12 Malignant Changes Synonyms: not otherwise specified (NOS). Invasive ductal carcinoma (IDC) is defined as a malignant tumor of the breast partially or wholly lacking specific histological differentiation patterns and therefore not falling into any of the other categories for invasive mammary carcinoma. The tumor cells initially develop in the terminal ducts, and tumor cells infiltrate the basal membrane continuously or in several distinct locations. Invasive ductal carcinomas possess a strong fibrotic component. In addition, an extensive intraductal component, which can comprise an area more than one-quarter the size of the primary invasive tumor, is often present. Isointense signal in comparison with the surrounding breast parenchyma and therefore no specific changes allowing demarcation of lesion when located within breast parenchyma (Fig. 12.1b). Lesions surrounded by adipose tissue are seen as hypointense, occasionally round (Fig. 12.3b) or ovoid, more often spiculated masses (Fig. 12.2b). The spatial resolution of MR mammography does not allow the depiction of pleomorphic microcalcifications. Typically intermediate or slightly decreased signal intensity in comparison with the surrounding breast parenchyma. Occasional hyperintense zone of peritumoral edema (Figs. 12.2c, 12.3c). Tumor matrix shows increased signal intensity (water content) in ~20% of cases (Fig. 12.8b). High signal intensity in the center of the tumor if central necrosis is present (Fig. 12.7b). Enhancement is obligatory. Depending upon size, lesions are focal (Fig. 12.1a,d), mass (Figs. 12.3a, d, 12.4a, d, 12.6b), or rarely nonmasslike (Fig. 12.2a,d). Tumor borders are often ill-defined or spiculated (Figs. 12.2d, 12.8a). Enhancement is inhomogeneous (Fig. 12.3d), or stronger in the tumor periphery (rim-enhancement) (Fig. 12.7a). Peritumoral linear enhancement is a potential indication of an EIC (Fig. 12.5a,d). Time–signal intensity analysis is recommended for mass lesions. Initial signal increase is often intermediate or rapid (> 100%) (Figs. 12.3e, 12.4e). A slow initial signal increase is very rare (< 5%). The postinitial signal course is often an unspecific plateau or suspicious washout (Figs. 12.3e, 12.4e). A persistent signal increase is very rare. Nodular carcinomas never display “late enhancement.” This expression originated from historic measurement protocols and there is no pathophysiological explanation for such enhancement dynamics.
Invasive Ductal Carcinoma
 IDC is the most common and morphologically heterogeneous malignant tumor of the breast.
IDC is the most common and morphologically heterogeneous malignant tumor of the breast.
 MR Mammography: Invasive Ductal Carcinoma
MR Mammography: Invasive Ductal Carcinoma
T1-Weighted Sequence (Precontrast)
T2-Weighted Sequence
T1-Weighted Sequence (Contrast Enhanced)
Invasive ductal carcinoma: General information | |
Incidence: | Most common form of invasive breast cancer (65%–75%). |
Age peak: | All ages, peak between 50th and 60th years. |
Grading: | Histological grade: well-differentiated (G1), intermediate (G2), poorly differentiated (G3). |
Prognosis: | Dependent upon size, grading, N-stage, and receptor status among other factors. |
Multifocality: | 15%. |
Bilaterality: | 5%. |
Findings | |
Clinical: | Small tumors are clinically occult. Average diameter of palpable tumor: 2.3 cm. Typical criteria of breast cancer (usually not early signs): hard, ill-defined mass, poorly movable, indolent, skin or nipple retraction. |
Mammography: | Tumors not associated with microcalcifications located within dense breast tissue are often mammographically occult. Tumors associated with microcalcifications or located within lipomatous breast areas are reliably detected. Typical mammographic criteria of breast cancer: irregular shape, ill-defined or spiculated, high radiodensity. Associated clustered, pleomorphic microcalcifications in ~30% of all invasive breast cancers. |
Ultrasonography: | Small tumors are often sonographically occult. Tumor detectability increases with size from a minimum diameter of approx. 5 mm. Examples of typical sonographic criteria of breast cancer: ill-defined, hypoechoic mass with hyperechoic margins. Central or peripheral posterior acoustic shadowing. |
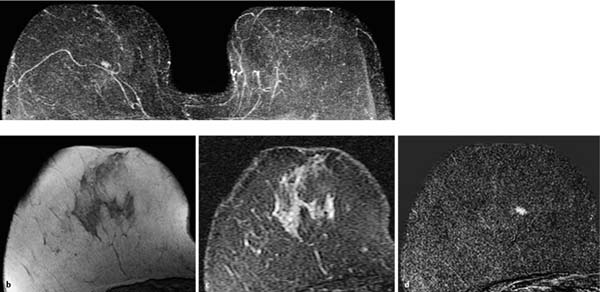
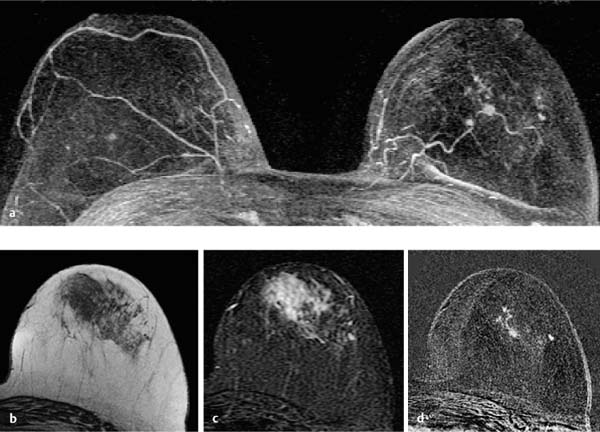
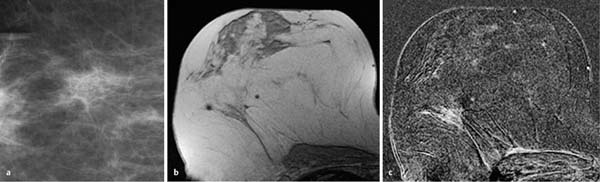
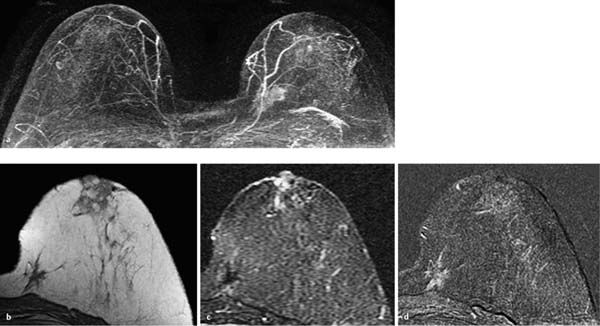
Fig. 12.1a–d Invasive ductal carcinoma of 4 mm diameter.
a Subtraction MIP: hypervascularized focus in the right breast. Unremarkable findings in the left breast.
b T1w precontrast slice image (right breast): unremarkable findings.
c IR T2w slice image (right breast): slightly decreased signal intensity (water content).
d Early subtraction slice image: oval, ill-defined focus. TICs are unspecific (not shown).
Histology: IDC (pT1a).
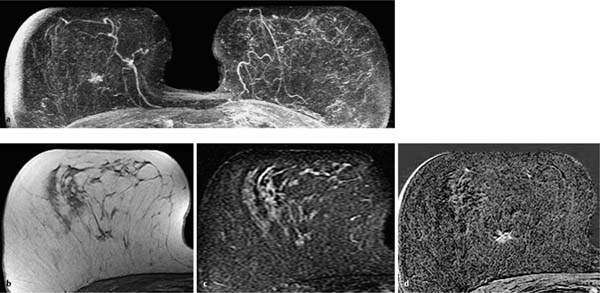
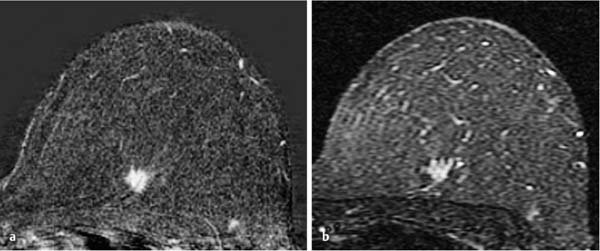
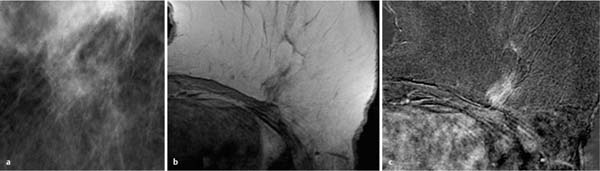
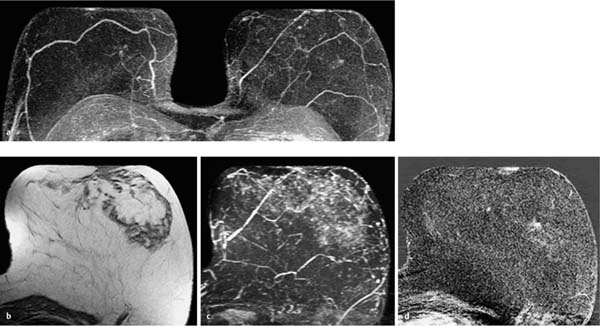
Fig. 12.2a–d Invasive ductal carcinoma (pT1b).
a Subtraction MIP: spiculated hypervascularized lesion in the center of the right breast. Otherwise unremarkable findings.
b T1w precontrast slice image (right breast): vague architectural distortion within the lipomatous breast area.
c IR T2w slice image (right breast): intermediate signal intensity in the center of the lesion.
d Early subtraction slice image (right breast): improved visualization of nonmasslike, spiculated enhancement. TICs unspecific, as is typical in nonmasslike lesions (not shown).
Histology: IDC (pT1b).
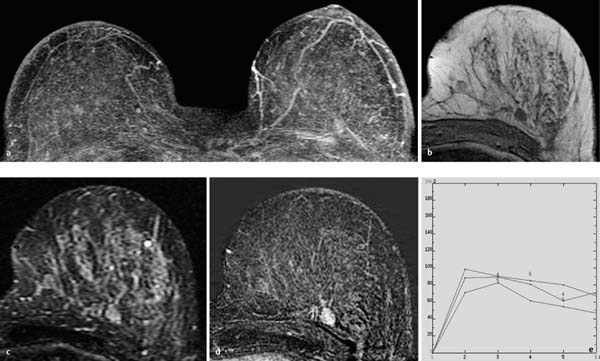
Fig. 12.3a–e Invasive ductal carcinoma (pT1b).
a Subtraction MIP: stippled enhancement pattern in both breasts. Vague, enhancing, nodular lesion near the chest wall in the left breast.
b T1w precontrast slice image (left breast): round, well-circumscribed mass with signal intensity equivalent to that of breast parenchyma.
c IR T2w slice image (left breast): intermediate signal intensity. The image gives the impression that the lesion may have internal septations.
d Early subtraction slice image (left breast): round, relatively wellcircumscribed mass lesion with inhomogeneous enhancement.
e TIC: early peak with pronounced postinitial washout.
Histology: IDC (pT1b).
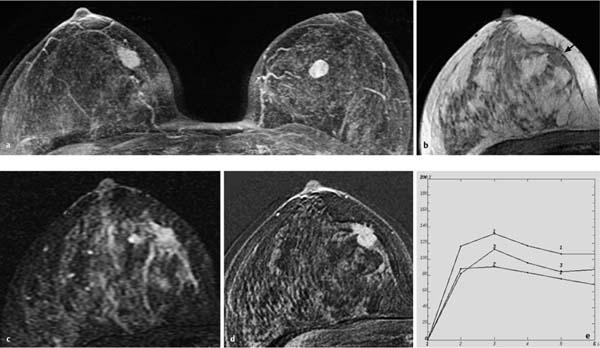
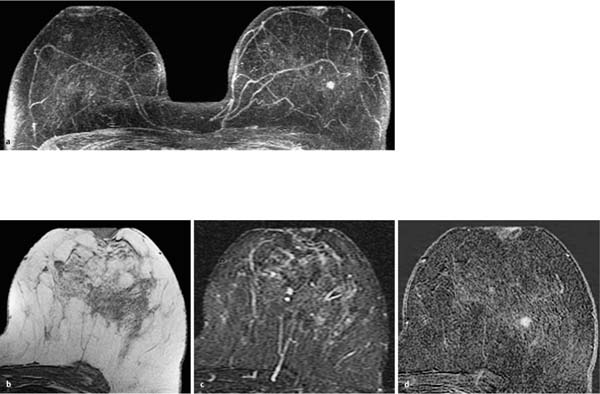
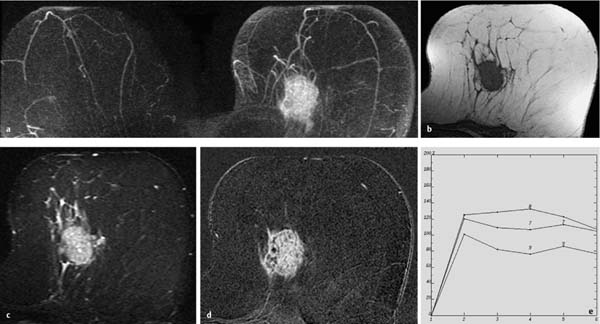
Fig. 12.4a–e Invasive ductal carcinoma (pT2) and fibroadenoma.
a Subtraction MIP: bilateral hypervascularized lesions. Right lesion: 12 mm, lobulated, relatively well-circumscribed lesion with inhomogeneous enhancement (carcinoma). Left lesion: 10 mm, oval lesion with inhomogeneous enhancement (fibroadenoma).
b T1w precontrast slice image (right breast): vague, space-occupying lesion with signal intensity equivalent to that of breast parenchyma (arrow).
c IR T2w slice image (right breast): increased signal intensity.
d Early subtraction slice image (right breast): irregular, partially illdefined, hypervascularized lesion.
e TIC: early peak with pronounced postinitial washout.
Histology: IDC (pT1c, right breast). Fibroadenoma (left breast).
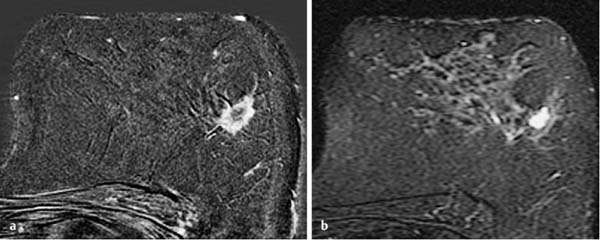
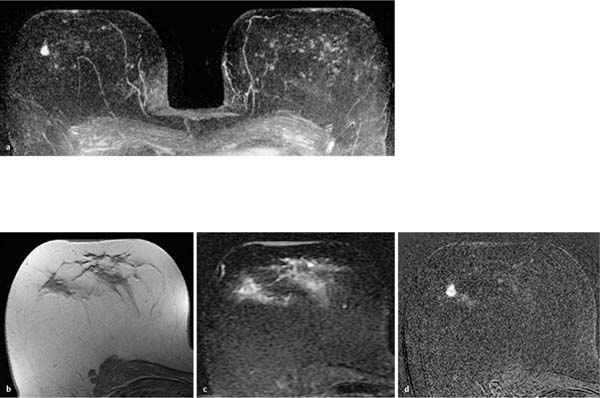
Fig. 12.5a–d Invasive ductal carcinoma with extensive intraductal component (EIC).
a Subtraction MIP: hypervascularized focus of 3 mm diameter in the center of the left breast. Adjacent linear-branching enhancement. Unremarkable findings in the right breast.
b T1w precontrast slice image (left breast): unremarkable findings.
c IR T2w slice image (left breast): unremarkable findings.
d Early subtraction slice image (left breast): Improved visualization of peritumoral ductal enhancement correlating to EIC. TICs are unspecific (not shown).
Histology: IDC (pT1a) + EIC.
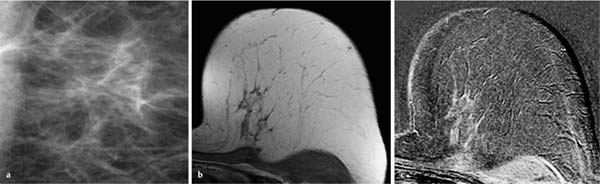
Fig. 12.6a, b Invasive ductal carcinoma in a location that is easy to overlook.
a T1w precontrast slice image (left breast): presumptive extension of breast parenchymal body far into the axillary tail.
b Early subtraction slice image with swapped phase-encoding gradient (left breast): now discernible, oval, ill-defined lesion with accentuated enhancement in the periphery of the lesion. Dynamic examination was performed using the zebra protocol (see Chapter 3) for better assessment of the lateral breast aspects.
Histology: IDC (pT1c).
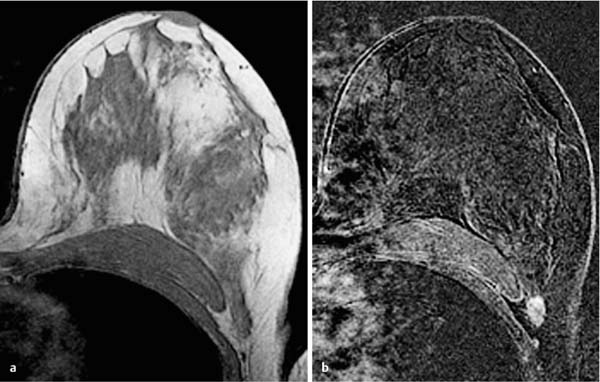
Fig. 12.7a, b Invasive ductal carcinoma with rim-enhancement.
a Early subtraction slice image (left breast): classical breast carcinoma with ill-defined borders, rim-enhancement, and peritumoral linear extensions as an indication of concomitant EIC.
b IR T2w slice image (left breast): pronounced necrosis in tumor center with high signal intensity equivalent to that of a cyst.
Histology: IDC (pT1c).
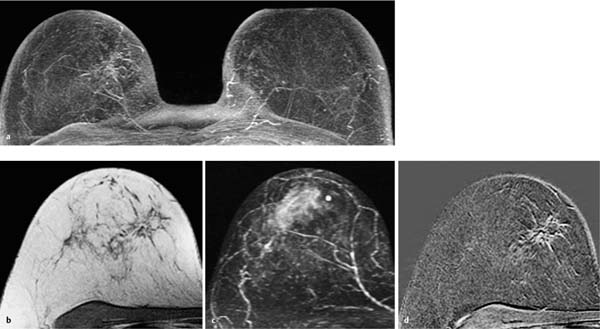
Fig. 12.8a, b Invasive ductal carcinoma with high water content.
a Early subtraction slice image (left breast): hypervascularized, spiculated breast carcinoma.
b IR T2w slice image (left breast): high signal intensity of the entire tumor matrix.
Histology: IDC (pT1b).
Invasive Lobular Carcinoma
Synonym: infiltrating lobular carcinoma.
Invasive lobular carcinoma (ILC) is characterized histologically by a desmoplastic stromal reaction. Tumor cells are small, monomorphic, and round, and grow in a single-file (“Indian-file”) or targetoid pattern encircling ducts that are dispersed throughout the fibrous matrix. Some tumors contain so-called “signet ring cells” (tumor cells with central mucoid globules) in addition to the small cells. Growth patterns are differentiated into diffuse and nodular types.
 Invasive lobular carcinomas (ILC) with a diffuse growth pattern have the greatest false-negative detection rate in all breast imaging techniques.
Invasive lobular carcinomas (ILC) with a diffuse growth pattern have the greatest false-negative detection rate in all breast imaging techniques.
 MR Mammography: Invasive Lobular Carcinoma
MR Mammography: Invasive Lobular Carcinoma
T1-Weighted Sequence (Precontrast)
Diffuse type. When located in lipomatous areas of the breast: linear-streaky, isointense architectural distortion that respects the fat lamellae and is not space-occupying (Figs. 12.9b, 12.10b, 12.11b, 12.12b). No characteristic changes when located within the breast parenchyma.
Nodular type. When located in lipomatous areas of the breast: nodular, an isointense lesion (Fig. 12.14b). When located within breast parenchyma, often unremarkable findings (Figs. 12.15b, 12.16b).
T2-Weighted Sequence
Typically intermediate or slightly decreased signal intensity in comparison to the surrounding breast parenchyma (Figs. 12.15c, 12.16c). Occasional hyperintense zone of peritumoral edema (Fig. 12.13c).
T1-Weighted Sequence (Contrast Enhanced)
Diffuse type. Nonmasslike lesion that respects the fat tissue boundaries and displays a linear-streaky, sometimes “Indianfile” enhancement (Figs. 12.9c, 12.10c, 12.11c, 12.12d). Enhancement pattern correlates with histology and the often very discrete morphological changes seen on mammography (Figs. 12.9a, 12.10a, 12.11a). It is not advisable to generate TICs. The ROIs always include nontumor, lipomatous areas, producing TICs that may be misleading.
Nodular type. The nodular type of ILC displays the same features as an IDC: hypervascularized focus (Fig. 12.14a,d) or mass, most often with ill-defined borders (Fig. 12.15a,d) and rim-enhancement (Fig. 12.16a,d). TIC shows intermediate or rapid initial signal increase, often with postinitial plateau or washout.
Invasive lobular carcinoma: General information | |
Incidence: | Second most common form of invasive breast cancer (10%–15%). |
Age peak: | All ages, peak between 40th and 60th years. |
Grading: | Histological grade: well-differentiated (G1), intermediate (G2), poorly differentiated (G3). |
Prognosis: | Dependent upon size, grading, N-stage, and receptor status among other factors. |
Multicentricity: | > 20%. |
Multifocality: | 10%–20%. |
Coincidence: | Association with LCIS or ADH. |
Findings | |
Clinical: | Often clinically occult. Tumors with large volume may cause palpable firmness. Nodular types may present as palpable mass. |
Mammography: | Diffuse type: architectural distortion, localized changes in parenchymal density, structural irregularities. Rarely tumor-associated microcalcifications (< 10%). Nodular type: may show more typical signs of malignancy. Often focal, ill-defined hyperdensity. Detection rate is higher when located in lipomatous breast areas than within dense parenchyma. |
Ultrasonography: | Diffuse type: often no specific changes. Occasionally diffuse echo-alterations with multiple fine posterior acoustic shadowings. Nodular type: focal lesions with typical characteristics of malignancy. |
Fig. 12.9a–c Invasive lobular carcinoma, diffuse type.
a Enlarged detail mammography image: typical nonmass density.
b T1w precontrast slice image (right breast): susceptibility artifacts due to prior surgery. Vague linear-streaky area within lipomatous breast tissue.
c Early subtraction slice image (right breast): regional, linear nonmasslike enhancement.
Histology: ILC (pT1c).
Fig. 12.10a–c Invasive lobular carcinoma, diffuse type.
a Enlarged detail of mammography (Mx) image: localized architectural distortion (nonmasslike lesion).
b T1w precontrast slice image (left breast): linear-streaky, nonlipomatous area corresponding to mammography.
c Early subtraction slice image (left breast): Regional, linear nonmasslike enhancement excluding the lipomatous areas.
Histology: ILC (pT1c).
Fig. 12.11a–c Invasive lobular carcinoma, diffuse type.
a Enlarged detail of mammography (Mx) image: localized architectural distortion (nonmasslike lesion).
b T1w precontrast slice image (left breast): nonlipomatous, tissue streaks within lipomatous breast tissue.
c. Early subtraction slice image (left breast): linear nonmasslike enhancement (“Indian-file”) excluding the lipomatous areas.
Histology: ILC (pT1c).
Fig. 12.12a–d Invasive lobular carcinoma, diffuse type.
a Subtraction MIP: reticular enhancement in the medial aspect of the right breast.
b T1w precontrast slice image (right breast): corresponding linearbranching architectural distortion within intramammary lipomatous tissue.
c IR T2w slice image (right breast): unremarkable findings in the medial aspect of the right breast.
d Early subtraction slice image (right breast): improved visualization of the nonmasslike, linear-branching enhancement which excludes the lipomatous areas. TICs are unspecific and not informative (not shown).
Histology: ILC (pT2).
Fig. 12.13a–d Invasive lobular carcinoma, diffuse type.
a Subtraction MIP: irregular area of enhancement in the medial aspect of the left breast.
b T1w precontrast slice image (left breast): irregular shape is wellvisualized within lipomatous tissue.
c IR T2w slice image (left breast): unusual signal suppression within the tumor in this fat-suppressed image. Slight peripheral edema.
d Early subtraction slice image (left breast): nonmasslike enhancement pattern within the tumor is evident. Topographical closeness to the chest wall. TICs are unspecific and not informative (not shown).
Histology: ILC (pT2).
Fig. 12.14a–d Invasive lobular carcinoma, nodular type.
a Subtraction MIP: barely discernible focus in the centrolateral aspect of the left breast.
b T1w precontrast slice image (left breast): As expected, unremarkable findings.
c IR T2w slice image (left breast): Unremarkable findings.
d Early subtraction slice image (left breast): improved visualization of hypervascularized focus with fine-linear peritumoral enhancement. TICs are unspecific (not shown).
Histology: ILC (pT1a).
Fig. 12.15a–d Invasive lobular carcinoma, nodular type.
a Subtraction MIP: ill-defined mass lesion of 5 mm diameter in the central aspect of the left breast.
b T1w precontrast slice image (left breast): unremarkable findings.
c IR T2w slice image (left breast): no correlating tumor findings.
d Early subtraction slice image (left breast): improved visualization of the lobulated, hypervascularized mass lesion with possible internal septations. TICs are unspecific with initial increase > 100% and postinitial plateau (not shown).
Histology: ILC (pT1b).
Fig. 12.16a–d Invasive lobular carcinoma, nodular type.
a Subtraction MIP: lobulated, partially ill-defined mass lesion of 7 mm diameter in the right breast. Multiple harmless foci in the left breast.
b T1w precontrast slice image (right breast): unremarkable findings.
c IR T2w slice image (right breast): unremarkable findings.
d Early subtraction slice image (right breast): improved visualization of the ill-defined borders and rim-enhancement (decreased perfusion in tumor center). TICs are unspecific with initial increase 80% and postinitial plateau (not shown).
Histology: ILC (pT1b).
Medullary Carcinoma
The histopathological features that define the typical form of medullary carcinoma are a tendency for tumor cells to grow in broad sheets without distinct cell borders (syncytial growth), high nuclear grade (pleomorphic nuclei with prominent nucleoli, usually accompanied by numerous mitotic figures), well-circumscribed tumor margins, and an intense lymphoplasmacytic reaction around and within the tumor. Carcinomas that have most, but not all, of these microscopic features are referred to as atypical medullary carcinomas. The median size of medullary carcinomas is 2–3 cm. Lesions larger than 5 cm tend to show central tumor necrosis and calcification.
 MR Mammography: Medullary Carcinoma
MR Mammography: Medullary Carcinoma
T1-Weighted Sequence (Precontrast)
Round, oval, or lobulated, well-circumscribed (Fig. 12.17b) or partially ill-defined (Fig. 12.18b), isointense or hypointense mass. Difficult to detect when located within breast parenchyma.
T2-Weighted Sequence
Signal intensity is occasionally intermediate (Fig. 12.18c), often increased (Fig. 12.17c) relative to breast parenchyma. Occasional peritumoral hyperintense edematous zone (Figs. 12.17c, 12.18c).
T1-Weighted Sequence (Contrast Enhanced)
Enhancement typical of malignancy. Often round, oval, or lobulated mass. Boundaries are often partially well-circumscribed, but are almost always partially ill-defined (Fig. 12.17d) and/or microlobulated (Fig. 12.18d). Rim-enhancement is rare. TIC analysis shows an intermediate, more often a rapid initial signal increase (Figs. 12.17e, 12.18e). A slow initial increase is very rare. The postinitial signal course often displays a washout (Figs. 12.17e, 12.18e). A postinitial persistent signal increase is very rare.
Medullary carcinoma: General information | |
Incidence: | Rare; ~1% of all breast cancers. Often associated with BRCA1 gene mutations. |
Age peak: | All ages. Usually under 50 years, common under 35 years. |
Prognosis: | Typical form: more favorable than that of invasive ductal carcinoma. Atypical form: identical with that of invasive ductal carcinoma. |
Findings | |
Clinical: | Small tumors are clinically occult. Larger tumors are often well-circumscribed masses. |
Mammography: | Tumors located within dense parenchyma are often mammographically occult. Good detection of tumors located within lipomatous breast tissue. Rare microcalcifications. Larger tumors appear as well-circumscribed, round or lobulated mass lesions. Occasionally ill-defined borders due to lymphocytic infiltrates. |
Ultrasonography: | Small tumors are sonographically occult. Larger tumors appear round, well-circumscribed, and hypoechoic. |
Because the typical appearance of a medullary breast carcinoma can make it difficult to differentiate from a benign breast mass lesion (e.g., fibroadenoma), it has a special significance in breast cancer diagnostics. Along with the mucinous carcinomas, medullary carcinomas are unlike other carcinomas in their tendency to have a high endotumoral water content (T2w signal). | |
Fig. 12.17a–e Medullary carcinoma.
a Subtraction MIP: large oval tumor in the left breast.
b T1w precontrast slice image (left breast): round, predominantly well-circumscribed mass.
c IR T2w slice image (left breast): high signal intensity of the entire lesion with punctate areas of very high signal intensity. Peritumoral edema.
d Early subtraction slice image (left breast): improved visualization of the partially ill-defined borders and very inhomogeneous enhancement.
e TIC: early peak with postinitial washout.
Histology: medullary carcinoma (pT2).