Risk Factors
Selected risk factors for NTDs are listed in Table 14-1, and genetic factors represent the largest category. Isolated NTDs display multifactorial inheritance. The recurrence risk is approximately 3 to 5 percent if a couple has previously had a child with either anencephaly or spina bifida, 5 percent if either parent was born with an NTD, and as high as 10 percent if a couple has two affected children. Importantly, almost 95 percent of NTDs develop in the absence of a family history. Polymorphisms in the methylene tetrahydrofolate reductase gene, which leads to impaired homocysteine and folate metabolism, have been associated with increased risk for anencephaly and spina bifida, as well as for cardiac malformations (Aneji, 2012; Harisha, 2010; Munoz, 2007; Yin, 2012). NTDs are a component of more than 80 genetic syndromes, many including other anomalies that may be amenable to prenatal diagnosis (Milunsky, 2004).
TABLE 14-1. Risk Factors for Neural-Tube Defects
Genetic Cause
Family history—multifactorial inheritance
MTHFR mutation—677C→T
Syndromes with autosomal recessive inheritance—Meckel Gruber, Roberts, Joubert, Jarcho-Levin, HARDE (hydrocephalus-agyria-retinal dysplasia-encephalocele)
Aneuploidy—trisomy 13 and 18, triploidy
Environmental Exposures
Diabetes—hyperglycemia
Hyperthermia—hot tub or sauna, fever (controversial)
Medications—valproic acid, carbamazepine, coumadin, thalidomide, efavirenz
Geographical—Ethnicity, Diet, and Other Factors
United Kingdom, India, China, Egypt, Mexico, Southern Appalachian United States
Other risk factors for NTDs include hyperthermia, medications that disturb folic acid metabolism, and hyperglycemia from insulin-dependent diabetes. Although the exact mechanism by which diabetes causes these anomalies is unknown, rodent studies have found that oxidative stress from embryonic hyperglycemia is associated with apoptosis in the developing neural tube (Li, 2012; Sugimura, 2009; Yang, 2008). The risk for these defects is also increased in certain racial or ethnic groups as well as in populations from selected geographical regions. For example, recent data from population-based registries indicate an NTD prevalence of 1.0 to 1.3 per 1000 births in the United Kingdom, which compares with 0.9 per 1000 in the United States (Cragan, 2009; Dolk, 2010). In the United States, the risk may be twice as high among Mexican-born women (Velie, 2006).
Prevention
Most women at increased risk for NTDs benefit from 4 mg folic acid taken daily before conception and through the first trimester. This is particularly important if a woman has one or more prior affected children or if either the pregnant woman or her partner has such a defect. Folic acid supplementation may not decrease the risk for NTDs in those with valproic acid exposure, pregestational diabetes, first-trimester fever or hot tub exposure, or defects associated with a genetic syndrome (American College of Obstetricians and Gynecologists, 2013b).
The policy of routine fortification of cereal grains with folic acid, which has been in place in the United States since 1998, provides approximately 200 additional micrograms of folic acid daily and may reduce the first occurrence of NTDs in low-risk women by approximately 20 percent (Honein, 2001). It is recommended that all women at low risk take 400 μg of folic acid orally every day before conception and through the first trimester, to reduce the NTD risk by as much as 80 percent (Chap. 8, p. 159).
 Maternal Serum Alpha-Fetoprotein Screening (MSAFP)
Maternal Serum Alpha-Fetoprotein Screening (MSAFP)
Alpha-fetoprotein (AFP) is a glycoprotein synthesized by the fetal yolk sac and later by the fetal gastrointestinal tract and liver. It is the major serum protein in the embryo and fetus and is thus analogous to albumin. As shown in Figure 14-1, its concentration increases steadily in both fetal serum and amnionic fluid until 13 weeks’ gestation, after which, levels rapidly decline. Conversely, AFP is found in steadily increasing quantities in maternal serum after 12 weeks. The normal concentration gradient between fetal plasma and maternal serum is on the order of 50,000:1. Defects in fetal integument, such as neural-tube and ventral wall defects, permit AFP to leak into the amnionic fluid, resulting in dramatically increased maternal serum AFP levels.
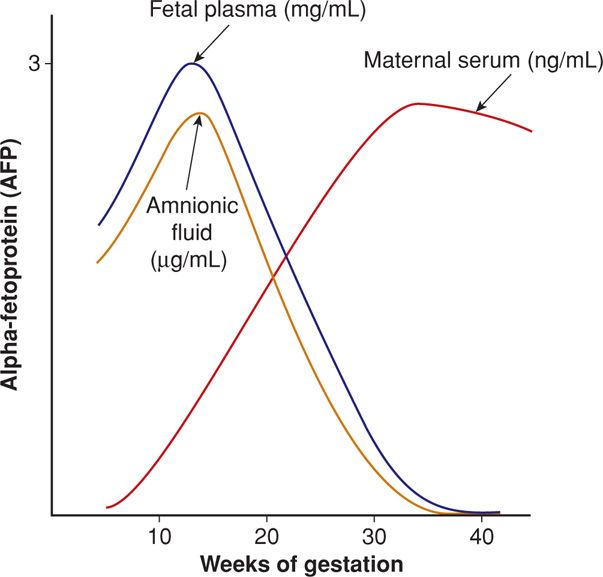
FIGURE 14-1 Diagram of alpha-fetoprotein (AFP) concentration across gestational age in fetal plasma, amnionic fluid, and maternal serum. The scale refers to the fetal plasma level, which is approximately 150 times greater than the amnionic fluid concentration and 50,000 times greater than the maternal serum concentration.
It was shown more than 30 years ago that maternal serum AFP concentrations at 16 to 18 weeks exceeded 2.5 multiples of the median (MoM) in a large proportion of women carrying fetuses with either anencephaly or spina bifida (Wald, 1977). Since the mid-1980s, MSAFP concentration has been routinely measured as a screening test for NTDs.
Maternal serum AFP screening is generally performed from 15 through 20 weeks, within a protocol that includes quality control, counseling, and follow-up. AFP is measured in nanograms per milliliter and reported as multiples of the median (MoM) of the unaffected population. Using MoM normalizes the distribution of AFP levels and permits comparison of results from different laboratories and populations. Using a maternal serum AFP level of 2.0 or 2.5 MoM as the upper limit of normal, most laboratories report a detection rate—test sensitivity—of at least 90 percent for anencephaly and 80 percent for spina bifida at a screen-positive rate of 3 to 5 percent (Milunsky, 2004). The positive predictive value—the proportion with AFP elevation that have an affected fetus—is only 2 to 6 percent. This is explained by the overlap in AFP distributions in affected and unaffected pregnancies, as shown in Figure 14-2.
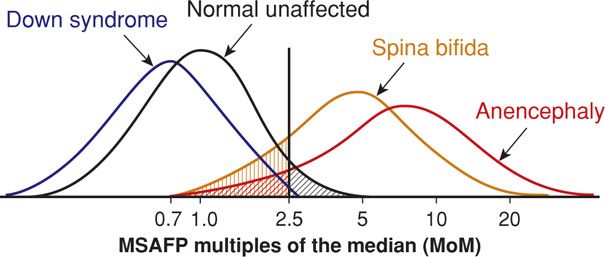
FIGURE 14-2 Maternal serum alpha-fetoprotein distribution for singleton pregnancies at 15 to 20 weeks. The screen cut-off value of 2.5 multiples of the median is expected to result in a false-positive rate of up to 5 percent (black hatched area) and false-negative rates of up to 20 percent for spina bifida (tan hatched area) and 10 percent for anencephaly (red hatched area).
Several factors influence maternal serum AFP levels and are considered when calculating the AFP MoM:
1. Maternal weight—The AFP concentration is adjusted for the maternal volume of distribution.
2. Gestational age—The maternal serum concentration increases by approximately 15 percent per week during the second trimester (Knight, 1992). In general, the MoM should be recalculated if the biparietal diameter differs from the stated gestational age by more than 1 week.
3. Race/ethnicity—African American women have at least 10-percent higher serum AFP concentrations but are at lower risk for fetal NTDs.
4. Diabetes—Serum levels may be 10 to 20 percent lower in women with insulin-treated diabetes, despite a three- to fourfold increased risk for NTDs (Greene, 1988; Huttly, 2004). There is controversy whether such adjustment remains necessary or if results should apply to all types of diabetes (Evans, 2002; Sancken, 2001; Thornburg, 2008).
5. Multifetal gestation—Higher screening threshold values are used in twin pregnancies (Cuckle, 1990). At Parkland Hospital, an AFP level is considered elevated in a twin pregnancy if greater than 3.5 MoM, but other laboratories use 4.0 or even 5.0 MoM.
According to the American College of Obstetricians and Gynecologists (2013b), all pregnant women should be offered screening for NTDs. Women who present for care early in pregnancy often have the option of several different screening tests for aneuploidy, as discussed subsequently. Those who elect second-trimester multiple marker serum screening will have a maternal serum AFP level measured as a component. Those who elect first-trimester screening or chorionic villus sampling may receive neural-tube defect screening either with serum AFP at 15 to 20 weeks or with sonography (American College of Obstetricians and Gynecologists, 2013c).
 MSAFP Elevation
MSAFP Elevation
One algorithm for evaluating elevated maternal serum AFP levels is shown in Figure 14-3. The evaluation begins with a standard sonogram, if not already performed earlier in gestation, as this can reliably exclude three common causes of AFP level elevation: underestimation of gestational age, multifetal gestation, and fetal demise. Virtually all cases of anencephaly and many cases of spina bifida may be detected or suspected during a standard second-trimester sonographic examination (Dashe, 2006). Once the gestational age is verified and the screening test is confirmed to be abnormal, a patient is offered diagnostic evaluation.
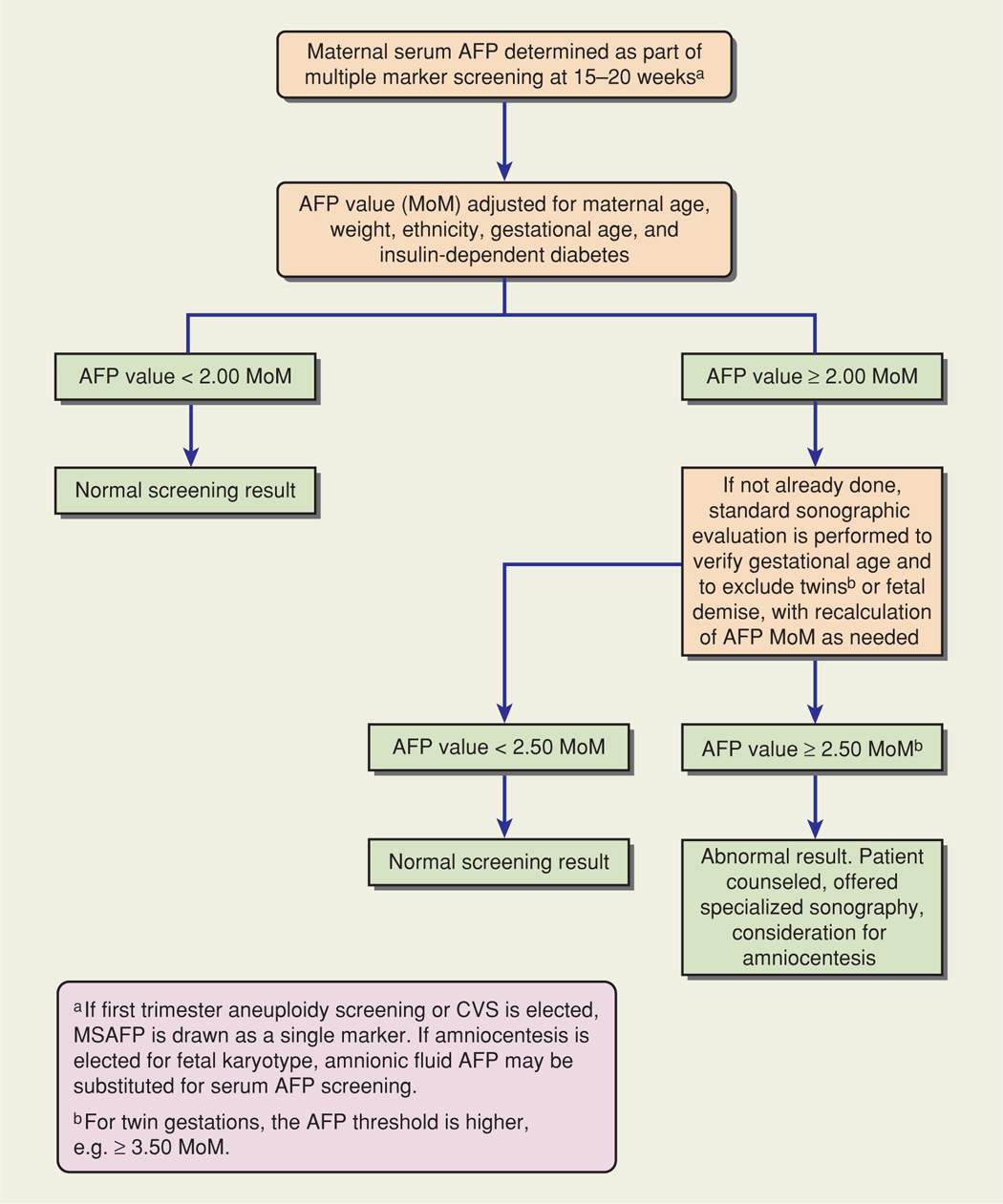
FIGURE 14-3 Example of an algorithm for evaluating maternal serum alpha-fetoprotein screening values (MSAFP). CVS = chorionic villus sampling; MoM = multiples of the median.
Numerous fetal and placental abnormalities have been associated with AFP elevation (Table 14-2). The likelihood of one of these abnormalities or of an adverse pregnancy outcome in the absence of a recognized abnormality increases in proportion to the AFP level. More than 40 percent of pregnancies may be abnormal if the AFP level is greater than 7 MoM (Reichler, 1994).
TABLE 14-2. Conditions Associated with Abnormal Maternal Serum Alpha-Fetoprotein Concentrations
Elevated Levels
Underestimated gestational age
Multifetal gestationa
Fetal death
Neural-tube defect
Gastroschisis
Omphalocele
Cystic hygroma
Esophageal or intestinal obstruction
Liver necrosis
Renal anomalies—polycystic kidneys, renal agenesis, congenital nephrosis, urinary tract obstruction
Cloacal exstrophy
Osteogenesis imperfecta
Sacrococcygeal teratoma
Congenital skin abnormality
Pilonidal cyst
Chorioangioma of placenta
Placenta intervillous thrombosis
Placental abruption
Oligohydramnios
Preeclampsia
Fetal-growth restriction
Maternal hepatoma or teratoma
Low Levels
Obesitya
Diabetes mellitusa
Trisomies 21 or 18
Gestational trophoblastic disease
Fetal death
Overestimated gestational age
For the foregoing reasons, women with a confirmed serum AFP level elevation should be referred for additional counseling and offered a diagnostic test, either specialized sonography or amniocentesis. Some women have risk factors that warrant referral for a diagnostic test even in the setting of a normal AFP level. These include personal history of or first-degree relative with an NTD, insulin-treated diabetes, and first-trimester exposure to a medication associated with increased risk.
Targeted Sonography
More than 25 years ago, Nicolaides and colleagues (1986) described frontal bone scalloping—the lemon sign, and anterior curvature of the cerebellum with effacement of the cisterna magna—the banana sign—in second-trimester fetuses with open spina bifida (Fig. 14-4). These investigators also frequently noted a small biparietal diameter and ventriculomegaly in such cases. Watson and coworkers (1991) reported that 99 percent of fetuses with open spina bifida had one or more of these findings. In addition to these cranial findings, transverse and sagittal images of the spine are increasingly used to characterize the size and location of spinal defects (Chap. 10, p. 202). Using these findings, experienced investigators have described nearly 100-percent detection of open NTDs (Norem, 2005; Sepulveda, 1995). Overall NTD risk may be reduced by at least 95 percent when no spine or cranial abnormality is observed (Morrow, 1991; Van den Hof, 1990).
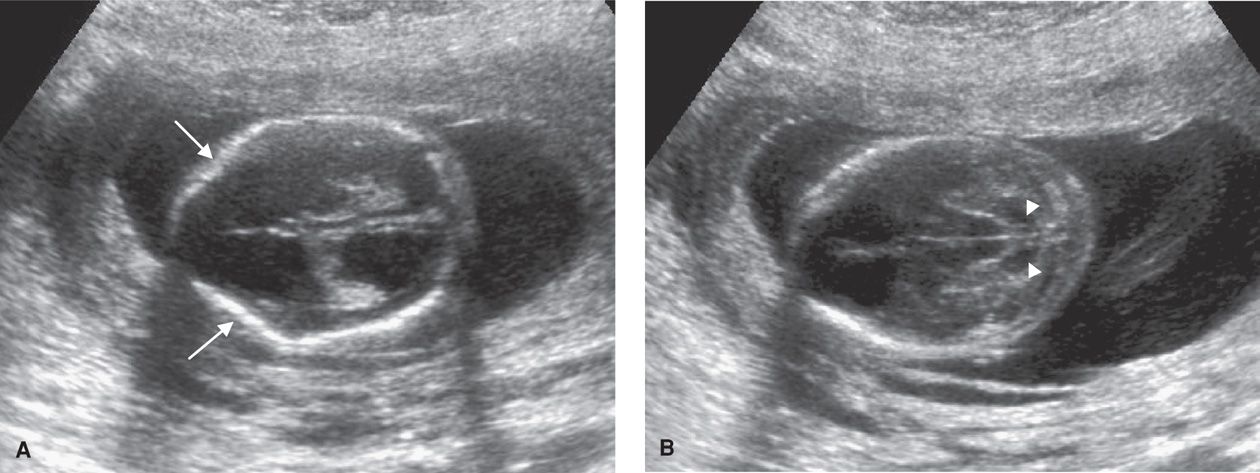
FIGURE 14-4 A. Image of the fetal head at the level of the lateral ventricles in the setting of spina bifida, demonstrating inward bowing or scalloping of the frontal bones (arrows)—the lemon sign. The image also depicts ventriculomegaly. B. Image of the fetal head at the level of the posterior fossa, demonstrating anterior curvature of the cerebellum (white arrows) with effacement of the cisterna magna—the banana sign.
Most centers use targeted sonography as the primary method of evaluating maternal serum AFP level elevation. The American College of Obstetricians and Gynecologists (2013b) recommends that women be counseled regarding the risks and benefits of targeted sonography and amniocentesis, the risk associated with the degree of AFP level elevation or other risk factors, and the quality and findings of the sonographic examination before making a decision.
Amniocentesis
Although amniocentesis for amnionic fluid AFP measurement was once considered the standard for open NTD diagnosis, it has been replaced in most centers by targeted sonography. If the amnionic fluid AFP level was elevated, an assay for acetylcholinesterase was performed, and if positive, was considered diagnostic of an NTD. Acetylcholinesterase leaks directly from exposed neural tissue into the amnionic fluid. The overall sensitivity of amniocentesis is approximately 98 percent for open NTDs, with a false-positive rate of 0.4 percent (Milunsky, 2004). Other fetal abnormalities associated with elevated amnionic fluid AFP levels and positive assay for acetylcholinesterase include ventral wall defects, esophageal atresia, fetal teratoma, cloacal exstrophy, and skin abnormalities such as epidermolysis bullosa.
Unexplained Maternal Serum AFP Level Elevation
If no fetal or placental abnormality is detected after a specialized sonographic evaluation, with or without amniocentesis, then an MSAFP elevation is considered unexplained. These pregnancies are at increased risk for various subsequent adverse pregnancy outcomes. These include fetal abnormalities or genetic syndromes not detectable sonographically, fetal-growth restriction, oligohydramnios, placental abruption, preterm membrane rupture, preterm birth, and fetal death. Many of these complications are assumed to result from placental damage or dysfunction. Importantly, AFP level elevation is not considered to be clinically useful as a screening tool for adverse pregnancy outcomes, due to its low sensitivity and positive predictive value. No specific program of maternal or fetal surveillance has been found to favorably affect pregnancy outcomes (Dugoff, 2010). At Parkland Hospital, prenatal care for these women is not altered unless a specific complication arises. Despite the extensive list of possible adverse outcomes, it is reassuring that most women with unexplained AFP level elevation have normal outcomes. Abnormally high or low values of other serum analytes used in aneuploidy screening protocols are reviewed on page 290.
 Management of the Fetus with Spina Bifida
Management of the Fetus with Spina Bifida
The optimal mode of delivery for a fetus with open spina bifida remains controversial. Some have recommended cesarean delivery before the onset of labor, positing that it may reduce the risk of mechanical trauma and spinal infection. Although some have found improved motor function in children delivered operatively, others have not identified benefit in short- or long-term outcomes (Lewis, 2004; Luthy, 1991; Merrill, 1998). The American College of Obstetricians and Gynecologists (2013b) recommends that the route of delivery for the fetus with spina bifida should be individualized.
Open fetal surgery to repair NTDs has been the subject of several clinical studies. A landmark trial designed to compare open fetal surgery for spina bifida with standard postnatal care was described by Adzick and colleagues (2011). These authors of the Management of Myelomeningocele or MOMS trial found that in selected cases, fetal surgery resulted in improved motor outcomes and reduced need for ventriculoperitoneal shunt placement at age 2 to 3 years. They also reported, however, that the surgery itself was associated with significant maternal and fetal risks. This is discussed further in Chapter 16 (p. 325).
DOWN SYNDROME AND OTHER ANEUPLOIDIES
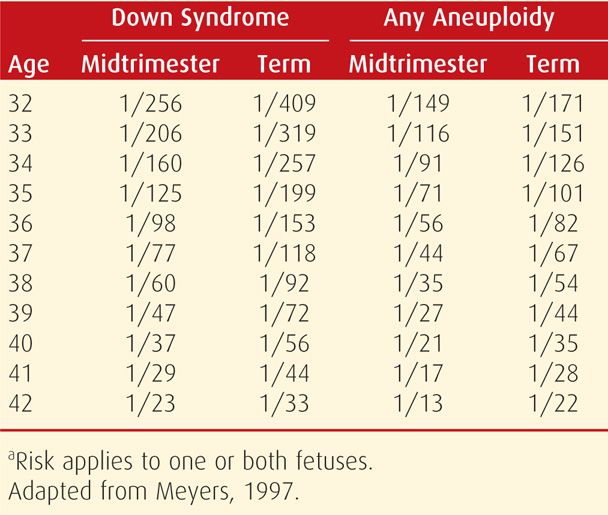
At least 8 percent of conceptuses are aneuploid, accounting for 50 percent of first-trimester abortions and 5 to 7 percent of all stillbirths and neonatal deaths. As discussed in Chapter 13 (p. 260), the risk of fetal trisomy increases with maternal age, particularly after age 35. Specific maternal age-related aneuploidy risks for singleton and twin pregnancies are shown in Tables 14-3 and 14-4. Other significant risk factors include a prior pregnancy with autosomal trisomy or triploidy or a woman or her partner with a numerical chromosomal abnormality or structural chromosomal rearrangement, such as a balanced translocation.
TABLE 14-3. Maternal Age-Related Risk for Down Syndrome and Any Aneuploidy at Midtrimester and at Term in Singleton Pregnancies
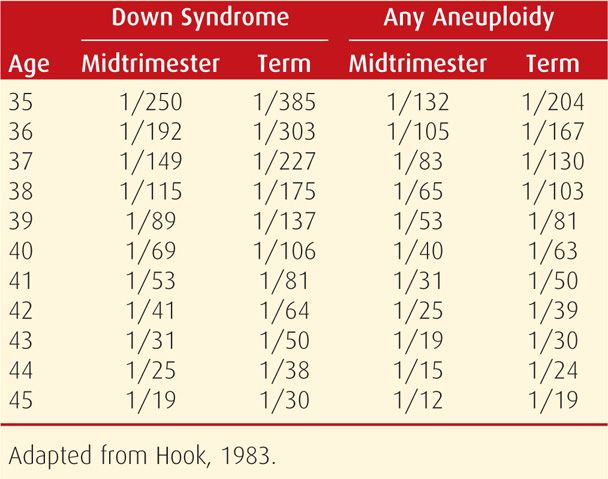
TABLE 14-4. Maternal Age-Related Risk for Down Syndrome and Any Aneuploidy at Midtrimester and at Term in Dizygotic Twin Pregnanciesa
 Types of Screening Tests
Types of Screening Tests
Until the mid-1980s, prenatal diagnostic testing for fetal aneuploidy was offered for “advanced maternal age.” However, age alone is a poor screening test, because approximately 70 percent of Down syndrome pregnancies are in women younger than 35 years. Nearly 30 years ago, Merkatz and associates (1984) observed that pregnancies with Down syndrome were characterized by lower maternal serum AFP levels at 15 to 20 weeks, and screening became available for younger women. During the past two decades, there have been four major advances in the area of aneuploidy screening:
1. The addition of other serum analytes to second-trimester screening has improved Down syndrome detection rates to approximately 80 percent for the quadruple marker test (Table 14-5).
2. First-trimester screening at 11 to 14 weeks’ gestation, using the fetal nuchal translucency measurement together with serum analytes, has achieved Down syndrome detection rates comparable to those for second-trimester screening in women younger than 35 years (American College of Obstetricians and Gynecologists, 2013c).
3. Combinations of first- and second-trimester screening yield Down syndrome detection rates as high as 90 to 95 percent (Malone, 2005b).
4. Maternal serum cell-free fetal DNA testing for trisomy 21, 18, and 13 has become available as a screening test for high-risk pregnancies, with a 98-percent detection rate and a false-positive rate of 0.5 percent (American College of Obstetricians and Gynecologists, 2012b; Bianchi, 2012; Palomaki, 2011, 2012).
TABLE 14-5. Selected Down Syndrome Screening Strategies and Their Detection Rate
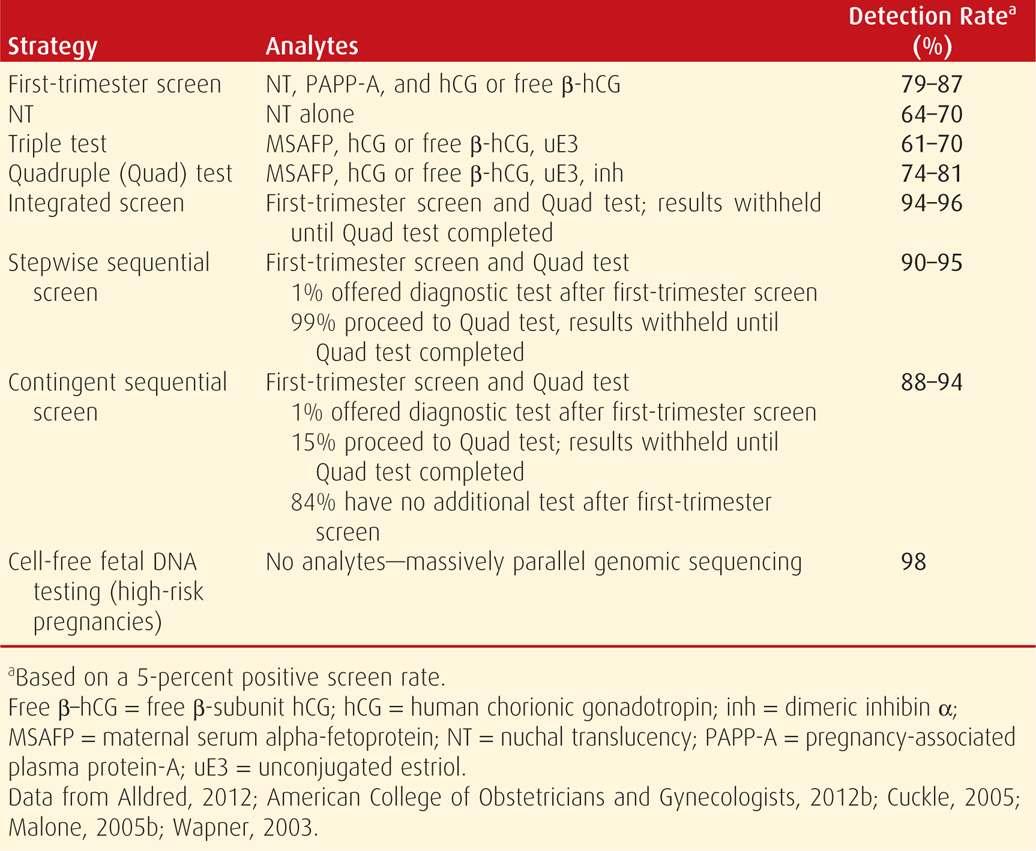
With the exception of cell-free fetal DNA testing, each first- and/or second-trimester aneuploidy screening test is based on a composite likelihood ratio, and the maternal age-related risk is multiplied by this ratio. This principle also applies to modification of the Down syndrome risk by selected sonographic findings (p. 292). Each woman is provided with a specific risk, expressed as a ratio—1:X. However, each screening test has a predetermined value at which it is deemed “positive” or abnormal. For second-trimester tests, this threshold has traditionally been set at the risk for fetal Down syndrome in a woman aged 35 years—approximately 1 in 385 at term (see Table 14-3). Women with a positive screening test result should be offered a diagnostic test for fetal karyotype by either chorionic villus sampling or amniocentesis (American College of Obstetricians and Gynecologists, 2012a).
Counseling
Because technology advances have resulted in improved aneuploidy detection with available screening tests, the American College of Obstetricians and Gynecologists (2013c) recommends that all women who present for prenatal care before 20 weeks be offered screening. Available screening paradigms are shown in Table 14-5. A positive screening test result indicates increased risk, but it is not diagnostic of aneuploidy. Conversely, a negative screening test indicates that the risk is not increased, but it does not guarantee a normal fetus. Although Down syndrome is the focus of most aneuploidy screening protocols, it accounts for only half of all fetal chromosomal abnormality cases. Invasive diagnostic tests such as chorionic villus sampling and amniocentesis are safe and effective. Regardless of age, all women are counseled regarding the differences between screening and diagnostic tests, and they are given the option of invasive diagnostic testing.
 First-Trimester Screening
First-Trimester Screening
The most commonly used protocol involves measurement of sonographic nuchal translucency and two maternal serum analytes. This is performed between 11 and 14 weeks’ gestation.
Nuchal Translucency (NT)
This is the maximum thickness of the subcutaneous translucent area between the skin and soft tissue overlying the fetal spine at the back of the neck (Fig. 14-5). It is measured in the sagittal plane, when the crown-rump length measures between 38 and 84 mm. Specific criteria for NT measurement are listed in Table 10-3 (p. 196). The NT measurement is expressed as a multiple of the gestational age-specific median, similar to serum markers used for aneuploidy screening. An increased NT thickness itself is not a fetal abnormality, but rather is a marker that confers increased risk. Approximately one third of fetuses with increased nuchal translucency thickness will have a chromosome abnormality, nearly half of which are Down syndrome (Snijders, 1998).
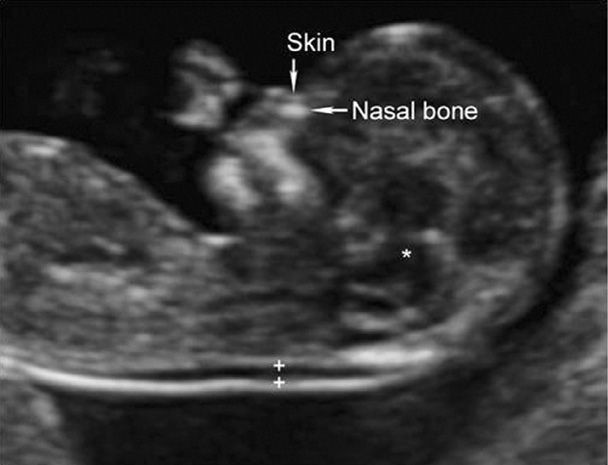
FIGURE 14-5 Sagittal image of a normal, 12-week fetus demonstrating correct caliper placement (+) for nuchal translucency measurement. The fetal nasal bone and overlying skin are indicated. The nasal tip and the 3rd and 4th ventricles (asterisk), which are other landmarks that should be visible in the nasal bone image, are also shown. (Image contributed by Dr. Michael Zaretsky.)
As shown in Table 14-5, as an isolated marker, NT detects 64 to 70 percent of fetuses with Down syndrome at a false-positive rate of 5 percent, and it has maximal sensitivity at 11 weeks (Malone, 2005b). The risk conferred by an increased NT thickness is independent of that of serum analytes, and combining NT with serum analyte values results in greatly improved aneuploidy detection (Spencer, 1999). Thus, NT is generally used as an isolated marker only in screening for multifetal gestations, in which serum screening is not as accurate or may not be available (American College of Obstetricians and Gynecologists, 2013c). An exception is that if the NT measurement is increased to 3 to 4 mm, then the aneuploidy risk is unlikely to be normalized using serum analyte assessment, and invasive testing should be offered (Comstock, 2006).
Increased NT thickness is also associated with other aneuploidies, genetic syndromes, and various birth defects, especially fetal cardiac anomalies (Atzei, 2005; Simpson, 2007). Because of this, if the NT measurement is 3.5 mm or greater, the patient should be offered targeted sonography, with or without fetal echocardiography, in addition to fetal karyotyping (American College of Obstetricians and Gynecologists, 2013c).
The NT must be imaged and measured with a high degree of precision for aneuploidy detection to be accurate. This has led to standardized training, certification, and ongoing quality review programs. In the United States, training, credentialing, and monitoring are available through the Nuchal Translucency Quality Review (NTQR) program (www.ntqr.org). Training is also available through the Fetal Medicine Foundation (www.fetalmedicineusa.com). In addition to nuchal translucency, NTQR provides an educational process leading to certification in measurement of the fetal nasal bone, which is discussed on page 294 and shown in Figure 14-5.
Serum Analytes
Two analytes used for first-trimester aneuploidy screening are human chorionic gonadotropin—either intact or free β-hCG—and pregnancy-associated plasma protein A (PAPP-A). In cases of fetal Down syndrome, the first-trimester serum free β-hCG level is higher, approximately 2.0 MoM, and the PAPP-A level is lower, approximately 0.5 MoM. With trisomy 18 and trisomy 13, levels of both analytes are lower (Cuckle, 2000; Malone, 2005b; Spencer, 1999, 2000; Tul, 1999). If gestational age is correct, the use of these serum markers—without NT measurement—results in detection rates for fetal Down syndrome up to 67 percent at a false-positive rate of 5 percent (Wapner, 2003). Aneuploidy detection is significantly greater if these first-trimester analytes are either: (1) combined with the sonographic NT measurement or (2) combined with second-trimester analytes, which is termed serum integrated screening (p. 291).
In twin pregnancies, serum free β-hCG and PAPP-A levels are approximately doubled compared with singleton values (Vink, 2012). Even with specific curves, a normal dichorionic cotwin will tend to normalize screening results, and thus, the aneuploidy detection rate is at least 15-percent lower (Bush, 2005).
Combined First-Trimester Screening
The most commonly used screening protocol combines the NT measurement with serum hCG and PAPP-A. Using this protocol, Down syndrome detection rates in large prospective trials range from 79 to 87 percent, at a false-positive rate of 5 percent (see Table 14-5). The detection rate is approximately 5-percent higher if performed at 11 compared with 13 weeks (Malone, 2005b). The detection rate for trisomies 18 and 13 is approximately 90 percent, at a 2-percent false-positive rate (Nicolaides, 2004; Wapner, 2003).
Maternal age does affect the performance of first-trimester aneuploidy screening tests. In prospective trials, combined first-trimester screening resulted in Down syndrome detection rates of 67 to 75 percent in women younger than 35 years at delivery, which are 10-percent lower than the overall detection rates in these studies (Malone, 2005b; Wapner, 2003). Among women older than 35 at delivery, however, Down syndrome detection rates were 90 to 95 percent, albeit at a higher false-positive rate of 15 to 22 percent.
Unexplained Abnormalities of First-Trimester Analytes
There is a significant association between serum PAPP-A levels below the 5th percentile and preterm birth, growth restriction, preeclampsia, and fetal demise (Dugoff, 2004). Similarly, low levels of free β-hCG have been associated with fetal demise (Goetzl, 2004). The sensitivity and positive-predictive values of these markers are considered too low to be clinically useful as screening tests. As with other serum analyte level abnormalities, no management strategies have been demonstrated to improve pregnancy outcomes when these marker levels are abnormally low (Dugoff, 2010).
 Second-Trimester Screening
Second-Trimester Screening
Pregnancies with fetal Down syndrome are characterized by lower maternal serum AFP levels—approximately 0.7 MoM, higher hCG levels—approximately 2.0 MoM, and lower unconjugated estriol levels—approximately 0.8 MoM (Merkatz, 1984; Wald, 1988). This triple test can detect 61 to 70 percent of Down syndrome cases as shown in Table 14-5 (Alldred, 2012). Levels of all three markers are decreased in the setting of trisomy 18, with a detection rate similar to that for Down syndrome at a false-positive rate of only 0.5 percent (Benn, 1999).
Levels of a fourth marker—dimeric inhibin alpha—are elevated in Down syndrome, with an average value of 1.8 MoM (Spencer, 1996). The addition of dimeric inhibin to the other three markers is the quadruple or quad test, which has a trisomy 21 detection rate of approximately 80 percent at a false-positive rate of 5 percent (see Table 14-5). As with first-trimester screening, aneuploidy detection rates will be slightly lower in younger women and higher in women older than 35 years at delivery. If second-trimester serum screening is used in twin pregnancies, aneuploidy detection rates are significantly lower (Vink, 2012).
The quad test is the most commonly used second-trimester serum screening test for aneuploidy. As a stand-alone test, it is generally used if women do not begin care until the second-trimester or if first-trimester screening is not available. As subsequently discussed, combining the quad test with first-trimester screening yields even greater aneuploidy detection rates.
Unexplained Abnormalities of Second-Trimester Analytes
There is a significant association between second-trimester elevation of either hCG or dimeric inhibin alpha levels and adverse pregnancy outcomes. The outcomes reported are similar to those associated with AFP level elevation and include fetal-growth restriction, preeclampsia, preterm birth, fetal demise, and stillbirth. Moreover, the likelihood of adverse outcome is increased when multiple marker levels are elevated (Dugoff, 2005). However, the sensitivity and positive predictive values of these markers are considered too low to be useful for screening or management (Dugoff, 2010).
Low Maternal Serum Estriol Levels. A maternal serum estriol level < 0.25 MoM has been associated with two uncommon but important conditions. The first, Smith-Lemli-Opitz syndrome, is an autosomal recessive condition characterized by mutations in the 7-dehydrocholesterol reductase gene. It may be associated with central nervous system, heart, kidney, and extremity abnormalities, with ambiguous genitalia, and with fetal-growth restriction. For this reason, the Society for Maternal-Fetal Medicine has recommended that sonographic evaluation be performed if an unconjugated estriol level is < 0.25 MoM (Dugoff, 2010). If abnormalities are identified, an elevated amnionic fluid 7-dehydrocholesterol level can confirm the diagnosis.
The second condition is steroid sulfatase deficiency, also known as X-linked ichthyosis. It is typically an isolated condition, but it may also occur in the setting of a contiguous gene deletion syndrome (Chap. 13, p. 266). In such cases, it may be associated with Kallmann syndrome, chondrodysplasia punctata, and/or mental retardation (Langlois, 2009). If the estriol level is < 0.25 MoM and the fetus appears to be male, fluorescence in situ hybridization to assess the steroid sulfatase locus on the X-chromosome may be considered (Dugoff, 2010).
 Combined First- and Second-Trimester Screening
Combined First- and Second-Trimester Screening
Combined screening strategies enhance aneuploidy detection. For this reason, the American College of Obstetricians and Gynecologists (2013c) recommends that a strategy incorporating both first- and second-trimester screening should be offered to women who seek prenatal care in the first trimester. Three types of screening strategies are available:
1. Integrated screening combines results of first- and second- trimester tests. This includes a combined measurement of fetal NT and serum analyte levels at 11 to 14 weeks’ gestation plus quadruple markers at 15 to 20 weeks. An aneuploidy risk is then calculated from these seven parameters. As expected, integrated screening has the highest Down syndrome detection rate—94 to 96 percent at a false-positive rate of 5 percent (see Table 14-5). If NT measurement is not available, serum integrated screening includes all six serum markers to calculate risk. This screening, however, is less effective.
2. Sequential screening discloses the results of first-trimester screening to women at highest risk, who are then offered invasive testing with chorionic villus sampling or amniocentesis. There are two testing strategies in this category:
• With stepwise sequential screening, women with first-trimester screen results that confer risk for Down syndrome above a particular threshold are offered invasive testing, and the remaining women receive second-trimester screening. The threshold is set at approximately 1 percent, because in a screened population, the 1 percent at highest risk includes approximately 70 percent of Down syndrome pregnancies (Cuckle, 2005). This method of screening may achieve up to a 95-percent detection rate (see Table 14-5).
• With contingent sequential screening, women are divided into high-, moderate-, and low-risk groups. Those at highest risk, for example, the top 1 percent, are offered invasive testing. Women at moderate risk, who comprise 15 to 20 percent of the population, undergo second-trimester screening. The remaining 80 to 85 percent, who are at or below a 1:1000 risk, receive negative screening test results and have no further testing (Cuckle, 2005). Thus, most of those screened are provided with results almost immediately while still maintaining a high detection rate. This rate ranges from 88 to 94 percent (see Table 14-5). This option is also more cost-effective because a second-trimester test is obviated in up to 85 percent of patients.
Integrated and sequential screening strategies require coordination between the provider and laboratory to ensure that the second sample is obtained during the appropriate gestational age window, sent to the same laboratory, and linked to the first-trimester results.
 Cell-Free Fetal DNA Screening
Cell-Free Fetal DNA Screening
Using massively parallel sequencing or chromosome selective sequencing to isolate cell-free fetal DNA from maternal plasma, fetal Down syndrome and other autosomal trisomies may be detected as early as 10 weeks’ gestation (Chap. 13, p. 279). Recent trials of these techniques in high-risk pregnancies have yielded detection rates for trisomies 21, 18, and 13 of approximately 98 percent at a false-positive rate of 0.5 percent or less (American College of Obstetricians and Gynecologists, 2012b; Bianchi, 2012; Palomaki, 2011, 2012; Sparks, 2012). This novel technology has recently become clinically available as a screening test, but it is not considered a replacement diagnostic test. Pretest counseling is recommended. If an abnormal result is identified, genetic counseling should be performed, and invasive prenatal diagnostic testing should be offered to confirm the results. The American College of Obstetricians and Gynecologists (2012b) currently recommends that the test may be offered to the following groups:
• Women 35 years or older at delivery
• Those with sonographic findings indicating increased risk for fetal aneuploidy
• Those with a prior pregnancy complicated by trisomy 21, 18, or 13
• Patient or partner carries a balanced robertsonian translocation indicating increased risk for fetal trisomy 21 or 13
• Those with an abnormal first-, second-, or combined first- and second-trimester screening test result for aneuploidy.
The College does not recommend offering the test to women with low-risk pregnancies or multifetal gestations (American College of Obstetricians and Gynecologists, 2012b).
 Sonographic Screening
Sonographic Screening
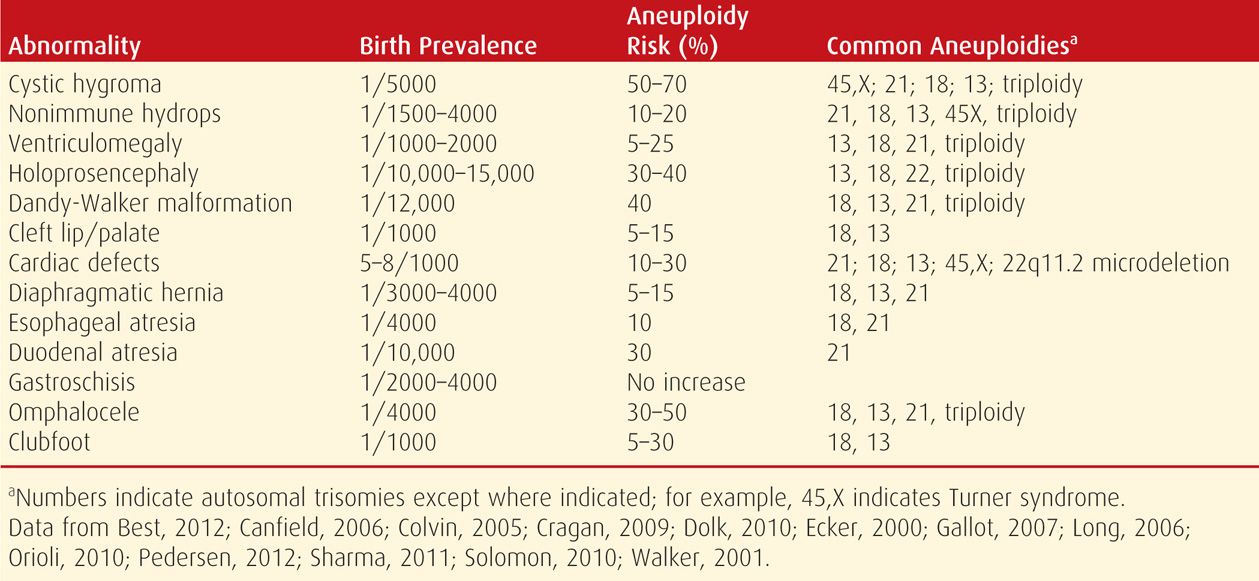
Major abnormalities and minor sonographic markers contribute significantly to aneuploidy detection. As shown in Table 14-6, with few exceptions, the aneuploidy risk associated with any major abnormality is high enough to warrant offering an invasive test for fetal karyotype and/or chromosomal microarray analysis (Chap. 13, p. 275). Importantly, a fetus with one abnormality may have others that are less likely to be detected sonographically or even undetectable sonographically but that greatly affect the prognosis nonetheless. Most fetuses with aneuploidy that is likely to be lethal in utero—such as trisomy 18 and 13 and triploidy—usually have sonographic abnormalities that can be seen by the second trimester. However, only 25 to 30 percent of second-trimester fetuses with Down syndrome will have a major malformation that can be identified sonographically (Vintzileos, 1995).
TABLE 14-6. Aneuploidy Risk Associated with Selected Major Fetal Anomalies
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


