Neoplasia
Robert J. Arceci
Howard J. Weinstein
Although neoplasia in infancy is quite rare, it presents important and unique biologic, diagnostic, and therapeutic problems. Such neoplasms often show peculiarities that distinguish them from those occurring later in life. Many tumors in early life are composed of persistent embryonal or fetal tissues, suggesting a failure of proper maturation or cytodifferentiation during intrauterine or early postnatal life. The failure of proper maturation of fetal tissue may be sometimes difficult to distinguish from neoplasia. Additionally, an unexpectedly large number of neoplasms of early life are associated with growth disturbances and congenital anomalies. Spontaneous regression and cytodifferentiation also occur most frequently in tumors of early life.
The unique physiology of the developing neonate provides the clinician with special problems in terms of therapeutic interventions and their long-term sequelae. The neonatal period is also one in which different types of treatment and exposures may affect the long-term risk of secondary malignancies.
EPIDEMIOLOGY
From the data in The Third National Cancer Survey (1969 to 1971), Bader and Miller (1) reported that, in the United States, the annual incidence of malignant neoplasms in infants younger than 1 year of age was 183.4 per 1 million live births and within the first 28 days of life was 36.5 per 1 million live births. They further estimated that approximately 653 infants per year in the United States are diagnosed with cancer and that about 130 (20%) of these patients are neonates. In a later study from Denmark, the incidence of neonatal cancer was calculated to be between 1.88 to 2.98 cases per 100,000 births (2). Approximately one-half of the neonatal malignancies are noted on the first day of life (3). The incidence of childhood cancers also has been observed to show regional differences over time and, over the past 20 years, an absolute increase in childhood cancer has been reported (4,5).
When incidence for all malignancies is compared with mortality as determined from death certificates, the incidence in patients younger than 1 year is 3.5 times greater than mortality, whereas the incidence in patients younger than 29 days old is 4.8 times greater than mortality (1,6). These figures offer an interesting comparison to those reported in children up to 15 years of age, among whom the incidence of malignancy is only about 1.3 to 1.8 times greater than mortality. When individual diseases are considered, there are marked differences in incidence vs. mortality. For example, in neonates, the incidence of neuroblastoma is 10 times greater than the mortality, whereas the incidence of leukemia is only 1.8 times the mortality. The distribution of the types of malignancies found in infants younger than 1 year of age differs from that found in later childhood. In infants younger than 1 year of age, neuroblastoma is the most common malignancy and accounts for about 50% of malignancies in the neonatal period; it is followed by leukemia, renal tumors, sarcomas, central nervous system (CNS) tumors, and hepatic malignancy (2,3,7,8). However, when one considers the total spectrum of neoplastic disorders of infancy, teratoma is usually reported as the most frequently encountered neoplasm, followed by hemangiomas, lymphangiomas, and small nevi lesions (9,10,11,12). In older children younger than 15 years of age, leukemia is the most common malignancy (about 30%), followed by CNS tumors, lymphoma, neuroblastoma, sarcoma, and renal tumors. Thus, the incidence and types of neoplastic disorders of infancy contrast greatly compared to later childhood and define the neonatal period as epidemiologically distinct in terms of these disorders.
DEVELOPMENTAL GROWTH DISTURBANCES, GENETIC ABERRATIONS, AND CANCER PATHOGENESIS
Primary, inherited, cytogenetic syndromes usually occur as a result of chromosomal aneuploidy, deletions, translocations, increased fragility or altered epigenetic imprinting, which in turn represent the end result of germline chromosomal defects. An example of aneuploidy is Down syndrome (trisomy 21), in which the frequency of acute leukemia is approximately 15 times the normal. Additionally, an increased incidence of solid tumors has been reported for persons with trisomy 8, 9, 13, and 18 (13). Deletion of part
of the long arm of chromosome 13 is associated with psychomotor retardation, microcephaly, cardiac and skeletal defects, and the early development of retinoblastoma. The deletion of the short arm of chromosome 11 results in mental retardation, microcephaly, aniridia, ear and genital anomalies, and an increased incidence of Wilms tumor (WAGR syndrome). These syndromes provided support for the assignment of a retinoblastoma locus to chromosome 13q14 and a Wilms tumor locus (WT1) to 11p13. These mutant genes are heterozygous in constitutional cells and homozygous in retinoblastoma and Wilms tumor cells. WAGR syndrome results from loss of several genes from the 11p13 region. Deletion of one copy of PAX 6 is responsible for aniridia, and loss of one WT1 allele results in genitourinary anomalies. Deletion of the WT1 gene in patients with WAGR syndrome is thought to be the first “genetic hit” in the genesis of a Wilms tumor. The function of WT1 is not clear, but it is an important transcription factor that contributes to the regulation of cell proliferation and differentiation. Homozygosity at the “Wilms tumor locus” on chromosome 11 has also been found in embryonal rhab- domyosarcomas (RMS) and hepatoblastomas, suggesting a common pathogenesis for these embryonal tumors (14). The specific loss of constitutional heterozygosity and its relationship to oncogenesis has been confirmed in studies of transgenic mice that lack a functional tumor suppressor gene, p53. This phenotype, termed Li-Fraumeni Syndrome in humans, is characterized by a higher incidence of embryopathy, and an increased incidence of malignancies developing early in life (15).
of the long arm of chromosome 13 is associated with psychomotor retardation, microcephaly, cardiac and skeletal defects, and the early development of retinoblastoma. The deletion of the short arm of chromosome 11 results in mental retardation, microcephaly, aniridia, ear and genital anomalies, and an increased incidence of Wilms tumor (WAGR syndrome). These syndromes provided support for the assignment of a retinoblastoma locus to chromosome 13q14 and a Wilms tumor locus (WT1) to 11p13. These mutant genes are heterozygous in constitutional cells and homozygous in retinoblastoma and Wilms tumor cells. WAGR syndrome results from loss of several genes from the 11p13 region. Deletion of one copy of PAX 6 is responsible for aniridia, and loss of one WT1 allele results in genitourinary anomalies. Deletion of the WT1 gene in patients with WAGR syndrome is thought to be the first “genetic hit” in the genesis of a Wilms tumor. The function of WT1 is not clear, but it is an important transcription factor that contributes to the regulation of cell proliferation and differentiation. Homozygosity at the “Wilms tumor locus” on chromosome 11 has also been found in embryonal rhab- domyosarcomas (RMS) and hepatoblastomas, suggesting a common pathogenesis for these embryonal tumors (14). The specific loss of constitutional heterozygosity and its relationship to oncogenesis has been confirmed in studies of transgenic mice that lack a functional tumor suppressor gene, p53. This phenotype, termed Li-Fraumeni Syndrome in humans, is characterized by a higher incidence of embryopathy, and an increased incidence of malignancies developing early in life (15).
A number of inherited syndromes, including Bloom syndrome, Fanconi Anemia, ataxia-telangiectasia, xeroderma pigmentosum, and Werner’s syndrome, are characterized by developmental abnormalities and increased incidence of various types of cancer (16,17). These syndromes also demonstrate increased defects in DNA recombination, increased sensitivity to genotoxic agents, increased chromosomal fragility, and abnormal DNA repair (18). Of particular interest is that these syndromes now are known to be caused by defects in genes encoding several novel proteins involved in deoxyribonucleic acid (DNA)/ribonucleic acid (RNA) recombination and repair, such as the DEAD-box helicase of Bloom syndrome or excision repair enzymes associated with some of the complementation groups of xeroderma pigmentosum (18,19,20). It is intriguing that when these defective genes are inherited through the germline, patients show both developmental abnormalities and an increased incidence of cancer. It will be important to determine whether somatic cell mutations of such genes will increase the chances of a cell becoming malignant, thus showing how the identification of altered genes leading to developmental defects can also increase our understanding of cancer in older individuals.
Malformations (21) and malformation syndromes without obvious cytogenetic abnormalities include hemihypertrophy and Beckwith-Wiedemann syndrome (BWS), which consists of mental retardation, gigantism, macroglossia, omphalocele, and organomegaly; both of these disorders are associated with the development of Wilms tumor, hepatoblastoma, and adrenocortical carcinoma. BWS, which occurs in approximately 1 in 13,000 births, usually is sporadic, although an autosomal dominant inheritance pattern with incomplete penetrance also has been proposed. Patients with BWS have an approximately 7.5% to 10% risk of developing a tumor (22). Sacrococcygeal teratomas and teratocarcinomas are associated with anomalies of the lower spine and urogenital region.
Hamartomas are benign proliferations of cells in their normal anatomic location. Hamartomas in which malignant neoplasms arise include congenital melanotic nevi, which can progress to melanoma, and familial polyposis, which may evolve into colonic carcinoma. Examples of malignancies developing from persistent fetal rests include craniopharyngioma arising from tissue derived embryologically from the Rathke pouch, and persistent neuroblastic cellularity leading to adrenal neuroblastoma.
Naturally occurring DNA sequences homologous to transforming viral oncogenes exist in normal, untransformed cells of all metazoa. Such DNA sequences are called cellular oncogenes and are used in normal cells during growth, development, and differentiation in precise temporal and tissue-specific patterns (23,24). Some of their products function as potent cell growth and death (apoptosis) regulators. The protein products of some cellular oncogenes are quite similar to the products from the homologous viral oncogenes. Because of their expression and critical role during normal development, inherited or acquired mutations affecting cellular oncogene expression can lead to a variety of developmental abnormalities and congenital defects, such as hemihypertrophy syndromes and hamartomas. Additionally, the persistent expression beyond birth of certain growth-related oncogenes may play a role in such proliferative states as the transient myeloproliferative disorder associated with Down syndrome and stage IV-S neuroblastoma found in infants, which both are characterized by subsequent, spontaneous regression.
These predisposing conditions share at least one common element: an inherited or developmental disturbance of cellular growth and/or cell survival, which may be linked to the molecular pathways regulating these genetically determined cellular responses. The finding of these different classes of genes helps to define the molecular links between conditions of abnormal development (i.e., teratogenesis) and neoplastic transformation (25,26).
EXPOSURE TO MATERNAL MALIGNANCY
In addition to the susceptibility of the fetus to adverse effects of chemotherapy during pregnancy, there also is the possibility that the maternal cancer will metastasize to the placenta and fetus. Although many anecdotal reports have documented such involvement, it occurs only very rarely. The types of tumors shown to be transmitted from the
mother to the placenta or fetus are quite varied, with melanoma most commonly cited. Although lymphoma and leukemia may involve the placenta, they have usually not been found to be transmitted to the fetus (27,28,29,30,31). The evaluation of infants born to mothers with cancer has not been clearly established, in part because of the rarity of such events. However, recommendations would suggest the utility of close follow-up during the first year of life including physical exams, blood studies such as complete blood count (CBC) and liver function tests, and scans only when indicated. Careful examination of the placenta is an important component of this evaluation.
mother to the placenta or fetus are quite varied, with melanoma most commonly cited. Although lymphoma and leukemia may involve the placenta, they have usually not been found to be transmitted to the fetus (27,28,29,30,31). The evaluation of infants born to mothers with cancer has not been clearly established, in part because of the rarity of such events. However, recommendations would suggest the utility of close follow-up during the first year of life including physical exams, blood studies such as complete blood count (CBC) and liver function tests, and scans only when indicated. Careful examination of the placenta is an important component of this evaluation.
PRENATAL EXPOSURE TO MATERNAL GENOTOXINS
There are relatively few reports and/or studies of outcome in infants born to mothers undergoing chemotherapy and/or radiation therapy for cancer. Nevertheless, several important lessons can be learned from the available reports. The risk of developmental problems increases with decreased gestational age at the time of maternal treatment (32,33,34). For example, a recommendation to terminate a pregnancy is often reasonable when significant numbers and doses of anticancer drugs are used during the first trimester because of the increased risk of major birth defects and spontaneous abortions. Outcomes for infants whose mothers are treated during the second and third trimesters are significantly better, although there has been reported some risk of low birth weight, intrauterine growth retardation and stillbirths (32,33,34). Last, other prenatal drug exposures may lead to increased risk of cancer in offspring. For example, prenatal expsosure to diethyl stilbesterol has been closely linked to the development of clear cell adenocarcinoma of the vagina (35,36), dilantin exposure with neuroblastoma (37,38), nitrosourea compounds with central nervous system tumors (39) and topoisomeras II inhibitors (epipodophyllotoxins, flavonoids, catechins, caffeine) with leukemia associated with mixed lineage leukemia (MLL) gene rearrangements (40,41,42,43). Significant use of alcohol and tobacco/marijuana and exposure to pesticides has been reported to be associated with an increased risk of congenital leukemia (44,45,46). Radiation therapy or significant exposures to radiation through diagnostic testing such as computed tomography (CT) scanning is usually avoided whenever possible in pregnant mothers because of concerns of potential morbidity to the developing fetus (47,48,49,50).
TUMORS OF NEUROEPITHELIAL ORIGIN
Neuroectodermal cells of the neural tube differentiate to neuroblasts, which become nervous system tissue and melanocytes; free spongioblasts, which become either astrocytes or oligodendroglia cells; and ependymal spongioblasts, which become ependymal cells. These primitive neuroectodermal cells may be the target for neoplasia, giving rise to a group of morphologically similar tumors involving central and peripheral sites of the nervous system. Neonatal tumors originating from neuroectodermal cells include neuroblastoma, retinoblastoma, peripheral nerve tumors (i.e., neuroepithelioma), medulloblastoma, choroid plexus papilloma, ependymoblastoma, and melanotic neuroectodermal tumors. These tumors show varying degrees of cellular differentiation, have similar histologic features (e.g., small, primitive cells with rosettes or pseudorosettes), and tend to spread along cerebrospinal fluid pathways.
Neuroblastoma
Neuroblastoma is the most common malignant tumor in neonates. It originates from neural crest cells that normally give rise to the adrenal medulla and sympathetic ganglia. Its reported occurrence in siblings and other family members suggests that some cases are hereditary (51,52). In such cases, the tumors usually are diagnosed at an earlier age and often are characterized by having multifocal primary tumors (53,54). An interesting syndrome has been reported in several women who delivered infants diagnosed as having neuroblastoma during the first few months of life (55). The mothers had sweating, pallor, headaches, palpitations, hypertension, and tingling in their hands and feet during the last trimester of pregnancy. These symptoms were relieved after the birth of the affected infants. The authors of that study postulated that this symptom complex is caused by the introduction of fetal tumor catecholamines into the maternal circulation. Although at least half of infants present with an abdominal mass from tumors arising in the adrenal medulla or retroperitoneal sympathetic ganglia, neuroblastoma may arise anywhere along the sympathetic nervous system and present with disseminated disease. An abdominal sonogram or CT scan demonstrates displacement of the kidney without distortion of the calyceal system. The neoplasm also may originate in the posterior mediastinum, neck, or pelvis. Cervical sympathetic ganglion involvement in the neck may result in Horner’s syndrome; posterior mediastinal tumors may cause respiratory distress; paravertebral tumors tend to grow through the intervertebral foramina and cause symptoms of spinal cord compression; and presacral neuroblastomas may mimic presacral teratomas. Neuroblastoma also has been detected prenatally by ultrasonography, showing a solid and sometimes cystic suprarenal mass (56,57,58). Two unusual presentations of neuroblastoma are intractable diarrhea secondary to release of vasoactive intestinal peptide, and the syndrome of opsoclonus, myoclonus, and truncal ataxia (59,60), the etiology of which remains uncertain. The diarrhea secondary to vasoactive intestinal peptide abates after removal of the neuroblastoma. In contrast is the unpredictable improvement after the removal or treatment of neuroblastoma associated with opsoclonus-myoclonus. Nevertheless, survival for children with this syndrome is excellent.
Metastatic lesions are common presenting findings of neuroblastoma, especially in the neonate (60). The primary tumor often cannot be found in infants younger than 6 months of age. These infants present with bluish subcutaneous nodules and extensive hepatomegaly. The liver may be studded with tumor nodules and be so large that it causes respiratory distress secondary to abdominal distention. Clumps of tumor cells often are found in the bone marrow aspirates. Metastases to bones, skull, and orbit, which present as periorbital ecchymoses, are rare in the neonate. The unique metastatic pattern to liver, bone marrow, and skin in infants is classified as stage IV-S neuroblastoma (61,62,63,64,65).
The differential diagnosis for neuroblastoma is limited. The subcutaneous nodules appear similar to those found in congenital leukemia cutis and several congenital infections. The leukoerythroblastosis secondary to bone marrow metastases from neuroblastoma also is observed with congenital infection, severe hemolytic disease, and leukemia. More than 90% of children with neuroblastoma will have elevated urinary excretion of catecholamine metabolites, vanillylmandelic acid or homovanillic acid, or both (66). The diagnosis of neuroblastoma is made by biopsy of the primary tumor or metastatic lesions. The most histologically primitive lesion is neuroblastoma without differentiation and is composed of small, round cells with scant cytoplasm. The ganglioneuroma, its benign counterpart, is composed of large, mature ganglion cells. Ganglioneuroblastoma is intermediate in its degree of cellular differentiation. In the absence of a tissue specimen, the findings of elevated urinary catecholamines and tumor pseudorosettes in a bone marrow specimen usually are sufficient to make a definitive diagnosis.
The prognosis for children with neuroblastoma is inversely correlated to the age of the child at diagnosis and the extent of disease. The infant with stage IV-S disease has a better chance of survival than does the older child with less advanced disease. Evans and associates (61) proposed a clinical staging system for children with neuroblastoma that was prognostically useful (67,68). This system has evolved into an International Staging System, which takes into account many of these basic concepts (63,64,65).
Infants with stage IV-S have had spontaneous regression of disease, and in other patients malignant neuroblastomas apparently have undergone maturation into mature ganglioneuromas (68). The incidence of spontaneous regression of neuroblastoma may be more common than is clinically evident. Primitive sympathetic neuroblasts, which are derived from neural crest ectoderm, migrate in early embryonic life into the adrenal primordium, in which they arrange themselves in nodules before differentiation into adrenomedullary tissue. These nodules are present in all fetal adrenal glands at 14 to 18 weeks of gestation (69). Beckwith and Perrin (70) detected the presence of microscopic clusters of neuroblastoma cells (i.e., neuroblastoma in situ) in the adrenal glands in a number of autopsies from infants younger than age 3 months who had no clinical evidence of tumor. They estimated that neuroblastoma in situ occurs in 1 of 250 stillborn infants and infants younger than 3 months of age. Clinically detectable neuroblastoma is noted in only 1 of 10,000 live births. Also pertinent to these data are the observations of the catecholamine screening programs (71,72,73,74,75). These programs demonstrated that this type of screening resulted in an increased incidence of early stage neuroblastoma that, most likely, would not have presented as clinically detectable disease (76,77,78,79,80,81).
These observations raise the interesting question regarding whether stage IV-S neuroblastoma is not a true malignancy. If stage IV-S neuroblastoma is a classic malignant neoplasm, it should be clonally derived. One such example of clonality in stage IV-S neuroblastoma tumor specimens has been demonstrated by cytogenetic analysis. Although no consistent and specific chromosomal alteration has been found in all stage IV-S specimens, there are increasing numbers of examples of cytogenetic clonality. Additionally, stage IV-S neuroblastoma is often characterized by being hyperdiploid and having variable expression of 1p deletions and N-myc amplification (82,83,84,85,86). The reason(s) for spontaneous regression of stage IV-S disease remains a mystery.
The extent of treatment for neuroblastoma depends on the stage of the disease and biological factors such as histology, N-myc amplification, and DNA ploidy. The infant with stage IV-S disease should be observed for a period of weeks to months before treatment is initiated because of the reasonable likelihood of spontaneous regression (67,79,87,88). Respiratory difficulties, blood vessel (usually vena cava) obstruction, and gastrointestinal compression secondary to rapid tumor expansion can develop and should be considered a medical emergency, with more than 50% mortality in some reports (89). Hsu and associates (90) have described a monitoring and scoring system to follow these patients and predict when, and if, therapeutic interventions should be initiated. This scoring system is based on the presence of the severity of several clinical symptoms, including respiratory compromise, renal insufficiency, extent of lower extremity edema, disseminated intravascular coagulopathy, and the rate of increasing abdominal girth (90). Although both chemotherapy (often single-agent cyclophosphamide) or radiation therapy have been used to effect symptomatic relief for such patients, some reports suggest that 200 to 600 cGy delivered tangentially to the liver to avoid other organs, such as kidneys and gonads, provides for a rapid response and possibly an improved outcome (90,91). Embolization approaches have also been reported to be successful (92).
Complete surgical removal of neuroblastoma is usually accomplished in infants with stage I or II disease. Postoperative treatment generally is not indicated for these patients, and their long-term survival is excellent (93,94,95). For infants with stage III disease in whom gross residual tumor remains after surgery, postoperative chemotherapy, radiotherapy, or both may be indicated (96). Chemother-apy is the treatment of choice for infants with stage IV disease. The active chemotherapeutic agents against neuroblastoma
include alkylating compounds (e.g., cisplatinum, cyclophosphamide, dacarbazine, nitrogen mustard), vincristine, and doxorubicin. Combinations of several of these agents administered for 6 months to 1 year have resulted in greater than 50% long-term survival for children younger than 1 year of age with stage IV neuroblastoma and, in some subgroups up to 90% cure rates (97). This is in contrast to the dismal prognosis for similarly staged children who are older than 1 year.
include alkylating compounds (e.g., cisplatinum, cyclophosphamide, dacarbazine, nitrogen mustard), vincristine, and doxorubicin. Combinations of several of these agents administered for 6 months to 1 year have resulted in greater than 50% long-term survival for children younger than 1 year of age with stage IV neuroblastoma and, in some subgroups up to 90% cure rates (97). This is in contrast to the dismal prognosis for similarly staged children who are older than 1 year.
Retinoblastoma
Retinoblastoma is a congenital malignant tumor arising from the nuclear layer of the retina. Although an extremely rare tumor, it is the most common ocular tumor of childhood. The median age at presentation is 18 months, or 14 months for bilateral cases, but a small percentage of infants are diagnosed during the first few months of life (98). Prenatal diagnosis by sonography of retinoblastoma has been reported (99). Approximately 10% of children with retinoblastoma have a family history of the disease. Approximately 30% with bilateral or multifocal unilateral tumors have a negative family history (100,101). These two groups are capable of transmitting the disease to their offspring in an autosomal dominant fashion. This hereditary tendency is governed by a genetic locus on the long arm of chromosome 13 (i.e., band 13q14), which includes the retinoblastoma (rb) tumor suppressor gene. Retinoblastoma has also been associated with chromosomal 13 deletion mosaicism (102). Preimplantation diagnosis of retinoblastoma has been reported (103). If there is a family history of retinoblastoma, an experienced ophthalmologist should examine the eyes of unaffected siblings regularly although they are under general anesthesia to detect cases early (104). Moll and associates have proposed such screening until age 4 years (105).
The most common initial signs of retinoblastoma include an abnormal white pupil (i.e., leukocoria), known as a cat’s-eye reflex and a squint or strabismus. A clinical picture resembling retinoblastoma may result from granulomatosis uveitis, congenital defects, and severe retrolental fibroplasia. Once the diagnosis of retinoblastoma is suspected, both eyes should be examined with the infant under general anesthesia. A bone marrow aspiration and spinal tap for malignant cells should be performed for staging. A staging system for retinoblastoma is based on the size, location, number of tumors in each eye, and distant hematogenous metastases. Vitreous seeding, tumors extending anteriorly to the ora serrata, tumors invading over one-half of the retina, residual orbital disease, and optic nerve or distant metastases are adverse prognostic features.
Retinoblastoma usually is curable when diagnosed early; vision often need not be sacrificed even when bilateral disease is present (106). More recent treatment approaches using cryosurgery in combination with chemotherapy and/or radiation therapy are being developed with the hope of preserving vision and reducing the risk of subsequent in-field radiation-induced secondary malignancies (106,107,108,109,110). Additionally, stereotactic and proton beam radiation methods are being tested to reduce the amount of normal tissue irradiated and the risk of radiation-induced secondary malignancies (106,111,112,113,114,115,116). Patients with adverse prognosis features often require enucleation. Children with very advanced and metastatic disease require aggressive chemotherapy regimens.
Brain Tumors and Other Neuroectodermal Tumors
Intracranial tumors presenting in the first year of life are uncommon. In a review from the Hospital for Sick Children in London, 107 of 1,296 children with brain tumors had symptoms before the age of 1 year; 17 were symptomatic within 2 months of birth (117,118). Brain tumors in children in this age group tend to be supratentorial, in contrast to those in older children, which tend to be infratentorial. In infants, the most common presenting symptom is macrocrania, with a bulging fontanelle secondary either to hydrocephalus or tumor volume. Seizures, vomiting, failure to thrive, abnormal eye movement, and irritability also are frequent. The histologic diagnoses of the neuroectodermal tumors are similar to those of tumors in later childhood, with gliomas accounting for most. Of particular interest, a majority of intracranial tumors in neonates have been reported to be teratomas (12,118,119,120,121). There is also a high frequency of choroid plexus papillomas in this age group (122). An association of choroid plexus papillomas and the presence of SV40 viral DNA has been reported (123).
Desmoplastic infantile gangliogliomas are rare, but massive, cystic tumors that usually occur supratentorially in the neonatal period (124). They present most commonly with signs of increased intracranial pressure, including seizures. Therapy has included surgery and chemotherapy, but without radiation therapy. For patients who have a complete surgical resection, additional therapy may not be required (125). Prognosis may be better than that observed with other tumors, such as high-grade astrocytoma. In contrast, pineoblastomas are malignant tumors with an extremely poor outcome, even when treated with surgery, chemotherapy, and radiation therapy. Part of the reason for the poor outcome in these infants may be the propensity of pineoblastoma to involve the leptomeninges and extraneural spread (125).
Treatment of infants with brain tumors historically included surgical removal or biopsy followed by radiotherapy (126,127,128). Operative mortality has been high, and few infants survived for longer than 1 year. In these few, brain radiotherapy resulted in severe intellectual and psychomotor retardation. In attempts to avoid the adverse effects of radiotherapy on the developing brain, more recent therapeutic approaches have evaluated surgery followed by preradiation chemotherapy (119,127,128,129,130,131).
Atypical teratoid/rhabdoid tumors (AT/RT) of the central nervous system, although rare, are usually large, aggressive
and have historically had very poor outcomes with the use of conventional therapies used in children with brain tumors (132,133,134). More aggressive, multidisciplinary approaches have started to show some improvement in patients with this type of brain tumor, although median survival is still estimated to be less than 2 years (134,135). Of particular note is the etiologic role of mutations in the SNF5/INI1 gene in these tumors which encodes for a chromatin remodeling factor (136,137).
and have historically had very poor outcomes with the use of conventional therapies used in children with brain tumors (132,133,134). More aggressive, multidisciplinary approaches have started to show some improvement in patients with this type of brain tumor, although median survival is still estimated to be less than 2 years (134,135). Of particular note is the etiologic role of mutations in the SNF5/INI1 gene in these tumors which encodes for a chromatin remodeling factor (136,137).
Primitive neuroectodermal tumors of peripheral nerve represent a group of soft tissue tumors known as neuroepitheliomas, medulloepitheliomas, and peripheral neuroblastomas (138,139). They are associated with major branches of peripheral nerves (i.e., tumor of the chest wall arising from intercostal nerve). These are extremely rare tumors, quite aggressive in their biologic behavior, with frequently occurring distant metastases, including to the CNS. Treatment approaches include wide excision, if possible, and chemotherapy modeled after either neuroblastoma or brain tumor protocols.
The melanotic neuroectodermal tumor of infancy has its origin in the neural crest population. Most of these tumors are diagnosed between 1 and 8 months of age and occur in the maxilla, although extremely rare cases have been reported in other sites such as the epididymis (140). They are considered a benign neoplasm, with a local recurrence rate of about 15%. These tumors originate from pluripotential neural crest cells that give rise to both melanoblasts and neuroblasts. The rate of malignancy for this tumor is reported to be approximately 5%. Recommended treatment is wide local excision.
CONGENITAL LEUKEMIA
Leukemia in the newborn is extremely rare with approximately 5 cases reported per million live births (141,142,143). It has been customary to categorize leukemia as congenital when it is diagnosed within a few days after birth and as neonatal when it manifests itself during the first 4 to 6 weeks of life (144). The kinetics of leukemic cell growth and the estimated leukemic cell burden at the time of diagnosis make it reasonable to assume that clinically detectable leukemia during the first 4 weeks of life originated in utero (145). Molecular studies indicate a prenatal initiation of acute lymphoblastic leukemia (ALL) and some acute myeloid leukemias, in children diagnosed in infancy or even later in life (146,147,148,149,150,151,152,153). In the following discussion, congenital leukemia is considered as leukemia diagnosed from birth to 4 weeks of age.
The etiology and pathogenesis of congenital leukemia, and other leukemias, are unknown. The strongest evidence for a genetic predisposition to acute leukemia is its occurrence in identical twins in which there is a very high concordance (154). If leukemia develops in one of a set of identical twins before 6 years of age, the risk of disease in the other twin is 20%. Leukemia usually develops in the other twin within weeks to months of the first case. In some of these cases, intrauterine exchange of leukemic cells from one twin to the other has been strongly suggested by the identity of molecular and genetic changes observed postnatally in the leukemias from each twin. For fraternal twins and siblings, the risk of development of leukemia is two and four times higher than in the general population. Congenital leukemia has been associated with trisomy 9, trisomy 13, Turner syndrome, and Down syndrome and rearrangements of the MLL gene (147, 155,156,157,158). More than 95% of the childhood leukemias, including congenital leukemia, are classified as acute, because they are characterized by a predominance of immature lymphoid or myeloid precursors. In children, the proportion of cases of ALL to acute myelogenous leukemia (AML) is approximately 4 to 1, but this ratio is reversed in the congenital leukemias (141,142). Whereas lymphoblasts from most children with ALL express the common acute lymphoblastic leukemia antigen (CALLA or CD10) on the cell surface and express markers of early B-cell differentiation (e.g., cytoplasmic immunoglobulin or Ig gene rearrangement), the lymphoblasts of congenital and infant ALL are often pre-B, but CD10-negative (159,160). These infants have a higher incidence of CNS leukemia at diagnosis, a higher leukocyte count, increased frequency of hepatosplenomegaly, and a poorer prognosis than older children with ALL. A translocation of the long arms of chromosomes 4 and 11, t(4;11), involving the MLL gene at chromosome 11q23, is commonly observed in infant leukemia. This translocation is associated with greater than 80% of ALL in infancy and carries a poor prognosis (161,162,163,164,165,166).
The most common subtype of AML in the neonate is acute monocytic leukemia, which accounts for only 20% of AML in older children (167). It is associated with a high incidence of extramedullary leukemia, especially in the CNS. Translocations involving chromosome 11q23 with MLL gene rearrangements are associated with this subtype of AML (167).
Cutaneous manifestations are the most frequent clinical findings noted at birth. In addition to petechiae and purpura, leukemic skin nodules (i.e., leukemia cutis) have been observed in approximately 50% of cases (168,169,170). These skin nodules may vary in size from a few millimeters to a few centimeters, are bluish to slate gray in color, may appear in all sites, and are palpated as firm tumors of the deep skin (Fig. 53-1). Neonatal leukemia cutis may undergo a spontaneous, temporary regression, but tends to recur in a more generalized form within a few weeks to months.
Hepatosplenomegaly is common, but lymphadenopathy is not. Respiratory distress, secondary to leukostasis within the pulmonary vasculature, may complicate the clinical course. Other nonspecific symptoms of neonatal leukemia include lethargy, pallor, poor feeding, and umbilical, gastrointestinal, or genitourinary bleeding.
In the report of congenital leukemia by Pierce (141), the mean hemoglobin concentration at birth was normal, with a wide range of values (7 to 20 g/dL); the mean leukocyte counts were 150,000 per mm3 (range 2,000-850,000); and mean platelet counts were 70,000 per mm3 (range
6,000-300,000). The diagnosis of leukemia is confirmed by examination of a bone marrow aspirate obtained from the posterior iliac crest.
6,000-300,000). The diagnosis of leukemia is confirmed by examination of a bone marrow aspirate obtained from the posterior iliac crest.
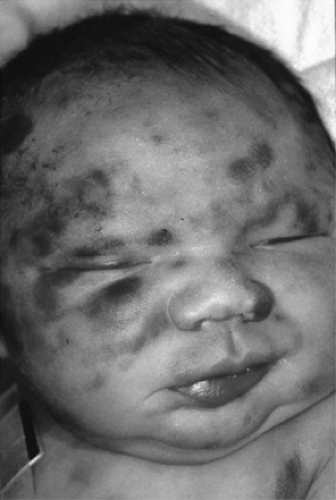 Figure 53-1 Congenital acute monocytic leukemia with skin nodules. (See color plate) |
A variety of disorders in the newborn imitate leukemia. The newborn bone marrow response to infection, hypoxemia, or severe hemolysis commonly is a leukemoid reaction and an increase in circulating nucleated erythrocytes. These conditions can easily be confused with congenital leukemia.
An enigmatic myeloproliferative disorder described as transient acute leukemia or transient myeloproliferative syndrome occurs primarily in infants with Down syndrome (171,172,173). This syndrome, noted during the first few days of life, mimics AML. Peripheral leukocyte counts can range from 25,000 to several hundred thousand; bone marrow aspirates reveal 30% to 70% blasts. Hepato-splenomegaly and thrombocytopenia are also common findings. The hematologic status of these neonates usually returns to normal in 1 to 4 months, with only supportive therapy. Several of these children who subsequently died of cardiac or pulmonary disease years after the resolution of their transient myeloproliferative syndrome showed no evidence of leukemia at autopsy. This syndrome has been observed in neonates with stigmata of Down syndrome and in phenotypically normal infants who have trisomy 21 mosaicism in their hematopoietic cells or skin fibroblasts (174). Molecular studies have identified the presence of truncated GATA1 mutations in essentially all cases of transient myeloproliferative syndrome and the acute mega-karyoblastic leukemia associated with Down Syndrome (175,176,177,178,179,180). GATA1, a critical transcription factor that regulates megakaryopoiesis and erythropoiesis, has thus been etiologically linked in these two hematologic disorders and in the increased drug sensitivity of leukemic mega-karyoblasts from children with Down Syndrome (181). There have also been reports of spontaneous remissions of congenital leukemia, even in those children without Down Syndrome (182,183), suggesting that some cases can be initially conservatively managed with observation and/or supportive care (144,184).
In approximately 20 to 30% of these infants, acute leukemia requiring systemic chemotherapy will develop within the first 4 years of life. This leukemia is nearly always acute megakaryoblastic leukemia (M7 subtype). In the setting of constitutional trisomy 21, acute megakaryoblastic leukemia usually has an excellent prognosis, even with less intense therapy than observed for non-Down syndrome children with the same subtype of leukemia (185,186,187,188). Although time is still the most definitive indicator of transiency, serial cytogenetic studies may be of value. If there is a chromosome marker in addition to trisomy 21 in spontaneously dividing bone marrow cells, a more aggressive leukemic clone is likely to be present.
Congenital ALL is fatal if untreated and should be managed with systemic chemotherapy. Several national trials are ongoing to improve the outcome of these infants with this poor prognosis leukemia. Age has been an important prognostic variable in childhood ALL, with the most favorable prognosis for patients between 2 and 9 years of age. Prognosis for young infants with ALL remains poor (162, 189,190,191,192). Because untreated congenital AML also is fatal, aggressive combination chemotherapy in an institution with maximal supportive services is mandatory. One report has described the successful treatment of several neonates with acute monocytic leukemia with either VP-16 or VM-26, but this experience has not been confirmed (193). Aggressive AML treatment regimens have met with considerable success in achieving long-term disease-free survival in neonates with AML (194,195,196,197,198,199).
NEOPLASMS OF THE KIDNEY
Mesoblastic Nephroma
Most abdominal masses presenting in infancy are renal in origin, and most can be accounted for by cystic disease of the kidney and congenital malformations of the urinary tract leading to hydronephrosis. Although neoplasms of the kidney are rare in infancy, they do occur and have important prognostic implications, making it mandatory that they be included in the evaluation of abdominal masses.
The most common renal tumor found in infants is mesoblastic nephroma, which accounts for nearly 80% of renal tumors in the neonatal period. It also has been called fetal renal hamartoma, mesenchymal hamartoma of infancy, and leiomyomatous hamartoma (200). Mesoblastic nephroma commonly presents as an asymptomatic, enlarging abdominal mass during the first few months of life (201). It is not associated with congenital anomalies and has no race predilection. Of note is the more frequent occurrence of polyhydramnios and premature labor in women whose infants have mesoblastic nephroma (202). The differential diagnosis includes renal cystic disease, congenital malformations of the urinary tract resulting in hydronephrosis, and Wilms tumor. Molecular studies have demonstrated that mesoblastic nephroma shares with most Wilms tumors the expression of insulin-like growth factor II (IGF-II), but, unlike Wilms tumor, does not express the WT1 gene and shows no loss of heterozygosity at 11p13 or 11p15 (203). The bi-allelic expression of the IGF-II has shown that there is a relaxation of the normal epigenetic imprinting pattern of this gene (i.e., the paternal allele is normally expressed with the maternal allele silent) (204). An intriguing finding has been the identification of a chromosomal translocation resulting in a fusion protein, ETV6-NTRK3, in both mesoblastic nephroma and infantile fibrosarcoma (205,206). Of further interest is the requirement of IGF-1 signaling for the transformation potential of the ETV6-NTRK3 to be realized (207).
Most patients with mesoblastic nephroma are cured by surgical excision without adjuvant chemotherapy or radiotherapy (208,209,210). The addition of chemotherapy has resulted in increased morbidity and, in some instances, fatal complications. In rare cases, such as older infants presenting with metastatic disease or when there is tumor rupture and spillage, chemotherapeutic intervention with regimens containing actinomycin D, vincristine, cyclophos- phamide, and doxorubicin have been used effectively. Similar regimens have been used in the rare recurrences (209, 211,212,213). Radiotherapy also has shown efficacy in patients for recurrences, which are relatively uncommon (210,214,215).
Persistent Renal Blastema, Nephroblastomatosis, and Wilms Tumor
The adult or metanephric kidney arises from a complex, inductive interaction between the evaginating uteric bud and its bifurcations with the metanephric, mesodermally derived blastema. By 36 weeks of gestation, normal nephrogenesis is complete, with no residual metanephric blastema. When these metanephric blastemal elements persist, they usually are characterized by microscopic clusters of primitive blastema and occasionally some tubular differentiation (i.e., persistent metanephric blastema). If these fetal rests proliferate, they may develop along several different histologic pathways, each of which has particular relevance to the evolution of Wilms tumor (216,217,218,219). Nephroblastomatosis represents the persistence and cellular expansion of metanephric blastema beyond the cessation of nephrogenesis. The proliferation may occur in characteristic patterns, either multifocal or diffuse.
Multifocal nephroblastomatosis refers to the widespread proliferation of blastemal cells, most prominently in the subcapsular cortex and along the penetrating columns of Bertin. Nephromegaly is not always evident. Unlike mesoblastic nephroma, multifocal nephroblastomatosis is associated with congenital malformation syndromes and chromosomal abnormalities. Within the category of multifocal nephroblastomatosis, there are several characteristic lesions. When persistent blastema proliferate in small 100- to 300-mm foci separated by normal renal parenchyma, they are referred to as nodular renal blastema. Nodular renal blastema may regress or evolve into what has been called sclerosing metanephric hamartoma and into Wilms tumorlets, which are 0.3 to 3.5 cm in diameter, noninfiltrating, often multiple, neoplastic tumors separated by normal renal parenchyma. They usually consist of blastema with a monomorphous epithelial pattern of differentiation. Although they resemble true Wilms tumor, they are distinguishable by their smaller size and their noninfiltrating behavior.
A second type of nephroblastomatosis, which is quite rare but found more commonly in infants and young children, is diffuse nephroblastomatosis. The blastemal proliferation may be pan-nephric or superficial, with the latter lesion encasing a normal cortex and medulla. Diffuse nephroblastomatosis presents as bilateral, palpable nephromegaly in association with congenital malformations. Radiographic examination by intravenous pyelogram reveals distortion and elongation of the calyceal system without obstruction (220). On gross inspection, there is an exaggerated pattern of fetal lobulation of the enlarged kidneys.
That these various histologic lesions are related to one another and to the evolution of frank Wilms tumor has been strongly suggested by case studies and by epidemiologic and pathologic correlations (221,222,223). In about one-third of cases of Wilms tumor, there is suggestive pathologic evidence for the association of nodular renal blastema, nephroblastomatosis, Wilms tumorlets, and Wilms tumor; in bilateral Wilms tumor, this association is nearly always present (224,225,226).
Management of nephroblastomatosis involves surgery and sometimes chemotherapy, depending on the extent of disease (227,228). Radiation therapy is not very effective. If only one kidney is involved, surgical resection is sufficient, but exploration and biopsy of the contralateral kidney are critical. When both kidneys are extensively involved, nephroblastomatosis usually will respond to the combination chemotherapy used in Wilms tumor (i.e., vincristine and actinomycin). The goal of such treatment is to cause regression of the nephroblastomatosis or cause its development into an end-stage hamartoma. The duration of treatment is based on clinical response. Close follow-up with both radiographic and second-look operations is important in that patients may still progress to the development of true Wilms tumor despite therapy.
True Wilms tumor rarely is seen in the neonatal period (201). It generally presents as an asymptomatic abdominal mass that does not cross the midline but, occasionally, the mass is large enough to cause dystocia at the time of delivery. It is rarely associated with gross hematuria, hypertension, or polycythemia secondary to increased erythropoietin levels. The most common congenital abnormalities associated with Wilms tumor are genitourinary and musculoskeletal anomalies, hemihypertrophy, aniridia, and hamartomas (e.g., hemangiomas, nevi, cafe-au-lait spots). Additionally, there is the Wilms tumor-aniridia syndrome, associated with a deletion of part of the short arm of chromosome 11. CT scan and particularly magnetic resonance imaging (MRI), along with renal ultrasonography, can help define the extent of the tumor.
Pathologically, classic Wilms tumor consists of neoplastic blastemal elements with epithelial and stromal components. In the neonate, Wilms tumor is predominantly epithelial and localized, displaying little invasiveness or metastatic potential. The primary prognostic variables include histology, extent of disease, and age (209,211, 229,230,231).
The management of a patient with Wilms tumor depends primarily on staging. In the neonate, most patients will be classified as stage I, in that the tumors usually are relatively small (i.e., less than 550 g), localized, noninvasive, and completely resectable. Neonatal Wilms tumor commonly shows favorable histology and appears to metastasize infrequently. At the time of surgery, a frozen section diagnosis is important in ascertaining whether or not nephroblastomatosis is also present. If nephroblas tomatosis is present, wedge biopsy of the contralateral kidney is indicated, even if it is grossly normal. The management of bilateral Wilms tumor must often be individualized, with the intent of trying to spare as much normal renal parenchyma as possible (232,233). Careful follow-up in such cases is critical.
For patients with stage I disease, the National Wilms Tumor Study group has recommended 6 months of combination chemotherapy with vincristine and actinomycin; radiation therapy is not given. Disease-free, long-term survival is greater than 90%. Other studies have shown that in infants with localized, noninvasive, nonmetastatic, histologically favorable tumors weighing less than 550 g, are likely to do well with reduced courses of chemotherapy or even no further therapy apart from radical nephrectomy (209,230,234). For advanced stages and for tumors with unfavorable histology, more aggressive therapy, including radiation and intensive chemotherapy, is used (230,235).
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


