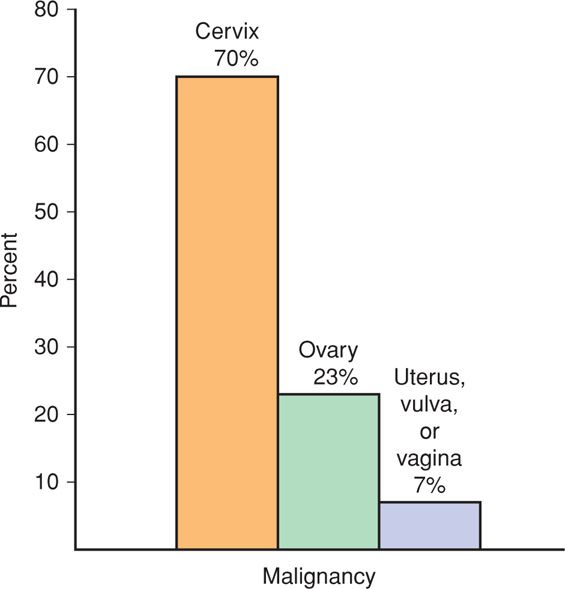
FIGURE 63-1 Proportion of malignancies during pregnancy and within 12 months of delivery in 4.85 million women from the California Cancer Registry. GI = gastrointestinal. (Data from Smith, 2003.)
PRINCIPLES OF CANCER THERAPY RELATED TO PREGNANCY
Management of the pregnant woman with cancer poses unique problems related to fetal concerns. Because of these, treatment must be individualized. Considerations include the type and stage of cancer and the desire for pregnancy continuation with the inherent risks associated with modifying or delaying treatment.
 Surgery
Surgery
Operative procedures indicated for cancer include those for diagnosis, staging, or therapy. Fortunately, most procedures that do not interfere with the reproductive tract are well tolerated by both mother and fetus (Chap. 46, p. 927). Although many procedures have classically been deferred until after 12 to 14 weeks’ gestation to minimize abortion risks, this probably is not necessary. We are of the opinion that surgery should be performed regardless of gestational age if maternal well-being is imperiled.
Both pregnancy and malignancy are risk factors for venous thromboembolism. There are no guidelines specific for thromboprophylaxis in pregnant women undergoing surgical procedures for cancer. Thus, depending on the complexity of the planned procedure, it seems reasonable to use mechanical prophylaxis compression or pneumatic stockings, low-molecular-weight heparin, or both (Chap. 52, p. 1044). There are general evidenced-based practice guidelines available from the American College of Chest Physicians (Guyatt, 2012).
 Diagnostic Imaging
Diagnostic Imaging
Sonography is a preferred imaging tool during pregnancy when appropriate. In addition, according to the American College of Obstetricians and Gynecologists (2009), most diagnostic radiographic procedures have very low x-ray exposure and should not be delayed if they would directly affect therapy (Chap. 46, p. 932). Computed tomography is useful for imaging extraabdominal tumors, and abdominal shielding usually helps to decrease fetal exposure. Routine magnetic resonance (MR) imaging use is not recommended, and if needed, is used preferentially after the first trimester. Gadolinium contrast administered with MR imaging crosses the placenta, resulting in high fetal concentrations. It should not be used in the first trimester and should be used later in pregnancy only when the benefits overwhelmingly outweigh the risks (Kanal, 2007). Some radioisotopes are relatively safe, but positron emission tomography (PET) scanning is usually not performed during pregnancy. Moreover, 18F-FDG (fludeoxyglucose) is concentrated in both breast tissue and milk. Thus, in the puerperium, breast feeding should be discontinued for 72 hours following a procedure (Rizack, 2009).
 Radiation Therapy
Radiation Therapy
Therapeutic radiation often results in significant fetal exposure depending on the dose, tumor location, and field size. The most susceptible period is organogenesis, although there is no gestational age considered safe for therapeutic radiation exposure. Adverse effects include cell death, carcinogenesis, and mutagenic changes in future generations (Brent, 1999; Hall, 1991). Characteristic fetal effects are microcephaly and mental retardation, and even late exposure can cause growth restriction and brain damage. Thus, radiotherapy to the maternal abdomen is contraindicated. With some head and neck cancers, however, radiotherapy to supradiaphragmatic areas can be used relatively safely with abdominal shielding. In others, such as breast cancer, significant scatter doses can accrue to the fetus.
 Chemotherapy
Chemotherapy
Various antineoplastic drugs may be given for primary treatment or for adjunctive therapy. Although chemotherapy often improves long-term maternal outcomes, many are reluctant to employ it during pregnancy. As discussed in Chapter 12 (p. 248), fetal concerns include malformations, growth restriction, mental retardation, and the risk of future malignancies. Risks are dependent primarily on fetal age at exposure, and most agents are potentially detrimental given in the first trimester during organogenesis. Indeed, embryonic exposure to cytotoxic drugs may cause major malformations in up to 20 percent of cases (Pavlidis, 2011).
After organogenesis, most antineoplastic drugs are without immediate obvious adverse sequelae (Abdel-Hady, 2012; Cardonick, 2004). Similarly, late mutagenic effects appear limited. Children born to 68 women receiving chemotherapy and who were assessed at 18 months had no excessive adverse effects to their central nervous system, heart, or hearing or to their general health or growth (Amant, 2012b).
Although not always practicable, some recommend that chemotherapy be withheld in the 3 weeks before expected delivery because neutropenia or pancytopenia might cause undue risk for maternal infection or hemorrhage. Another concern is that neonatal hepatic and renal clearance of chemotherapy metabolites is limited (Ko, 2011). For these reasons, chemotherapy is also contraindicated with breast feeding.
 Molecular Therapy
Molecular Therapy
Drugs designed to stimulate hemopoiesis are commonly used with cancer treatments. Some of these include the granulocyte colony-stimulating factors filgrastim (Neupogen) and pegfilgrastim (Neulasta). If required in pregnancy, limited data support the safety of these factors (Dale, 2003). Red blood cells can be stimulated by erythropoietin alfa (Procrit), which likely is also safe in pregnancy (Briggs, 2011).
 Immunotherapy
Immunotherapy
Hybridized monoclonal antibodies directed against tumor-specific antigens are designed to treat an ever-increasing list of malignant neoplasms. Some of these drugs are described in the discussion of tumors for which they are used. Little is known regarding their fetal effects, but concerns for teratogenicity by tyrosine kinase inhibitors—for example, imatinib (Gleevec)—have been raised (Ali, 2009).
 Fertility and Pregnancy after Cancer Therapy
Fertility and Pregnancy after Cancer Therapy
The number of cancer survivors continues to increase. From the Childhood Cancer Survival Study (CCSS), a fourth of survivors have significant morbidity at 25 years (Diller, 2011). One is that subsequent fertility may be diminished after chemotherapy or radiotherapy, and couples should be counseled regarding potential risks (American Society for Reproductive Medicine, 2013). Guidelines for this counseling have been developed by the American Society of Clinical Oncology (Loren, 2013).
Embryo cryopreservation is standard and widely available, however, other methods are considered investigational and limited to referral centers. Some are oocyte or ovarian cryopreservation, preradiotherapy ovarian transposition, and/or ovarian suppression with gonadotropin-releasing hormone agonists before therapy. Educational materials concerning these methods have been provided by a National Institutes of Health (NIH) interdisciplinary group—the Oncofertility Consortium (Woodruff, 2010). This is available online at: http://oncofertility.northwestern.edu.
Not surprisingly, abdominopelvic radiation impairs subsequent reproductive function (Wo, 2009). Data from the CCSS are informative for women who conceive following cancer therapy. Since the database was established in 1994, 4029 pregnancies have been reported in 1953 women. A number of reports have described increased adverse pregnancy outcomes by type of cancer treatment given. For example, fetuses born to women who had undergone prior pelvic radiation were more likely to have a birthweight < 2500 g (Green, 2002; Robison, 2005). There was a higher rate of preterm birth in cancer survivors—especially those given pelvic radiotherapy—compared with their siblings (Signorello, 2006). In another study of 917 pregnancies, cancer survivors had slightly higher rates of preterm birth and postpartum hemorrhage (Clark, 2007). In yet another study of 1657 women from the CCSS, Signorello and colleagues (2010) reported that uterine or ovarian irradiation significantly increased the risk of stillbirth and neonatal death. One possible explanation for adverse reproductive effects is that radiotherapy, but not chemotherapy, at a young age irreversibly reduces the uterine volume in survivors (Larsen, 2004). Perhaps the most reassuring findings from all of these studies are that fetuses of women who were treated in childhood with radiotherapy did not have significantly increased risks for congenital malformations (Lawrenz, 2012; Winther, 2009).
 Placental Metastases
Placental Metastases
Tumors infrequently metastasize to the placenta. The most common types are malignant melanomas, leukemias, lymphomas, and breast cancer (Al-Adnani, 2007). Placentas from pregnancies in these and all other women with a malignancy should be sent for histological evaluation. Because tumor cells are usually confined within the intervillous spaces, fetal metastases are infrequent. Melanoma is the most common example, and typically fetal metastases are found in the liver or subcutaneous tissue with an 80-percent associated mortality rate (Alexander, 2003; Altman, 2003; Gottschalk, 2009).
REPRODUCTIVE TRACT NEOPLASMS
Benign neoplasms are common in the female reproductive tract and include leiomyomas, ovarian neoplasms, and endocervical polyps. Malignancies of these organs are the most frequent cancers encountered in pregnant women, and of these, cervical cancers make up the majority (Fig. 63-2).
FIGURE 63-2 Frequency of reproductive-tract malignancies in 844 pregnant women. (Data from Haas, 1984; Lutz, 1977; Smith, 2003.)
 Cervix
Cervix
Endocervical Polyp
A number of benign, premalignant, and malignant cervical neoplasms are frequently recognized during pregnancy. Endocervical polyps are overgrowths of benign endocervical stroma covered by epithelium. If asymptomatic, these polyps are usually seen at the time of the initial pelvic examination, although they can cause bleeding at any time. They typically appear as single, red, smooth, elongated fleshy masses of variable size that extend outward from the endocervical canal. Typically benign, they can however be a source of Pap smear results describing atypical glandular cells of undetermined significance—AGUS. And although malignant transformation in these growths is uncommon, polypoid cervical cancers can mimic these (Chin, 2002; Younis, 2010). Thus, most recommend their removal and evaluation. Polyps with a slender stalk are removed by grasping the tumor with ring or polyp forceps. It is then twisted repeatedly about its base to strangulate feeding vessels. With repeated twisting, the base narrows and avulses. Monsel solution, which is ferric subsulfate, can be applied with pressure to the stalk stub for hemostasis. A thick-pedicle polyp may sometimes warrant surgical excision.
Epithelial Neoplasia
Pregnancy provides an opportune time to screen for premalignant and malignant cervical epithelial neoplasia, especially in women without regular access to health care. With prenatal Pap smear screening, the pregnancy status is noted on the requisition form because interpretation may be hindered by pregnancy-associated physiological changes (Connor, 1998). Some changes include glandular epithelial hyperplasia, Arias-Stella reaction, and presence of decidual cells.
Screening guidelines also applicable in pregnant women were updated in 2012 by the American Society for Colposcopy and Cervical Pathology (ASCCP). These include: (1) no screening until age 21, (2) cytology alone every 3 years in those aged 21 to 29 years, and (3) in those older than 30, human papillomavirus (HPV) and cytology co-testing every 5 years, or cytology alone every 3 years. Importantly, these broad guidelines do not cover women in high-risk populations who may need more intensive screening. Examples include diethylstilbestrol (DES)-exposed women, cervical cancer survivors, and the immunocompromised (Saslow, 2012).
Oncogenic Human Papillomaviruses. Certain serovars of HPV are associated with high-grade intraepithelial lesions and invasive cancer. Some of these high-risk viruses—HPV types 16, 18, 31, 35, 45, 51, 52, and 56—may have an increased incidence in pregnant compared with nonpregnant women (Fife, 1996). Cotesting for such high-risk serotypes at the time of the Pap test is now incorporated into the recent screening guidelines for women 30 years and older. Cells can be collected directly into liquid-based cytology medium, namely, PreservCyt Solution of the ThinPrep Pap Test. Additionally, the cobas HPV Test and Digene HC2 HPV DNA test also allow collection into a specific collection medium if traditional cytology is performed.
HPV Vaccination. The clear link between HPV infection and cervical neoplasia has prompted development of two Food and Drug Administration-approved vaccines. These are available for girls and women aged 9 to 26 years. The quadrivalent HPV-6/11/16/18 vaccine (Gardasil) and the bivalent HPV-16/18 vaccine (Cervarix) decrease the incidence of high-grade dysplasia, and thus, cervical carcinoma (American College of Obstetricians and Gynecologists, 2012b; FUTURE II Study Group, 2007; Paavonen, 2009).
Vaccination is not recommended during pregnancy, and contraception during the series is recommended by the American College of Obstetricians and Gynecologists (2010) and the Centers for Disease Control and Prevention (2010). That said, both vaccines are considered safe and are not associated with higher rates of adverse pregnancy outcomes if conception occurs during the series (Dana, 2009; Descamps, 2009; Garland, 2009). Completion of the series should be delayed until after delivery. Both of these inactivated vaccines can be given postpartum in a typical regimen of three doses, with injections given at 0, at 1 to 2, and at 6 months. The vaccines are safe for use in breast-feeding women.
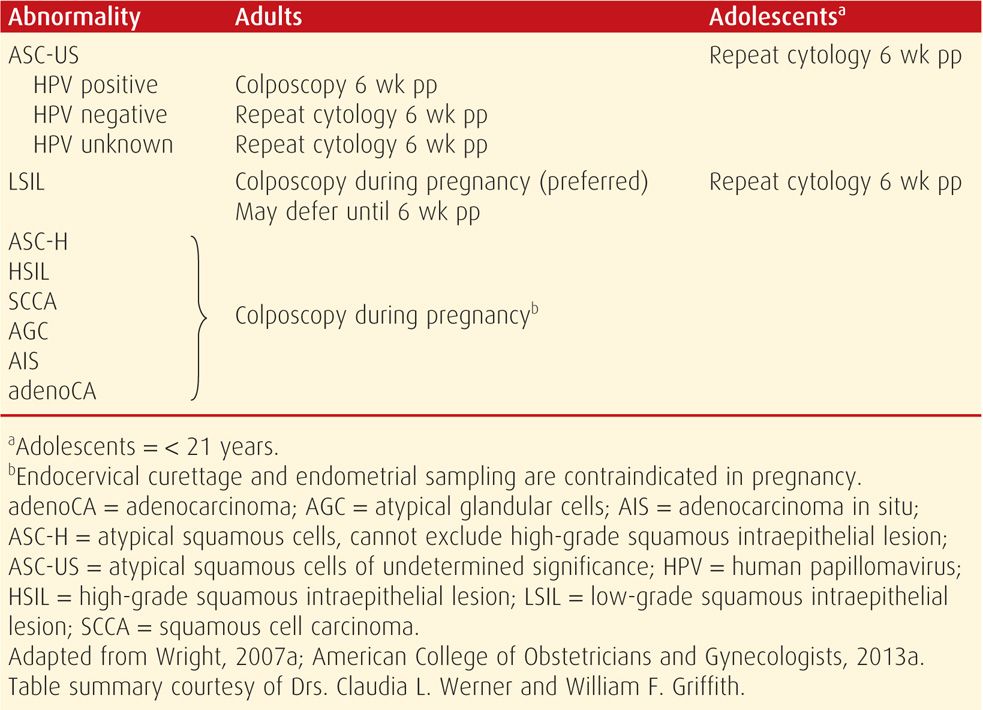
Abnormal Cytological Results. The incidence of abnormal cervical cytology during pregnancy is at least as high as that reported for nonpregnant women. Abnormal cytological findings and their suggested management according to consensus guidelines are summarized in Table 63-1. Many of these cytological abnormalities should prompt colposcopy to exclude invasive cancer. Accordingly, lesions suspicious for high-grade disease or cancer should undergo biopsy (American College of Obstetricians and Gynecologists, 2013a). These may actively bleed because of hyperemia, but this can be stopped with Monsel paste and pressure, silver nitrate application, vaginal packing, or suture.
TABLE 63-1. American Society for Colposcopy and Cervical Pathology (ASCCP) Guidelines for Initial Management of Epithelial Cell Abnormalities in Pregnancy
Unsatisfactory colposcopic evaluation is less common during pregnancy because the transformation zone is better exposed due to cervical eversion. With unsatisfactory visualization, colposcopy is repeated in 6 to 8 weeks, during which time the squamocolumnar junction will further evert (Randall, 2009). Once satisfactory examination is possible and advanced disease is excluded, additional colposcopic and cytological examinations during pregnancy are not encouraged. After delivery, repeat cytology and colposcopy should generally be delayed for at least 6 weeks to allow reparative healing.
Cervical Intraepithelial Neoplasia. Women with histologically confirmed intraepithelial neoplasia during pregnancy may be allowed to deliver vaginally, with further evaluation planned after delivery. For those with cervical intraepithelial neoplasia (CIN) 1, the recommended management is reevaluation postpartum. For those with CIN 2 or 3 in which invasive disease has been excluded, or with advanced pregnancy, repeat colposcopic and cytological evaluations are performed at intervals no more frequent than 12 weeks. Repeat biopsy is recommended if appearance of the lesion worsens or if cytology suggests invasive cancer. Alternatively, deferring reevaluation until at least 6 weeks postpartum is acceptable (Wright, 2007b).
Regression of CIN lesions is common during pregnancy or postpartum. In a study of 1079 pregnant women with cervical dysplasia in which biopsy correlated with colposcopic findings, 61 percent reverted to normal postpartum (Fader, 2010). In another study, Yost and colleagues (1999) reported postpartum regression in 70 percent of women with CIN 2 and CIN 3 lesions. And although 7 percent of women with CIN 2 lesions progressed to CIN 3, none progressed to invasive carcinoma. In another study of 77 women with carcinoma in situ (CIS) diagnosed during pregnancy, a third had postpartum regression, two thirds had persistent CIS, and only two women had microinvasive cancer on cone biopsy after delivery (Ackermann, 2006).
Adenocarcinoma in situ (AIS) is managed similarly to CIN 3 (Dunton, 2008). Thus, unless invasive cancer is identified, treatment of AIS is not recommended until 6 weeks postpartum (Wright, 2007b).
Cervical Conization. If invasive epithelial lesions are suspected, conization is indicated and may be done with loop electrosurgical excisional procedure (LEEP) or by cold knife conization. If possible, conization is avoided in pregnancy because of increased risks for abortion, membrane rupture, hemorrhage, and preterm delivery. The epithelium and underlying stroma within the endocervical canal cannot be extensively excised without the risk of membrane rupture. Residual disease is common. Of 376 conization procedures during pregnancy, Hacker and colleagues (1982) found residual neoplasia in 43 percent of subsequent specimens. In addition, nearly 10 percent of 180 pregnant women required transfusion after conization (Averette, 1970).
Women with CIN treated before pregnancy may encounter pregnancy complications. Cicatricial cervical stenosis is uncommon, but it may follow cervical trauma from conization, the loop electrosurgical excision procedure (LEEP), and laser surgery. Cervical stenosis almost always yields during labor. A so-called conglutinated cervix may undergo almost complete effacement without dilation, with the presenting part separated from the vagina by only a thin layer of cervical tissue. Dilatation usually promptly follows pressure with a fingertip, although manual dilatation or cruciate incisions may be required.
For decades, cold-knife conization has been associated with cervical incompetence and preterm birth. However, the relationship between preterm birth and LEEP continues to be debated, with some studies showing increased risk, but others not (Castanon, 2012; Jakobsson, 2009; Samson, 2005; Werner, 2010). An important study confounder is the increased risk of preterm birth in women with CIN compared with the general population even if they have not undergone an excisional procedure (Bruinsma, 2007; Shanbhag, 2009).
Invasive Cervical Cancer
The incidence of invasive cervical carcinoma has dramatically declined and approximates 1 in 8500 pregnancies in the United States (Pettersson, 2010; Smith, 2003). The diagnosis is confirmed with biopsies taken during colposcopy, with conization, or from a grossly abnormal lesion. Of the histological types, squamous cell carcinomas comprises 75 percent of all cervical cancers, whereas adenocarcinomas account for 20 to 25 percent. Cancers may appear as exophytic or endophytic growth; as a polypoid mass, papillary tissue, or barrel-shaped cervix; as a cervical ulceration or granular mass; or as necrotic tissue. A watery, purulent, foul, or bloody discharge may also be present. Biopsy with Tischler forceps is warranted for suspicious lesions.
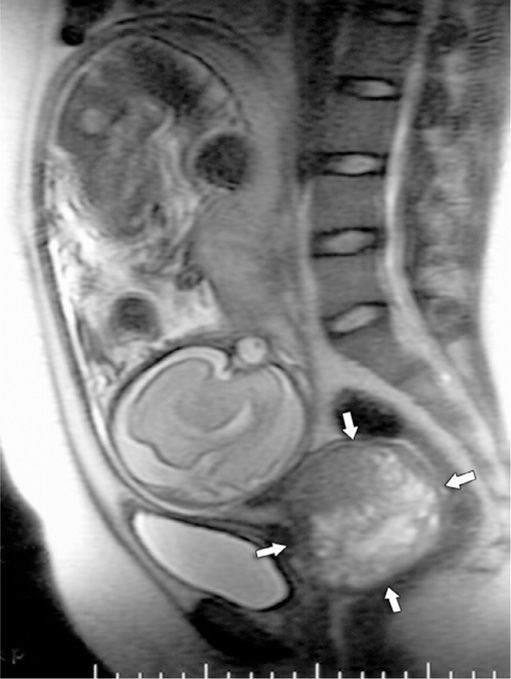
Cervical cancer is staged clinically, and 70 percent of cases that are diagnosed in pregnancy are stage I (Morice, 2012). Physiological pregnancy changes may impede accurate staging, and the extent of cancer is more likely to be underestimated in pregnant women (Oto, 2007). Specifically, induration of the broad ligament base, which characterizes tumor spread beyond the cervix, may be less prominent due to cervical, paracervical, and parametrial pregnancy-induced softening. Staging is modified and typically incorporates findings from pelvic examination and from renal sonography, chest radiography, cystoscopy, proctoscopy, and perhaps cone biopsy. Although MR imaging is not formally considered for clinical staging, it can be used without gadolinium contrast, ideally after the first trimester, to ascertain disease involvement of the urinary tract and lymph nodes (Fig. 63-3).
FIGURE 63-3 Sagittal T-2 weighted magnetic resonance image of a gravid uterus at 32 weeks’ gestation with a large cervical carcinoma (arrows).
Management and Prognosis. Treatment of cervical cancer in pregnant women is individualized and depends on the clinical stage, fetal age, and individual desire to continue pregnancy (Hunter, 2008). Stage IA1 is termed microinvasive disease and describes lesions with deepest invasion ≤ 3 mm and widest lateral extension ≤ 7 mm (FIGO Committee on Gynecologic Oncology, 2009b). If diagnosed by cone biopsy, treatment follows guidelines similar to those for intraepithelial disease. In general, continuation of pregnancy and vaginal delivery are considered safe, and definitive therapy is reserved until 6 weeks postpartum.
Truly invasive cancer demands relatively prompt therapy. During the first half of pregnancy, immediate treatment is advised by most, but this depends on the decision whether to continue pregnancy. With diagnosis during the latter half of pregnancy, most agree that pregnancy can safely be continued not only until fetal viability is reached, but also until fetal lung maturity is attained (Greer, 1989). In two studies with a total of 40 women past 20 weeks with either stage I or stage IIA carcinoma, it was concluded that delayed treatment was reasonable in women without bulky lesions (Takushi, 2002; van Vliet, 1998). Another option is to complete staging using laparoscopic lymphadenectomy and to delay treatment if metastases are excluded (Alouini, 2008; Favero, 2010). In a recent metaanalysis, neoadjuvant chemotherapy with platinum derivatives was found to be promising for treatment in pregnancy (Zagouri, 2013).
Although surgical therapy and radiation are equally effective, radical hysterectomy plus pelvic lymphadenectomy is the preferred treatment for invasive carcinoma in most women with stage I and early stage IIA lesions (American College of Obstetricians and Gynecologists, 2010a). Before 20 weeks, hysterectomy is usually performed with the fetus in situ. In later pregnancy, however, hysterotomy is often performed first. Radiotherapy for cervical cancer destroys ovarian and possibly sexual function, and frequently causes intestinal and urinary tract injury. In 49 women with pregnancy-associated stage IB cancer, there was a 30-percent severe complication rate from radiotherapy compared with only 7 percent with surgery (Nisker, 1983).
Although less commonly selected, fertility-preserving procedures for early-stage cervical cancers have been investigated. Ungár and colleagues (2006) performed abdominal radical trachelectomy before 20 weeks for stage IB1 carcinoma in five pregnant women. Yahata and associates (2008) treated four women at 16 to 23 weeks for stage IA1 adenocarcinoma with laser conization, and all delivered at term. Van Calsteren and coworkers (2008) reported similar success in a woman at 8 weeks with stage IB2 adenocarcinoma.
Radiotherapy is given for more advanced-stage cancer. External beam radiation is given for treatment early in pregnancy, and then if spontaneous abortion does not ensue, curettage is performed. During the second trimester, spontaneous abortion may not promptly occur and may necessitate hysterotomy in up to a fourth of cases.
Stage-for-stage, the survival rate for invasive cervical carcinoma is similar in pregnant and nonpregnant women (Sood, 1998). In a case-control study of 44 women with pregnancy-associated cervical cancer, the overall 5-year survival rate was approximately 80 percent in both pregnant women and nonpregnant controls (Van der Vange, 1995).
Delivery. Any adverse prognostic effects that vaginal delivery through a cancerous cervix might have are unknown. For this reason, the mode of delivery is controversial, especially for small, early-stage lesions. In some cases of bulky or friable lesions, there may be significant hemorrhage with vaginal delivery. Also, recurrences in the episiotomy scar, which result from tumor cells apparently “seeding” the episiotomy, have been reported (Goldman, 2003). Thus, most favor cesarean delivery and use a classical hysterotomy incision to avoid the risk of cutting through tumor.
Pregnancy after Radical Trachelectomy. There is growing experience with pregnancy in women who have previously undergone fertility-sparing radical trachelectomy for stage IB1 and IB2 cervical cancer before conception. During the typically vaginal procedure, the cervix is amputated at the level of the internal os, and a permanent-suture cerclage is placed around the isthmus for support in future pregnancies. The uterine isthmus is then reconstructed to the vagina. Because of the permanent cerclage, a classical cesarean incision is required for delivery.
Shepherd and colleagues (2006) presented outcomes for 123 such women cared for at their institution. Of 63 women who attempted pregnancy, 19 had 28 live births. All had classical cesarean deliveries, and a fourth were before 32 weeks. Similar findings were reported by Kim (2012) and Park (2014) and their colleagues.
 Uterus
Uterus
Leiomyomas
Also known as myomas and somewhat erroneously called fibroids, uterine leiomyomas are common benign smooth muscle tumors. Their incidence during pregnancy is approximately 2 percent, and the cited range depends on the frequency of routine sonography and population characteristics (Qidwai, 2006; Stout, 2010). For example, in a study of 4271 women, Laughlin and coworkers (2009) reported that first-trimester leiomyoma prevalence was highest in black women—18 percent—and lowest in whites—8 percent.
Leiomyomas vary in location and may develop as submucous, subserosal, or intramural growths. Less frequently, these develop in the cervix or broad ligament. Some become parasitic and their blood supply is derived from adjacent structures such as the highly vascularized omentum. In the rare manifestation of leiomyomatosis peritonealis disseminata, numerous subperitoneal benign smooth muscle tumors may appear similar to disseminated carcinomatosis. The tumors are likely caused by estrogen stimulation of multicentric subcoelomic mesenchymal cells to become smooth-muscle cells (Bulun, 2013). In their review of 45 cases, Lashgari and colleagues (1994) found that half were discovered during pregnancy. Often, these tumors regress after pregnancy.
The stimulatory effects of pregnancy on myoma growth are unpredictable and can be impressive. These tumors respond differently in individual women and may grow, regress, or remain unchanged in size during pregnancy (Laughlin, 2009; Neiger, 2006).
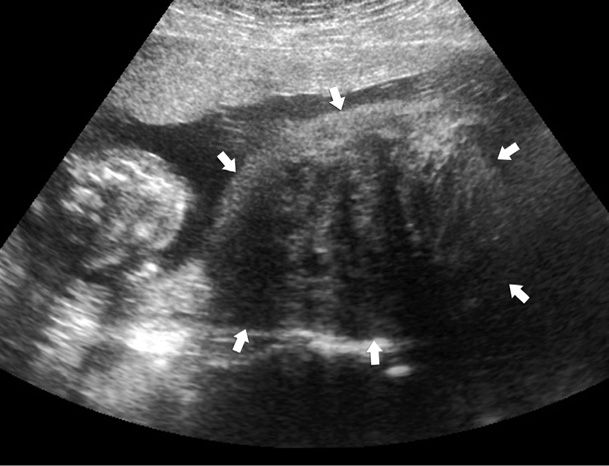
Especially during pregnancy, myomas can be confused with other adnexal masses, and sonographic imaging is indispensable (Fig. 63-4). In women in whom sonographic findings are unclear, MR imaging performed after the first trimester may be necessary (Torashima, 1997). Once diagnosed, leiomyomas do not require surveillance with serial sonography unless associated complications are anticipated.
FIGURE 63-4 Sonogram of a pregnant uterus with a large uterine leiomyoma. The heterogeneous mass (arrows) lies beside the fetus (seen in cross section) and has the classic appearance of a leiomyoma in pregnancy. The placenta is located anteriorly, and the mass originates from the posterior lower uterine segment and occupies more than half of the total uterine volume.
Symptoms. Most leiomyomas are asymptomatic, but acute or chronic pain or pressure may develop. For chronic pain secondary to large tumor size, nonnarcotic analgesic drugs may be given. More acutely, in some cases, myomas outgrow their blood supply and hemorrhagic infarction follows, which is termed red or carneous degeneration. Clinically, there is acute focal abdominal pain and tenderness, and sometimes a low-grade fever and leukocytosis is noted. As such, tumor degeneration may be difficult to differentiate from appendicitis, placental abruption, ureteral stone, or pyelonephritis. Sonographic imaging may be helpful, but close observation is requisite because an infarcted myoma is essentially a diagnosis of exclusion. In some women, preterm labor is stimulated by associated inflammation.
Treatment is analgesic medications, and most often, symptoms abate within a few days. If further treatment is given to forestall preterm labor, then close observation to exclude a septic cause is imperative. Although surgery is rarely necessary during pregnancy, myomectomy in highly selected cases has resulted in good outcomes (Celik, 2002; De Carolis, 2001). Most of 23 reported women were 14 to 20 weeks, and in almost half, surgery was performed because of pain. In some, an intramural leiomyoma was in contact with the implantation site. Except for one loss immediately following surgery at 19 weeks, most underwent cesarean delivery later at term.
Occasionally, pedunculated subserosal myomas will undergo torsion with subsequent painful necrosis. In such cases, laparoscopy or laparotomy can be used to ligate the stalk and resect the necrotic tumor. That said, we are of the opinion that surgery should be limited to tumors with a discrete pedicle that can be easily clamped and ligated.
Pregnancy Complications. Myomas are associated with a number of complications including preterm labor, placental abruption, fetal malpresentation, obstructed labor, cesarean delivery, and postpartum hemorrhage (Klatsky, 2008; Qidwai, 2006; Sheiner, 2004). In a review of pregnancy outcomes in 2065 women with leiomyomas, Coronado and coworkers (2000) reported that placental abruption and breech presentation were each increased fourfold; first-trimester bleeding and dysfunctional labor, twofold; and cesarean delivery, sixfold. Salvador and associates (2002) reported an eightfold increased second-trimester abortion risk in these women but found that genetic amniocentesis did not increase this risk.
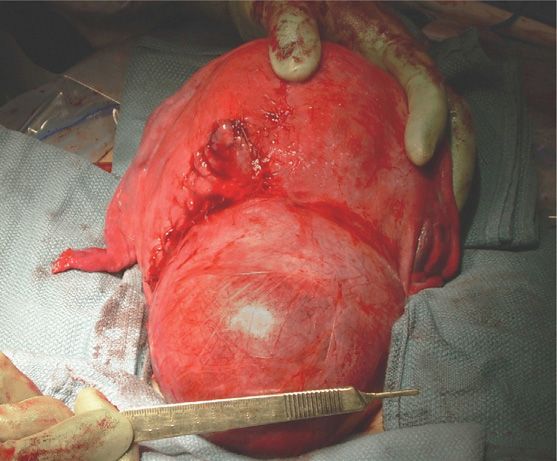
The two factors most important in determining morbidity in pregnancy are leiomyoma size and location (Shavell, 2012). The proximity of myomas to the placental implantation site is also a factor. Specifically, abortion, preterm labor, placental abruption, and postpartum hemorrhage all are increased if the placenta is adjacent to or implanted over a leiomyoma. Tumors in the cervix or lower uterine segment may obstruct labor, as did the one shown in Figure 63-5. Despite these complications, Qidwai and colleagues (2006) reported a 70-percent vaginal delivery rate in women in whom myomas were ≥ 10 cm. These data argue against empirical cesarean delivery for leiomyomas, and we allow a trial of labor unless myomas clearly obstruct the birth canal. If cesarean delivery is indicated, myomas are generally left alone unless they cause recalcitrant bleeding. An important caveat is that cesarean hysterectomy may be technically difficult because of lateral ureteral displacement by the tumors.
FIGURE 63-5 Cesarean delivery performed because of a large leiomyoma in the lower uterine segment. A classical vertical uterine incision, seen to the left of the myoma, was required for delivery of the fetus.
Bleeding due to myomas may develop during pregnancy from any of several factors. Especially common is bleeding with miscarriage, preterm labor, or placenta previa and abruption. Much less frequently, bleeding may result from a submucous myoma that has prolapsed from the uterus and into the cervix or vagina. In these unusual cases, although heavy or persistent bleeding may require earlier intervention, the stalk can be ligated vaginally near term if accessible to avoid tumor avulsion during delivery.
Fortunately, myomas rarely become infected (Genta, 2001). This is most frequent postpartum, especially if the tumor is located immediately adjacent to the implantation site (Lin, 2002). They also may become infected with an associated septic abortion and myoma perforation by a sound, dilator, or curette.
Fertility Considerations. Despite the relatively high prevalence of myomas in young women, it is not clear whether they diminish fertility, other than by possibly causing miscarriage as discussed in Chapter 18 (p. 359). In a review of 11 studies, Pritts (2001) concluded that submucous myomas did significantly affect fertility. He also found that hysteroscopic myomectomy improved infertility and early miscarriage rates in these women. Infertility-related myomas in other locations may require laparoscopy or laparotomy for excision.
Some of these methods of treatment for infertility may affect successful pregnancies. For example, after myomectomy, there is concern for uterine rupture before and during labor (American College of Obstetricians and Gynecologists, 2012a). Management is individualized, and review of the prior operative report is prudent. If resection resulted in a defect into or immediately adjacent to the endometrial cavity, then cesarean delivery is usually done before labor begins.
Although less effective than surgery, uterine artery embolization of myomas has also been used to treat infertility or myoma symptoms (Mara, 2008). Women so treated have increased rates of miscarriage, cesarean delivery, and postpartum hemorrhage (Homer, 2010). At this time, the Society of Interventional Radiology (Stokes, 2010) considers myoma embolization relatively contraindicated in women who plan future pregnancies.
Endometriosis and Adenomyosis
Rarely do endometriomas cause problems in pregnancy. If they are identified sonographically, they can be resected if cesarean delivery is performed. With vaginal delivery, endometriotic tumors or cysts may be removed after the puerperium or followed clinically depending on symptoms and cyst characteristics (Levine, 2010). Occasionally, endometriosis can develop after delivery from endometrial implants within abdominal incisions made at cesarean delivery or within episiotomy scars (Bumpers, 2002).
Adenomyosis is traditionally found in older women. Its acquisition may be at least partially related to disruption of the endometrial-myometrial border during sharp curettage for abortion (Curtis, 2002). Although problems during pregnancy are rare, in an 80-year review, Azziz (1986) reported concurrent adenomyosis to be associated with uterine rupture, ectopic pregnancy, uterine atony, and placenta previa.
Endometrial Carcinoma
This estrogen-dependent neoplasia is usually found in women older than 40 years. Thus, it is seen only rarely with pregnancy. Hannuna and colleagues (2009) found 27 cases that were identified during pregnancy or within the first 4 months postpartum. Most were found in first-trimester curettage specimens.
These are usually early-stage well-differentiated adenocarcinomas for which treatment consists primarily of total abdominal hysterectomy and bilateral salpingo-oophorectomy. Much less commonly, to preserve future fertility, curettage with or without postprocedural progestational therapy has been used for the rare patient with cancer identified in a miscarriage curettage specimen (Schammel, 1998).
Many more studies describe a conservative approach for well-selected nonpregnant women diagnosed with endometrial cancer who wish to preserve fertility. In one study of 13 women treated with progestins for early-stage well-differentiated adenocarcinoma, nine had liveborn infants, and four of six women with a recurrence responded to another course of therapy (Gotlieb, 2003). Similar outcomes were described in 12 women by Niwa and associates (2005) and in 21 women by Signorelli and coworkers (2009). Despite these acceptable pregnancy rates, recurrences and death have been reported, and conservative management is not considered standard (Erkanli, 2010).
 Ovary
Ovary
Ovarian masses found during pregnancy are relatively common, and the reported incidences are population- and investigation- dependent. Factors include whether studies are referral or primary care sources, the frequency of prenatal sonography, and what size constitutes a “mass.” It is thus not surprising that the incidence has been reported as 1 in 100 to 1 in 2000 pregnancies (Whitecar, 1999; Zanetta, 2003). Of ovarian malignancies, the absolute incidence in the California Cancer Registry was 1 in 19,000 pregnancies (Smith, 2003).
The most frequent types of ovarian masses are corpus luteum cysts, endometriomas, benign cystadenomas, and mature cystic teratomas, colloquially termed dermoids. Because pregnant women are usually young, malignant tumors and those of low malignant potential are proportionately uncommon and vary from 4 to 13 percent (Hoffman, 2007; Sherard, 2003). Our experiences from Parkland Hospital are similar to those reported by Leiserowitz and colleagues (2006), who found that 1 percent of 9375 ovarian masses were frankly malignant and that another 1 percent was of low malignant potential.
Most ovarian masses are asymptomatic in pregnant women. Some cause pressure or chronic pain, and acute abdominal pain may be due to torsion, rupture, or hemorrhage. Seldom is blood loss significant enough to cause hypovolemia.
Sonography
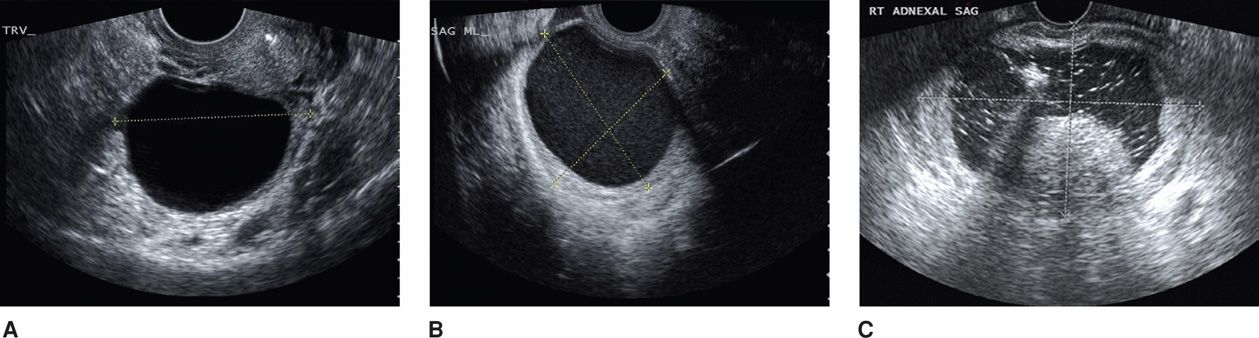
Many ovarian masses are detected during routine prenatal sonography or during imaging done for other indications, including evaluation of symptoms. The typical sonographic appearance of these masses is shown in Figure 63-6. In some instances, MR imaging can be used to evaluate complicated anatomy (American College of Obstetricians and Gynecologists, 2013b).
FIGURE 63-6 Sonographic characteristics of common adnexal masses in pregnancy. A. A simple anechoic cyst with smooth walls is characteristic of a physiological corpus luteum cyst or benign cystadenoma. B. Cystic structure with diffuse internal low-level echoes suggestive of an endometrioma or hemorrhagic corpus luteum. C. Mature cystic teratoma appears as an adnexal cyst (marked by calipers) with accentuated lines and dots that represent hair in both longitudinal and transverse planes. At the central inferior aspect of this cyst, a mural nodule—Rokitansky protuberance—is seen. These typically rounded protuberances range in size from 1 to 4 cm, they are predominantly hyperechoic, and they create an acute angle with the cyst wall. Although not seen here, fat-fluid levels are often identified with cystic teratomas. (Image contributed by Dr. Elysia Moschos.)
Tumor Markers
Cancer antigen 125 (CA125) levels are frequently elevated with ovarian malignancy. Importantly, concentrations in early pregnancy and early puerperium are normally elevated, possibly from the decidua (Aslam, 2000; Haga, 1986; Spitzer, 1998). From the second trimester until term, levels are not normally higher than those in the nonpregnant woman (Appendix, p. 1289). Other tumor markers that have not been proven for diagnosis or posttreatment surveillance in pregnancy include OVA1, human chorionic gonadotropin (hCG), alpha-fetoprotein, and inhibins A and B (Liu, 2011). Of these, OVA1 is a biomarker blood test that measures five analytes, one of which is CA125.
Complications
The two most common complications of any ovarian mass are torsion and hemorrhage. Torsion usually causes acute constant or episodic lower abdominal pain that frequently is accompanied by nausea and vomiting. Sonography often aids the diagnosis. With color Doppler, presence of an ovarian mass with absent flow strongly correlates with torsion. However, minimal or early twisting may compromise only venous flow, thus leaving arterial supply intact. If torsion is suspected, laparoscopy or laparotomy is warranted. Contrary to prior teaching, adnexectomy is generally unnecessary to avoid clot release, thus, most recommend attempts at untwisting (McGovern, 1999; Zweizig, 1993). With a salvageable ovary, within minutes, congestion is relieved, and ovarian volume and cyanosis diminish. If cyanosis persists, however, then removal of the infarcted adnexa is typically indicated.
If the adnexa is healthy, there are options. First, neoplasms are resected. However, ovarian cystectomy in an ischemic, edematous ovary may be technically difficult, and adnexectomy may be necessary. Second, unilateral or bilateral oophoropexy has been described to minimize the risk of repeated torsion (Djavadian, 2004; Germain, 1996). Techniques described include shortening of the uteroovarian ligament or fixing the uteroovarian ligament to the posterior uterus, the lateral pelvic wall, or the round ligament (Fuchs, 2010; Weitzman, 2008).
The most common case of ovarian hemorrhage follows rupture of a corpus luteum cyst. If the diagnosis is certain and symptoms abate, then observation and surveillance is usually sufficient. Concern for ongoing bleeding will typically prompt surgical evaluation. If the corpus luteum is removed before 10 weeks’ gestation, progestational support is recommended to maintain the pregnancy. Suitable regimens include: (1) micronized progesterone (Prometrium) 200 or 300 mg orally once daily; (2) 8-percent progesterone vaginal gel (Crinone) one premeasured applicator vaginally daily plus micronized progesterone 100 or 200 mg orally once daily; or (3) intramuscular 17-hydroxyprogesterone caproate, 150 mg. For the last, if given between 8 and 10 weeks’ gestation, only one injection is required immediately after surgery. If the corpus luteum is excised between 6 and 8 weeks’ gestation, then two additional doses should be given 1 and 2 weeks after the first.
Management of an Asymptomatic Adnexal Mass
Because most of these are incidental findings, management considerations include whether resection is necessary and its timing. A cystic benign-appearing mass that is < 5 cm often requires no additional antepartum surveillance. Early in pregnancy, this is likely a corpus luteum cyst, which typically resolves by the early second trimester. For cysts ≥ 10 cm, because of the substantial risk of malignancy, torsion, or labor obstruction, surgical removal is reasonable. Tumors between 5 and 10 cm should be carefully evaluated by sonography along with color Doppler and possibly MR imaging. If they have a simple cystic appearance, these cysts can be managed expectantly with sonographic surveillance (Schmeler, 2005; Zanetta, 2003). Resection is done if cysts grow, begin to display malignant qualities, or becomes symptomatic. However, if there are sonographic characteristics that suggest a cancer—thick septa, nodules, papillary excrescences, or solid components—then immediate resection is indicated (Caspi, 2000; Fleischer, 1990). Approximately 1 in 1000 pregnant women undergoes surgical exploration for an adnexal mass (Boulay, 1998). In general, we plan resection at 14 to 20 weeks because most masses that will regress will have done so by this time. Those with classic findings of endometrioma or mature cystic teratoma may be resected postpartum or during cesarean for obstetrical indications. Importantly, in any instance in which cancer is strongly suspected, the American College of Obstetricians and Gynecologists (2011) recommends consultation with a gynecologic oncologist.
Pregnancy-Related Ovarian Tumors
A group of adnexal masses result directly from the stimulating effects of various pregnancy hormones on ovarian stroma. These include pregnancy luteoma, hyperreactio luteinalis, and ovarian hyperstimulation syndrome.
Pregnancy Luteoma. These are rare, benign, ovarian neoplasms that are thought to arise from luteinized stromal cells and that classically cause elevated testosterone levels (Irving, 2011). Up to 25 percent of affected women will be virilized, and of these affected women, approximately half of their female fetuses will have some degree of virilization. Fortunately, most mothers and their fetuses are unaffected by the hyperandrogenemia because the placenta rapidly converts androgens to estrogens (Kaňová, 2011).
In some cases, an adnexal mass along with maternal virilization will prompt sonography and measurement of testosterone and CA125 levels. Luteomas range in size from microscopic to greater than 20 cm. They appear as solid tumors, may be multiple or bilateral, and may be complex because of hemorrhagic areas (Choi, 2000). Concerns for malignancy may be further investigated by MR imaging (Kao, 2005; Tannus, 2009).
Total testosterone levels are increased, but with the caveat that levels in normal pregnancy can be substantially elevated (Appendix, p. 1291). Levels of CA125 antigen tumor marker are normal with a pregnancy luteoma, and significantly elevated levels indicate another cause. Differential diagnoses are primary ovarian cancer or metastatic disease and include granulosa cell tumors, thecomas, Sertoli-Leydig cell tumors, Leydig cell tumors, stromal hyperthecosis, and hyperreactio luteinalis.
Generally, luteomas do not require surgical intervention unless there is torsion, rupture, or hemorrhage (Masarie, 2010). These tumors spontaneously regress during the first few months postpartum, and androgen levels drop precipitously during the first 2 weeks following delivery (Wang, 2005). Lactation may be delayed a week or so by hyperandrogenemia (Clement, 1993; Dahl, 2008). Recurrence in subsequent pregnancies is rare.
Hyperreactio Luteinalis. In this condition, one or both ovaries develop multiple large theca-lutein cysts, typically after the first trimester. Cysts are caused by luteinization of the follicular theca interna layer, and most are in response to stimulation by exceptionally high hCG levels (Russell, 2009). For this reason, they are more common with gestational trophoblastic disease, twins, fetal hydrops, and other conditions with increased placental mass. Similar to luteoma, maternal virilization develops in approximately a third of women, but there have been no reports of fetal virilization (Kaňová, 2011).
These ovarian tumors appear sonographically to have a “spoke wheel” pattern (Fig. 20-3, p. 399). If the diagnosis is confident, and unless complicated by torsion or hemorrhage, surgical intervention is not required. These masses resolve after delivery.
Ovarian Hyperstimulation Syndrome. This rare event is typified by multiple ovarian follicular cysts accompanied by increased capillary permeability. It most often is a complication of ovulation-induction therapy for infertility, although it rarely may develop in an otherwise normal pregnancy. Its etiopathogenesis is thought to involve hCG stimulation of vascular endothelial growth factor (VEGF) expression in granulosa-lutein cells (Soares, 2008). This causes increased vascular permeability that can lead to hypovolemia with ascites, pleural or pericardial effusion, acute kidney injury, and hypercoagulability. There is a clinical spectrum with symptoms that may include abdominal distention, rapid weight gain, dyspnea, and progressive hypovolemia. Serious complications are renal dysfunction, adult respiratory distress syndrome, ovarian rupture with hemorrhage, and thromboembolism.
Although most cases of ovarian hyperstimulation are associated with in vitro fertilization, increasing hCG levels from pregnancy can worsen the syndrome (Chen, 2012). Detailed guidelines for management have been outlined by the American Society for Reproductive Medicine (2008). Treatment is primarily supportive with attention to maintaining vascular volume and thromboprophylaxis.
Ovarian Cancer
Malignancies of the ovary are most common in older women and are the leading cause of death in all women from genital-tract cancers (American Cancer Society, 2012). That said, the incidence of ovarian malignancy during pregnancy ranges from 1 in 20,000 to 50,000 births (Palmer, 2009; Eibye, 2013; Smith, 2003). Fortunately, 75 percent of these found in pregnancy are early-stage malignancies with a 5-year survival rate between 70 and 90 percent (Brewer, 2011). The types of malignancy are also markedly different in pregnant women compared with those in older women. In pregnant women, these are, in decreasing order of frequency, germ cell and sex cord-stromal tumors, low malignant potential tumors, and last, epithelial tumors (Morice, 2012).
Pregnancy apparently does not alter the prognosis of most ovarian malignancies. Management is similar to that for nonpregnant women, with the usual proviso that it may be modified depending on gestational age. Thus, if frozen section histopathological analysis verifies malignancy, then surgical staging is done with careful inspection of all accessible peritoneal and visceral surfaces (Giuntoli, 2006; Yazigi, 1988). Peritoneal washings are taken for cytology; biopsies are taken from the diaphragmatic surface and peritoneum; omentectomy is done; and biopsies are taken from pelvic and infrarenal paraaortic lymph nodes. Depending on the uterine size, some of these components, especially lymphadenectomy, may not be technically feasible.
If there is advanced disease, bilateral adnexectomy and omentectomy will decrease most tumor burden. In early pregnancy, hysterectomy and aggressive surgical debulking procedures may be elected. In other cases, minimal debulking as described is done and the operation terminated. In some cases of aggressive or large-volume disease, chemotherapy can be given during pregnancy while awaiting pulmonary maturation. As discussed on page 1227, monitoring maternal CA125 serum levels during chemotherapy is not accurate in pregnancy (Aslam, 2000; Morice, 2012).
Adnexal Cysts
Paratubal and paroovarian cysts are either distended remnants of the paramesonephric ducts or are mesothelial inclusion cysts. Although most are ≤ 3 cm, they occasionally attain worrisome dimensions (Genadry, 1977). Their reported incidence is influenced by size, but one autopsy series in nonpregnant women cited this to be 5 percent (Dorum, 2005). The most common paramesonephric cyst is the hydatid of Morgagni, which is pedunculated and typically dangles from one of the fimbria. These cysts infrequently cause complications and are most commonly identified at the time of cesarean delivery or puerperal sterilization, at which time they can simply be drained or excised. Neoplastic paraovarian cysts are rare, histologically resemble tumors of ovarian origin, and rarely are of borderline potential or frankly malignant (Honore, 1980; Korbin, 1998).
 Vulva and Vagina
Vulva and Vagina
Preinvasive Disease
In young women, vulvar intraepithelial neoplasia (VIN) and vaginal intraepithelial neoplasia (VAIN) are seen more often than invasive disease and are commonly associated with HPV infection. As with cervical neoplasia, these premalignant conditions are treated after the puerperium.
Squamous Cell Carcinoma
Cancer of the vulva or vagina is generally a malignancy of older women, and thus, these are rarely associated with pregnancy. Even so, any suspicious lesions should be biopsied. Treatment is individualized according to the clinical stage and depth of invasion. In a review of 23 cases, investigators concluded that radical surgery for stage I disease was feasible during pregnancy—including in the last trimester (Heller, 2000). We and others question the necessity of resection in late pregnancy in that definitive therapy can often be delayed because of vulvar cancer’s typical slow progression (Anderson, 2001). It appears that vaginal delivery is not contraindicated if vulvar and inguinal incisions are well healed.
Vulvar sarcoma, vulvar melanoma, and vaginal malignancies are rare in pregnancy and are the subjects of case reports (Alexander, 2004; Kuller, 1990; Matsuo, 2009).
BREAST CARCINOMA
The incidence of breast cancer is age dependent, however, because of its overall high frequency in women, it is relatively common in young women. Recall from Figure 63-1 that breast malignancies are the most frequent cancer found in pregnant women. And, as more women choose to delay childbearing, the frequency of associated breast cancer is certain to increase (Amant, 2012a). As an example, postponed childbearing was considered partially responsible for the increase in pregnancy-associated breast cancer in Sweden from 16 to 37 percent during the 40-year period from 1963 to 2002 (Andersson, 2009). Similar findings have been reported from Denmark (Eibye, 2013).
 Risk Factors
Risk Factors
Some, but not all, studies suggest that women with a family history of breast cancer—especially those with BRCA1 and BRCA2 breast cancer gene mutations—are more likely to develop breast malignancy during pregnancy (Johannsson, 1998; Shen, 1999; Wohlfahrt, 2002). However, although there is a definitely increased long-term risk with BRCA1 and BRCA2 mutations, it may be that parity modifies this risk. Specifically, parous women older than 40 years with these mutations have a significantly lower cancer risk than nulliparas with these mutations (Andrieu, 2006; Antoniou, 2006). Women with BRCA1 and BRCA2 gene mutations who undergo induced abortion or those who breast feed do not have an increased risk of breast cancer (Beral, 2004; Friedman, 2006). Moreover, Jernström and associates (2004) found that breast feeding actually conveyed a protective effect against this cancer in those with BRCA1 gene mutation, but not in those with BRCA2 mutations. Of other congenital risks, it is controversial whether diethylstilbestrol exposure in utero is associated with an increased breast cancer risk (Hoover, 2011; Larson, 2006; Titus-Ernstoff, 2006).
 Diagnosis
Diagnosis
More than 90 percent of pregnant women with breast cancer have a palpable mass, and greater than 80 percent are self-reported (Brewer, 2011). There are usually slight delays in clinical assessment, diagnostic procedures, and treatment of pregnant women with breast tumors (Berry, 1999). This can partially be attributed to increased pregnancy-induced breast tissue that obscures masses.
Evaluation of pregnant women with a breast mass should not differ from that for nonpregnant women. Thus, any suspicious breast mass should be pursued to diagnosis. Pragmatically, a palpable discrete mass can be biopsied or excised. If imaging is desirable to distinguish between a solid mass and a cystic lesion, then sonography has high sensitivity and specificity (Navrozoglou, 2008). Mammography is appropriate if indicated, and the fetal radiation risk is negligible with appropriate shielding. Fetal exposure is only 0.04 mGy for the typical mammogram study. But, because breast tissue is denser in pregnancy, mammography has a false-negative rate of 35 to 40 percent (Woo, 2003). If the decision to biopsy is uncertain, then MR imaging may be used (Amant, 2010). With such techniques, masses can usually be described as either solid or cystic.
Cystic breast lesions are simple, complicated, or complex (Berg, 2003). Simple cysts do not require special management or monitoring, but they may be aspirated if symptomatic. Complicated cysts show internal echoes during sonography, and they sometimes are indistinguishable from solid masses. These are typically aspirated, and if the sonographic abnormality does not resolve completely, a core-needle biopsy is usually performed. Complex cysts have septa or intracystic masses seen on sonographic evaluation. Because some forms of breast cancer may be complex cysts, excision is usually recommended.
For solid breast masses, evaluation is with the triple test, that is, clinical examination, imaging, and needle biopsy. If all three suggest a benign lesion or if all three suggest a breast cancer, the test is said to be concordant. A concordant benign triple test is > 99-percent accurate, and breast lumps in this category can be followed by clinical examination alone. Fortunately, most masses in pregnancy have these three reassuring features. To the contrary, however, if any of the three assessments is suggestive of malignancy, then the mass should be excised regardless of the other two findings.
 Management
Management
Once breast cancer is diagnosed, a limited search of the most common metastatic sites is completed. For most women, this includes a chest radiograph, liver sonography, and skeletal MR imaging (Amant, 2012a).
Treatment of breast cancer is by a multidisciplinary team that includes an obstetrician, surgeon, and medical oncologist. Desires for pregnancy continuation are addressed up front. Data indicate that pregnancy interruption does not influence the course or prognosis of breast cancer (Cardonick, 2010). With pregnancy continuation, treatment in general is similar to that for nonpregnant women. However, chemotherapy and surgery are delayed to the second trimester, and adjuvant radiotherapy is withheld until after delivery (Brewer, 2011).
Surgical treatment may be definitive. In the absence of metastatic disease, either a wide excision or a modified or total mastectomy—each with axillary node staging—can be performed (Rosenkranz, 2006; Woo, 2003). Staging by sentinel lymph node biopsy and lymphoscintigraphy with technetium-99m is safe. Although often administered concurrently with lymphoscintigraphy, isosulfan blue and methylene blue dyes are pregnancy class C drugs, and some recommend that they be avoided (Mondi, 2007; Spanheimer, 2009). Breast reconstruction, if desired, is typically delayed until after delivery (Viswanathan, 2011).
Chemotherapy is usually given with either positive- or negative-node breast cancers. In premenopausal women, survival is improved even if lymph nodes are cancer free. For node-positive disease, multiagent chemotherapy is begun if delivery is not anticipated within several weeks. Drug classes are alkylating agents, anthracyclines, antimetabolites, and taxanes (Lippman, 2012). Cyclophosphamide, doxorubicin, methotrexate, and 5-fluorouracil are currently in use (García-Manero, 2009). After the first trimester, methotrexate can be substituted for doxorubicin. If an anthracycline-based agent such as doxorubicin is used, pretherapy maternal echocardiography is performed because of associated cardiotoxicity (Brewer, 2011). In 22 pregnant women treated with modified radical mastectomy that was followed by chemotherapy in most, Berry and colleagues (1999) reported minimal fetal risks. Moreover, Hahn and coworkers (2006) reported good short-term outcomes for the offspring of 57 women treated during pregnancy with multiagent chemotherapy for breast cancer.
Immunotherapy for breast cancers has become commonplace. Trastuzumab (Herceptin) is a monoclonal antibody to the HER2/neu receptor, which is found in approximately a third of invasive breast cancers (Hudis, 2007). The drug is used more commonly as adjunctive therapy for early disease in HER2/neu-positive tumors but is not recommended in pregnancy (Amant, 2010). This is because HER2/neu is strongly expressed in fetal renal epithelium, and trastuzumab has been linked with miscarriage, with fetal renal failure and related oligohydramnios, and with neonatal deaths from preterm birth (Azim, 2010). For these reasons, highly effective contraception is used during and for 6 months following trastuzumab treatment. For women who conceive after only brief exposure, fetal renal effects appear to be reversible, and short-term neonatal outcomes have been normal with no congenital anomalies reported (Azim, 2012).
 Prognosis
Prognosis
The effects of pregnancy on the course of breast cancer and its prognosis are complex. There is no doubt that breast cancer is more aggressive in younger women, but whether it is more aggressive during pregnancy in these same women is debatable. Clinically, most studies indicate little difference in overall survival rates with pregnancy-associated breast cancer compared with similarly aged and staged nonpregnant women (Beadle, 2009). There have been other reports of worse overall survival rates with pregnancy-associated breast cancer (Rodriguez, 2008). These investigators did both conclude, however, that later disease stages are more prevalent in pregnant women.
Thus, it appears that because breast cancer is usually found at a more advanced stage in pregnant women, overall prognosis is diminished. In an earlier review, Jacob and Stringer (1990) reported that approximately 30 percent of pregnant women had stage I disease, 30 percent had stage II, and 40 percent had stages III or IV. In addition, the aggregate of studies published after 1990 indicate that up to 60 percent of pregnant women have concomitant axillary node involvement at diagnosis. And although, stage for stage, the 5-year survival rate is comparable in pregnant and nonpregnant women, the more advanced stages that are typical of pregnant women worsen their prognosis (King, 1985; Nugent, 1985; Zemlickis, 1992).
 Pregnancy Following Breast Cancer
Pregnancy Following Breast Cancer
After breast cancer treatment, chemotherapy will render some women infertile, and options for childbearing are limited (Kim, 2011). For those who became pregnant, there appear to be no adverse effects on long-term maternal survival rates (Averette, 1999; Velentgas, 1999). Moreover, a metaanalysis of 10 studies found that for women with early breast cancer, pregnancy that occurs 10 months after diagnosis may, in fact, have a survival benefit (Valachis, 2010). There are also no data indicating that lactation adversely affects the course of previously diagnosed breast cancer. Also, lactation and breast feeding are possible after conservative surgery and radiation of the treated breast (Higgins, 1994).
Recommendations for subsequent pregnancies in women successfully treated for breast malignancy are based on several factors that include consideration for the recurrence risk. Simply because recurrences are more common soon after treatment, it seems reasonable to advise a delay of 2 to 3 years for observation. Hormonal contraceptive methods are contraindicated, and a copper-containing intrauterine device is an excellent long-acting reversible method for many. Even with contraceptive failure, women who conceive do not appear to have diminished survival (Ives, 2006). It is reassuring that women who undertake pregnancy after breast cancer treatment overall have good outcomes (Langagergaard, 2006). Of particular concern is that women treated with tamoxifen are at risk for several months after its discontinuation for having an infant with congenital anomalies. This is because of its extremely long half-life, and thus delaying conception is recommended for at least 2 months after its completion (Braems, 2011).
THYROID CANCER
Various thyroid gland malignancies constitute the most common endocrine cancers. Because most of these originate within discrete nodules, their evaluation is important. This typically includes sonography and measurement of serum thyroid-stimulating hormone (TSH) and free thyroxine levels. Fine-needle aspiration is indicated for any suspicious nodule (Yazbeck, 2012).
With a diagnosis of thyroid malignancy, pregnancy termination is not necessary (Alves, 2011). Primary therapy is total thyroidectomy performed ideally during the second trimester. Postoperatively, replacement thyroxine is given to maintain serum TSH levels between 0.1 and 0.5 mIU/L (Abalovich, 2007). Most thyroid cancers are well differentiated and follow an indolent course. Thus, delayed surgical treatment does not usually alter outcome (Yazbeck, 2012). If surgical therapy is postponed until after pregnancy, thyroxine is given to suppress any growth stimulus of TSH on tumor cells (Pacini, 2012).
In some types of thyroid malignancies, primary or postoperative treatment is given with radioiodine. This is contraindicated in both pregnancy and lactation. Transplacental iodine-131 is avidly trapped by the fetal thyroid gland to cause hypothyroidism (Chap. 58, p. 1149). During lactation, the breast also concentrates a substantial amount of iodide. This may pose neonatal risk due to 131I-containing milk ingestion and maternal risk from significant breast irradiation. To limit maternal exposure, a delay of 3 months between lactation and ablation will more reliably ensure complete breast involution (Sisson, 2011). Last, pregnancy should be avoided for 6 months to 1 year in women with thyroid cancer who receive 131I doses. This time ensures thyroid function stability and confirms thyroid cancer remission (Abalovich, 2007).
LYMPHOID CELL MALIGNANCIES
Hematological malignancies comprise 10 to 20 percent of cancers in pregnancy (Cohen, 2011; Eibye, 2013; Smith, 2003). Some of these present as leukemia involving bone marrow and blood. Others are solid lymphomas, which may be of B- or T-cell origin.
 Hodgkin Disease
Hodgkin Disease
This lymphoma is probably B-cell derived and is cytologically distinguished from other lymphomas by Reed–Sternberg cells. It is the most common malignant lymphoma in women of childbearing age. In a population-based review of approximately 4 million pregnancies, Smith and associates (2003) reported that Hodgkin lymphoma complicated only 1 in 34,000 live births. Our experiences are similar during the past 40 years at Parkland Hospital with nearly 500,000 births.
In more than 70 percent of Hodgkin disease cases, there is painless enlargement of lymph nodes above the diaphragm—the axillary, cervical, or submandibular chains. Approximately one third of patients have symptoms including fever, night sweats, malaise, weight loss, and pruritus. Diagnosis is by histological examination of involved nodes (Longo, 2012).
Staging and Treatment
The Ann Arbor staging system, shown in Table 63-2, is used for Hodgkin and other lymphomas. Recent studies have used risk categories—high, low, and very low—however, these are not succinctly defined (Armitage, 2010). Pregnancy limits the use of some radiographic studies for staging, but at minimum, chest radiography, abdominal imaging with sonography or MR imaging, and bone marrow biopsy are completed (Williams, 2001). MR imaging is excellent for evaluating thoracic and abdominal paraaortic lymph nodes (Fig. 63-7) (Brenner, 2012).
TABLE 63-2. Ann Arbor Staging System for Hodgkin and Other Lymphomas
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree