Anatomy of the most common types of atrial septal defect, viewed from the right atrium. RA = right atrium; RV = right ventricle.
Regardless of the anatomical position, any ASD permits blood flow from the left to the right atrium, which leads to enlargement of the right atrium and ventricle and increased pulmonary blood flow (Figure 14.2).
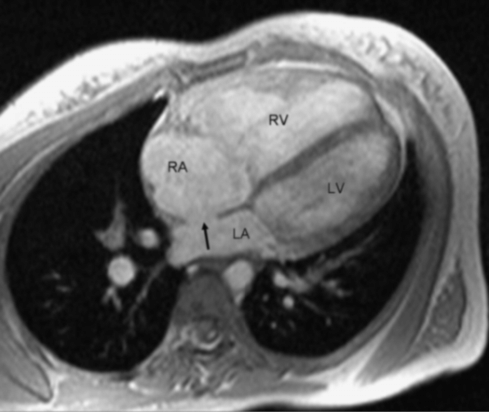
Cardiac magnetic resonance image of an atrial septal defect (ASD; arrow); the ASD results in left-to-right shunting; blood from the pulmonary veins enters the left atrium (LA), after which some of it crosses the ASD into the right atrium (RA), subsequently leading to enlargement of the RA and right ventricle (RV). LV = left ventricle.
Indications for ASD closure mostly depend on the size of the defect and the degree of right heart volume overload. A small defect without any evidence of right atrial or right ventricular enlargement does not warrant closure (unless there is previous history of paradoxical embolism with transient ischemic attack or stroke). An ASD leading to right heart enlargement (the ratio of pulmonary to systemic blood flow is usually >1.5) should ideally be closed before pregnancy. Pulmonary hypertension (PH) is not common in patients with ASD but has to be excluded before closure. Anticipated benefits of ASD closure are improved functional class and overall quality of life, as well as risk reduction for atrial arrhythmia and right heart failure.[14–17]
In the absence of PH and right ventricular dysfunction, pregnancy is well tolerated in a woman with an ASD, regardless of whether it is repaired or unrepaired.[18]
In patients with an unrepaired ASD, the pregnancy-related decrease in systemic vascular resistance reduces the magnitude of left-to-right shunting. Consequently, the effects of increased cardiac output and blood volume on the right ventricle are attenuated.
Atrial arrhythmias (from right atrial distension) and paradoxical embolism are potential hazards during pregnancy, although the latter is uncommon. A prepregnancy history of arrhythmia and maternal age older than 30 years are risk factors for maternal cardiac complications.[18] Low-dose aspirin can be considered to reduce the risk of thromboembolic events, and thromboprophylaxis with low-molecular-weight heparin (LMWH) should be commenced if bed rest is required. Furthermore, prevention of venous stasis (use of compression stockings and avoiding the supine position) and use of an air-filter or avoidance of air in intravenous lines is important to prevent systemic embolization due to potential right-to-left shunting during labor.[7,19]
Women with an unrepaired ASD have a slightly increased risk of preeclampsia, fetal loss, and low birth weight, whereas patients with repaired defects have a risk similar to that of the general population.[18]
Tetralogy of Fallot
ToF is the most common cyanotic congenital heart lesion and constitutes about 5–10% of cases of CHD.[20,21] ToF consists of a large VSD, right ventricular outflow tract obstruction, right ventricular hypertrophy, and overriding of the aorta (Figure 14.3).
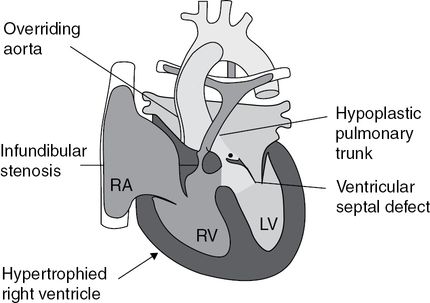
Tetralogy of Fallot is characterized by a large ventricular septal defect (VSD), an aorta that overrides the left and right ventricles, obstruction of the right ventricular outflow tract, and right ventricular hypertrophy; with substantial obstruction of the right ventricular outflow tract, blood is shunted through the VSD from right to left.
Surgical repair includes VSD closure and relief of the right ventricular outflow tract obstruction. In cases with a small pulmonary valve annulus, the outflow tract obstruction can be relieved by a transannular patch, leading to free pulmonary regurgitation. Over time, this massive pulmonary regurgitation may lead to right ventricular enlargement and dysfunction, which is associated with ventricular tachycardia and sudden cardiac death.[22] Atrial arrhythmias are not uncommon after repair of ToF, especially if right atrial enlargement is present.
Nevertheless, pregnancy is a relatively low-risk endeavor for most women with repaired ToF. The risks of pregnancy largely depend on the hemodynamic status after repair. Mild to moderate right ventricular outflow tract obstruction is well tolerated and so is pulmonary regurgitation, provided that right ventricular function is maintained. Severe pulmonary regurgitation after ToF repair with right ventricular dilatation has, however, been reported as a maternal risk factor during pregnancy; pulmonary valve replacement prior to pregnancy has therefore been recommended.[23] More recent data question this strategy by showing that severe pulmonary regurgitation and right ventricular dilatation are well tolerated unless additional risk factors such as twin pregnancy, branch pulmonary stenosis, right ventricular systolic dysfunction, and right ventricular hypertrophy are present.[24] Furthermore, the risk of adverse maternal events associated with pregnancy increases if there is a history of ablation, cardiovascular medication, right or left ventricular dysfunction, PH due to small pulmonary arteries, or previous central aortopulmonary shunt procedures.[25–28]
During pregnancy, regular assessment should focus on signs of right and left heart dysfunction and clinical arrhythmia. Maternal cardiovascular events during pregnancy are highly associated with perinatal complications for the fetus.[27]
The long-term impact of pregnancy on right ventricular size and function in repaired ToF patients is uncertain. There are some data to indicate that pregnancy in this patient group may result in persisting medium-term enlargement of the right ventricle, an effect that has been observed in nonpregnant ToF control patients.[29]
ToF can be part of the DiGeorge syndrome (present in 15% of ToF patients). Genetic testing should be offered before pregnancy because the recurrence risk of DiGeorge syndrome is 50%. In its absence, the risk of recurrence of CHD in the fetus is about 2–3%.[30]
Small retrospective controlled cohort studies show a mildly higher incidence of antenatal complications such as spontaneous rupture of membranes, gestational hypertension, and antepartum hemorrhage in women with repaired ToF. Neonatal outcomes were similar, despite a significantly lower birth weight: babies of mothers with a repaired ToF were on average about 500 g lighter than those of a healthy control population.[31]
Ebstein anomaly of the tricuspid valve
Ebstein anomaly is an apical displacement of the tricuspid valve leading to enlargement of the right atrium and, often, tricuspid regurgitation. The anomaly is frequently associated with an ASD that allows right to left shunting and cyanosis, as well as with Wolff–Parkinson–White syndrome in about 15% of patients. Survival into adulthood is common in mild to moderate cases, but patients can present with right heart failure, supraventricular arrhythmias, or cyanosis.
Owing to inefficient right heart function, patients have reduced cardiac output reserve, which can be further compromised by atrial reentry tachycardia. Chest radiography to assess heart size prior to conception (Figure 14.4) is an approximate, but reproducible, marker of severity of disease.
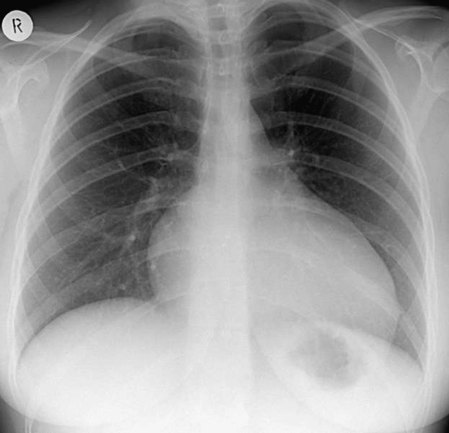
Typical chest X-ray of a moderate Ebstein anomaly of the tricuspid valve; note a large heart with normal and not increased pulmonary vascular markings and a small pedicle of the heart (small pulmonary artery and aorta), indicative of the low cardiac output status.
In women without cyanosis or signs of heart failure, pregnancy is usually well tolerated. However, Ebstein anomaly is associated with an increased risk of preterm birth and fetal loss, especially in cyanotic women.[32,33] Birth weight is often lower, especially in cyanotic women. Monitoring during pregnancy should focus on the potential development of cyanosis, heart failure, and arrhythmias. If an ASD is present, low-dose aspirin should be considered to reduce the risk of thromboembolic events, and thromboprophylaxis with LMWH should be commenced if bed rest is required.
Pulmonary stenosis
Pulmonary stenosis is an isolated congenital obstruction of the right ventricular outflow tract, usually at the level of the pulmonary valve and occasionally at the sub- or supravalvar level. It accounts for about 10% of cases of CHD. Catheter interventional or surgical relief of pulmonary stenosis is recommended for patients with a peak pressure gradient across the stenosis of >50 mmHg or a peak right ventricular pressure of >75 mmHg. Ideally, it should be performed before pregnancy.[34]
Adult patients with pulmonary stenosis are mostly asymptomatic, but when the stenosis is severe and longstanding they can develop right heart failure or atrial arrhythmia. If interatrial communication is present, right to left shunting may occur and result in cyanosis. Isolated right ventricular outflow tract obstruction—even when severe—is usually well tolerated during pregnancy when right heart function is preserved.[35] Balloon valvuloplasty should be considered in cases of right heart failure during early pregnancy (see Chapter 8).[36]
Pulmonary stenosis can be part of Noonan syndrome (clinical features: short stature, a short webbed neck, pectus excavatum, hypertelorism, and low-set ears). Noonan syndrome is of autosomal dominant inheritance and therefore carries a recurrence risk of 50%. Genetic testing should be offered before pregnancy if Noonan syndrome is suspected clinically. Furthermore, patients with Noonan syndrome have a more myxomatous type of pulmonary stenosis, often involving the supravalvar area, and usually do not respond well to balloon valvuloplasty.
Congenitally corrected transposition of the great arteries
In ccTGA, the ventricles and great arteries are “inverted.” Systemic venous return enters the left ventricle, which then ejects into the pulmonary artery. Pulmonary venous return enters the right ventricle, which fills the aorta. The circulation is therefore “physiologically corrected” but the right ventricle supports the systemic circulation (Figure 14.5).
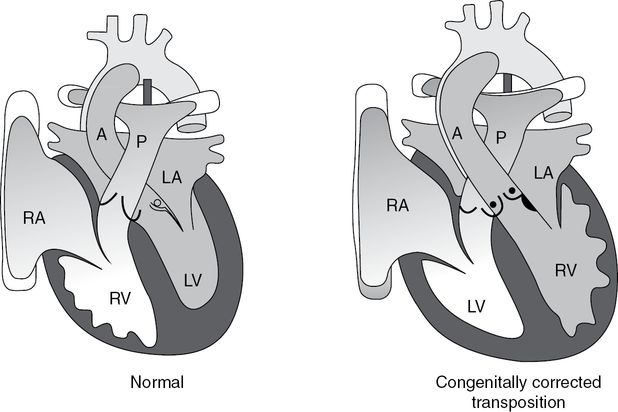
The ventricles are inverted in congenitally corrected transposition of the great arteries; systemic venous return enters the left ventricle (LV), which ejects into the pulmonary circulation (P); pulmonary venous return enters the right ventricle (RV), which supports the systemic circulation. A = aorta, RA = right atrium, LA = left atrium.
ccTGA is often associated with the following heart defects, which have an impact on prognosis:
systemic (tricuspid) atrioventricular valve abnormalities (Ebstein-like) with valve insufficiency
VSD
subpulmonary stenosis
complete heart block (acquired)
Wolff–Parkinson–White syndrome.
Successful pregnancy can be achieved by many women with ccTGA and a systemic right ventricle. The overall risk for this group depends largely on associated lesions, systemic ventricular function, function of the systemic atrioventricular valve (tricuspid valve), and the woman’s functional class.[37,38] While the long-term impact of successful and completed pregnancy on right ventricular function in women with ccTGA is somewhat unclear, a mortality rate of up to 4% and an increased risk of fetal loss has been reported in a small series of women with systemic right ventricles, and this needs to be discussed in preconception counseling.[8,39–41]
CMR imaging should be performed as part of the prepregnancy evaluation because it provides invaluable data on the function of the systemic right ventricle and the function of the tricuspid valve. A treadmill exercise before pregnancy will document the woman’s functional status and her ability to increase heart rate during exercise. Monitoring during pregnancy should include serial assessment of ventricular and tricuspid valve function, as well as of the heart rhythm. Ventricular and valve function may deteriorate when blood volume and cardiac output increase during pregnancy.
Transposition of the great arteries
TGA accounts for 5–7% of all congenital heart malformations. In TGA, the right ventricle gives rise to the aorta and the left ventricle gives rise to the pulmonary artery (Figure 14.6). The majority of adults with this condition will have had an “atrial switch operation” (Mustard or Senning operation) in childhood, in which the systemic venous return was directed to the subpulmonary left and the pulmonary venous return was directed to the systemic right ventricle using baffles. After an atrial switch operation, patients have the right ventricle supporting the systemic circulation, as in ccTGA.
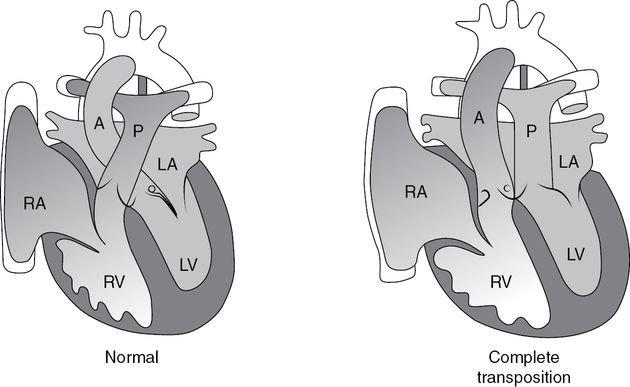
Transposition of the great arteries (TGA) and atrial switch operation; in TGA (complete transposition), systemic venous blood returns to the right atrium (RA), from which it goes to the right ventricle (RV) and then to the aorta (A); pulmonary venous blood returns to the left atrium (LA), from which it goes to the left ventricle (LV) and then to the pulmonary artery (P).
Late complications following the atrial switch operation include baffle leak or stenosis, sinus node dysfunction with bradycardia, atrial arrhythmias, and dysfunction of the systemic right ventricle with secondary tricuspid regurgitation. Women with good or only mildly impaired right ventricular function and no history of arrhythmia have a relatively low risk during pregnancy.[42,43]
Prepregnancy evaluation should include a thorough assessment of ventricular function. Patency of the atrial baffles should also be evaluated before pregnancy to ensure that an increased cardiac output can be accommodated (Figure 14.7). Both ventricular function and patency of the baffles are ideally assessed with cardiovascular magnetic resonance imaging.
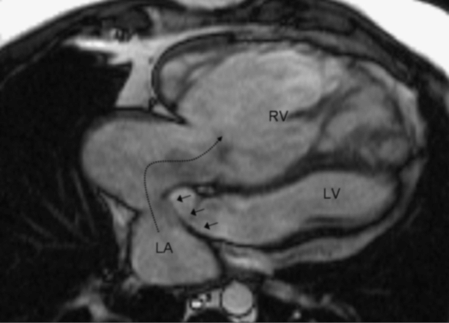
Cardiac magnetic resonance imaging of a patient with transposition of the great arteries and atrial switch operation; a baffle (small arrows) is created in the atria to redirect blood from the left atrium (LA) into the systemic right ventricle (RV); systemic venous blood returns to the subpulmonary left ventricle (LV); the systemic RV is markedly enlarged, squashing the LV.
As tachycardia is part of the physiological adaptation of the circulatory system to pregnancy, exercise testing may provide valuable information on chronotropic competence before pregnancy.
During pregnancy, clinical assessment should focus on the early signs of heart failure and arrhythmias. If atrial tachycardia occurs, sinus rhythm should be restored without delay. Direct current cardioversion is usually effective and safe when needed during pregnancy.
There is an increased risk of thromboembolic events during pregnancy in women who have undergone the atrial switch procedure, mainly as a consequence of atrial tachycardia and/or systemic ventricular dysfunction. As a consequence, anticoagulation with warfarin/LMWH should be considered when atrial tachyarrhythmia is present.[19]
Early evidence suggests that pregnancy may have an adverse long-term effect on ventricular function and on the functional class of women with TGA and previous atrial procedures; this needs to be investigated further.[39,44]
Univentricular hearts after Fontan-type operations
Many adults with functional “univentricular hearts” have undergone Fontan-type operations. Following the Fontan operation, the systemic venous return is diverted directly to the pulmonary circulation without incorporation of a subpulmonary ventricle. Blood flow to the lungs is driven mainly by systemic venous pressure and respiration. The “single” ventricle can be of right- or left-sided morphology and supports the systemic circulation (Figure 14.8).
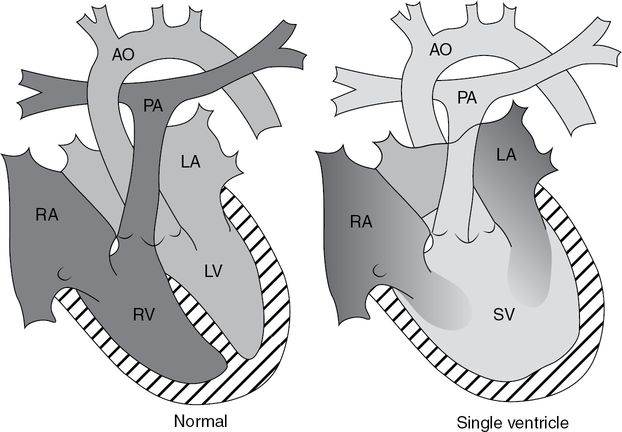
As opposed to in the normal heart, a “single” ventricle (SV) collects blood from the right atrium (RA) and the left atrium (LA). PA = pulmonary artery; AO = aorta; RV = right ventricle; LV = left ventricle.
Several Fontan-type operations exist; they connect either the right atrium or the caval veins with the pulmonary arteries. The latter is called total cavopulmonary connection (Figure 14.9).
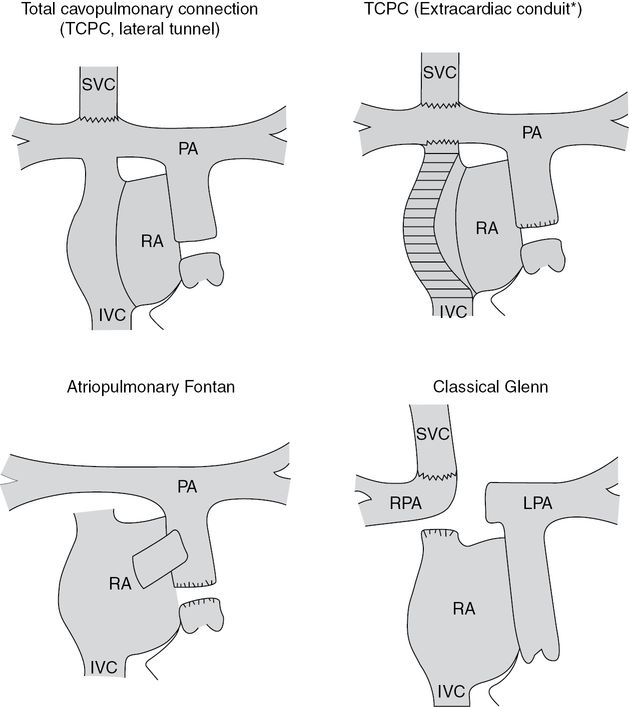
Various Fontan-type operations exist as palliative operations for patients with univentricular hearts; in all of them, systemic venous return is diverted to the lung without interposition of a subpulmonary ventricle; the operations differ regarding the type of connection between the systemic veins and the pulmonary arteries; RPA = right pulmonary artery; SVC = superior vena cava; LPA = left pulmonary artery; RA = right atrium; IVC = inferior vena cava; PA = pulmonary artery; TCPC = total cavo-pulmonary connection.
All “Fontan patients” are at risk of various complications related to surgery and/or abnormal circulatory physiology that persists after surgery (“low cardiac output status”):
1. Atrial arrhythmias related to scarring from surgery or to atrial distension from high venous pressure are common and can cause profound hemodynamic deterioration.
2. Thrombotic diathesis is present as a consequence of sluggish blood flow in the systemic venous pathways and/or in the right atrium.
3. Impairment of ventricular function is part of the “natural” history in these patients, especially if the systemic ventricle has right ventricular morphology.
4. Cyanosis can result from persistent systemic venous to left-sided bypass tracts, leading to right to left shunting.
Although data are limited, it can be assumed that the overall risk of maternal complications for women with single ventricle physiology palliated with a Fontan-type procedure is moderate, but there is a high risk of miscarriage.[45] Legitimate concerns mainly relate to the effect of pregnancy on ventricular function, a prothrombotic circulatory system, and a tendency to poorly tolerated atrial arrhythmias.
The risks of pregnancy mainly relate to the woman’s functional status, her history of arrhythmias, her ventricular function, and the degree of cyanosis.[45] The maternal risk is lower if the woman is in NYHA class I or II, ventricular function is preserved, there is no cyanosis at rest or during exercise, and there is no history of arrhythmia.
Prepregnancy evaluation should include CMR and cardiopulmonary exercise testing to assess ventricular function, venous pathway and pulmonary artery anatomy, and functional status, as well as the woman’s ability to increase cardiac output and preserve oxygen saturation during exercise.
Affected women should have the high risk of miscarriage and preterm delivery explained to them carefully before pregnancy. As the 20-year survival rate following a Fontan operation is 84–87%,[46,47] it is important to discuss maternal long-term prognosis during preconception counseling because the woman’s limited life expectancy is clearly likely to have a major impact on the long-term care of her child.
Close cardiovascular monitoring is mandatory throughout pregnancy, delivery, and the postpartum period. Monitoring should focus on the early signs of heart failure, arrhythmia, thromboembolic complications, and cyanosis.
Patients with the Fontan circulation should be considered for some form of antithrombotic therapy in pregnancy, either full anticoagulation with LMWH, prophylactic weight-adjusted LMWH, or aspirin.[7,48] Depending on the woman’s clinical status, inpatient bed rest and oxygen supplementation should be considered during the third trimester. If bed rest is necessary, anticoagulation with LMWH should be administered instead of aspirin for thromboprophylaxis.
Growth of the fetus should be monitored frequently throughout pregnancy.
Vaginal delivery usually carries the lowest risk of complications such as hemorrhage, thromboembolism, and infection. Epidural anesthesia has minimal effects on hemodynamic performance and may be introduced early in order to avoid pain and anxiety leading to an additional increase in cardiac output during delivery. A lateral position of the woman is preferable to avoid hypotension from uterus compression of the vena cava and the aorta, particularly during labor. Dehydration should be avoided.
After delivery, the risk of thromboembolic complications persists and anticoagulation with LMWH should be continued until the woman is fully mobile, or for at least a week if a cesarean section has been necessary.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


