careful physical examination is necessary to ensure independent firing. On occasion, dynamic electromyography can facilitate the decision-making process (Hutchinson et al. 2008).
The two main donors are the biceps and the posterior deltoid muscles (Mulcahey et al. 2003; LeClerq et al. 2008). Each procedure has its own nuances. The biceps to triceps is easier and has a less rigid postoperative rehabilitation. The posterior deltoid transfer requires an intervening graft that complicates the technique and adds greater postoperative restrictions.
Technique: Biceps to Triceps Transfer
Operative Prerequisites
Active brachialis and supinator muscles are requirements to biceps transfer to maintain elbow flexion and forearm supination (Kozin 2003; Kuz et al. 1999). The evaluation of their presence requires a vigilant physical examination of elbow flexion and forearm supination strength. The brachialis and supinator muscles can be assessed independent of the biceps muscle. Effortless forearm supination without resistance will cause supinator function that is palpable along the proximal radius. Likewise, powerless elbow flexion causes palpable brachialis contraction along the anterior humerus deep to the biceps muscle. Equivocal cases require supplementary evaluation to guarantee adequate supinator and brachialis muscle activity. Injection of the biceps muscle with a local anesthetic (e.g., bupivacaine) induces temporary paralysis. Subsequently, the examiner can assess independent assessment of brachialis and supinator function via elbow flexion and forearm supination, respectively.
A supple elbow with near complete range of motion is also necessary. Patients with an elbow contracture require resolution of the contracture prior to surgery. Therapy and/or serial casting can lessen the contracture. Surgery is deferred until the contracture is less than 20°. A greater contracture negates the biceps tendon from reaching the olecranon at surgery.
Technique: Biceps to Triceps Transfer
Biceps to triceps transfer |
|---|
Preoperative planning |
OR table: regular |
Position/positioning aids: supine, meticulous padding of all bony prominences |
Fluoroscopy location: none |
Equipment: standard |
Tourniquet: sterile |
The patient is placed supine on the operating room table. The entire extremity is prepped and draped. A sterile circular tourniquet is applied (HemaClear, OHK Medical Devices, Grandville, Michigan, USA) that exsanguinates during application. An S-shaped incision is made along the medial arm, across the antecubital fossa, and over the brachioradialis muscle belly. Skin flaps are elevated and the biceps tendon is traced to its insertion into the radial tuberosity. There are large crossing veins that require ligation. The lacertus fibrosus can be incorporated into the transfer or left behind (Kozin 2003; Kuz et al. 1999; Revol et al. 1999; Kozin et al. 2010). The tendon is released directly from the radial tuberosity to maximize length. The tendon and muscle belly are mobilized proximal into the arm to maximize excursion and to improve line of pull (Fig. 8). The musculocutaneous and lateral antebrachial nerves must be protected.
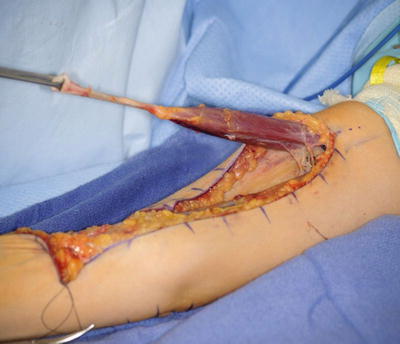

Fig. 8
An eighteen-year-old female with lack of elbow flexion necessitating biceps to triceps transfer. Biceps harvested from radial tuberosity and proximal dissection performed to improve excursion (Courtesy of Shriners Hospital for Children, Philadelphia)
Only the medial side of the arm, the median nerve, the brachial artery, and the ulnar nerve are isolated. A posterior incision is made around the olecranon and extended in a proximal direction along the triceps tendon. A subcutaneous tunnel is created between the medial brachium and posterior incision for tendon passage (Fig. 9). The tendon can be passed over the median and ulnar nerves; however, the advance in neuroregeneration has changed our practice. The tendon is passed over the median nerve, but under the ulnar nerve to avoid any iatrogenic ulnar nerve compression (Fig. 10). A Krackow suture is placed in the tendon with the two suture ends left long.
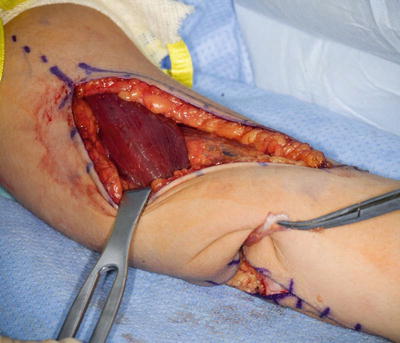
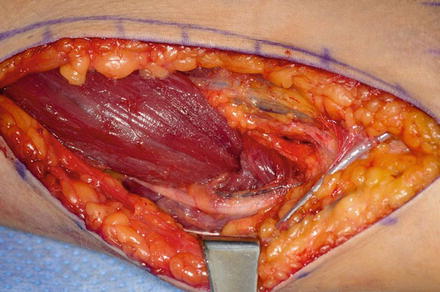

Fig. 9
Biceps passed along the medial side of the arm to the posterior incision beneath the ulnar nerve (Courtesy of Shriners Hospital for Children, Philadelphia)

Fig. 10
Biceps passed beneath the ulnar nerve (Courtesy of Shriners Hospital for Children, Philadelphia)
The triceps tendon is split and the tip of the olecranon is exposed. A large bore blind bone tunnel is made in the olecranon for the acceptance of the biceps tendon. The tendon is passed through the medial leaflet of the triceps tendon in preparation of docking into the bone tunnel (Fig. 11). The tendon is docked using two posterior unicortical holes and suture retrievers (Suture retriever, Smith Nephew, Andover, Massachusetts, USA) (Kozin and Zlotolow 2012) (Fig. 12). The elbow is placed in full extension and the two ends are drawn into the bone tunnel using suture retrievers (Fig. 13). The sutures are tied over the posterior olecranon cortex. Additional suturing is performed during the closure of the triceps split with incorporation of the biceps tendon. Following closure, a well-padded long-arm cast is applied with the elbow in extension. The wrist is included within the cast and the hand position depends upon concomitant procedures performed for hand function.
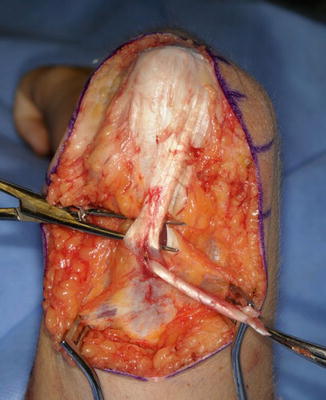
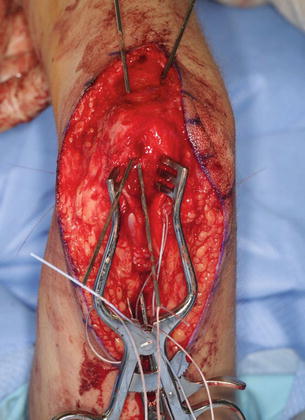
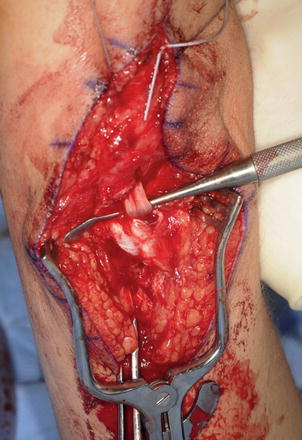

Fig. 11
Biceps tendon passed though triceps tendon prior to docking into olecranon (Courtesy of Shriners Hospital for Children, Philadelphia)

Fig. 12
Suture retrievers to facilitate docking biceps tendon into osseous tunnel (Courtesy of Shriners Hospital for Children, Philadelphia)

Fig. 13
Biceps tendon docked into olecranon (Courtesy of Shriners Hospital for Children, Philadelphia)
Biceps to triceps transfer |
|---|
Surgical steps |
S-shaped incision is made along the medial arm, across the antecubital fossa, and over the brachioradialis muscle belly |
The biceps tendon traced to insertion into the radial tuberosity, released, and mobilized in a proximal direction |
Posterior incision around the olecranon and extended in a proximal direction, the triceps tendon is split and the tip of the olecranon is exposed |
The biceps tendon passed from anterior to posterior via spacious tunnel that runs over median nerve and deep to ulnar nerve |
The biceps tendon passes through triceps leaflet and is secured within osseous tunnel using a docking technique |
Biceps to triceps transfer |
|---|
Postoperative protocol |
Elbow extension cast for 1 week |
Gradually mobilize the elbow and initiate transfer training at 15 degrees per week, using a dial-hinge brace, Bledsoe brace (Bledsoe Brace Systems, Grand Prairie, Texas, USA) |
Tendon transfer firing and reeducation instituted |
Protective splinting of the elbow is continued until 3 months after surgery |
Biceps to triceps transfer | |
|---|---|
Potential pitfalls and preventions | |
Potential pitfall | Pearls for prevention |
Pitfall #1 Bleeding during biceps harvest | Large crossing veins require careful ligation during dissection |
Pitfall #2 Inadequate biceps length to reach olecranon | Resolve any elbow contracture prior to surgery |
Pitfall #3 Difficulty firing transfer after surgery | Try alternative methods, such as biofeedback |
Pitfall #4 Difficulty regaining elbow flexion | Be patient; avoid passive elbow flexion until 3 months following surgery |
Technique: Posterior Deltoid Transfer
Operative Prerequisites
The posterior deltoid must have adequate strength to motor elbow extension. Shoulder extension and palpation best assess posterior deltoid strength and turgor. The examination can be performed with the patient seated, although placing the patient prone provides a more reliable assessment. Similar to biceps to triceps transfer, a supple elbow with near complete range of motion is also necessary. Patients with an elbow contracture require resolution of the contracture prior to surgery.
Posterior deltoid transfer
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|
|---|