On successfully completing this topic, you will be able to:
understand the definition and causes of major obstetric haemorrhage
recognise and manage maternal collapse caused by obstetric haemorrhage
understand the pharmacological and surgical options for the treatment of major obstetric haemorrhage
make a plan to facilitate optimum management in your environment
understand the concerns to be discussed with a woman declining blood and blood products during pregnancy.
Introduction
Major haemorrhage remains a leading cause of maternal mortality despite modern improvements in obstetric practice and transfusion services. In addition, all ‘near miss’ audits, in developed as well as developing countries, show major haemorrhage to be one of the leading causes of severe maternal morbidity.
Complications of haemorrhage associated with first-trimester bleeding due to miscarriage or ectopic pregnancy are not within the remit of the MOET course, although most general principles will apply.
Maternal mortality and incidence of major haemorrhage
The MBRRACE confidential enquiry 2009–2012 showed a slight (but not statistically significant) rise in deaths due to haemorrhage, but all cases could have had improvements in care.1 In particular, a lack of routine observations in the postpartum period and failure to appreciate that significant (concealed) bleeding was occurring led to a lack of appropriate action including no early senior involvement. The report restates previous comments that all units should have procedures in place for the identification and management of major obstetric haemorrhage, and that regular skills training sessions should take place. It also draws attention to the importance of the use of MEOWS charts for 24 hours after CS, with immediate action if abnormalities are detected. A further comment is the importance of detecting and treating anaemia antenatally, thereby mitigating the effects of major obstetric haemorrhage should it occur.
Mortality statistics give no data on the incidence of major haemorrhage or on the maternal and fetal morbidity associated with haemorrhage and transfusion. Specific complications include: transfusion-related reactions and infections; genital tract trauma; loss of fertility following hysterectomy; and the rare Sheehan’s syndrome (hypopituitarism).
Definition and epidemiology
Major obstetric haemorrhage is defined as the loss of more than 1000 ml blood as either antepartum or postpartum loss. Antepartum haemorrhage is often followed by postpartum haemorrhage.
Considerable problems are recognised in the accurate measurement of blood loss, and a definition based on volume alone has some shortcomings. Both visual and measured loss can be highly inaccurate, and loss from placental abruption, uterine rupture or post-CS may be partially or completely concealed. Underestimation of blood loss may delay active steps being taken to prepare for or prevent further bleeding.
Major causes of obstetric haemorrhage(primary or secondary)
Resulting initially in hypovolaemia:
uterine atony (multiple causes)
placenta praevia
retained or adherent placenta or placental fragments
genital tract injury including broad ligament haematoma
uterine rupture
uterine inversion
uterine anatomical abnormalities, e.g. multiple fibroids.
Associated with coagulation failure:
placental abruption
pre-eclampsia
septicaemia/intrauterine sepsis
retained dead fetus
amniotic fluid embolus
incompatible blood transfusion
existing coagulation abnormalities.
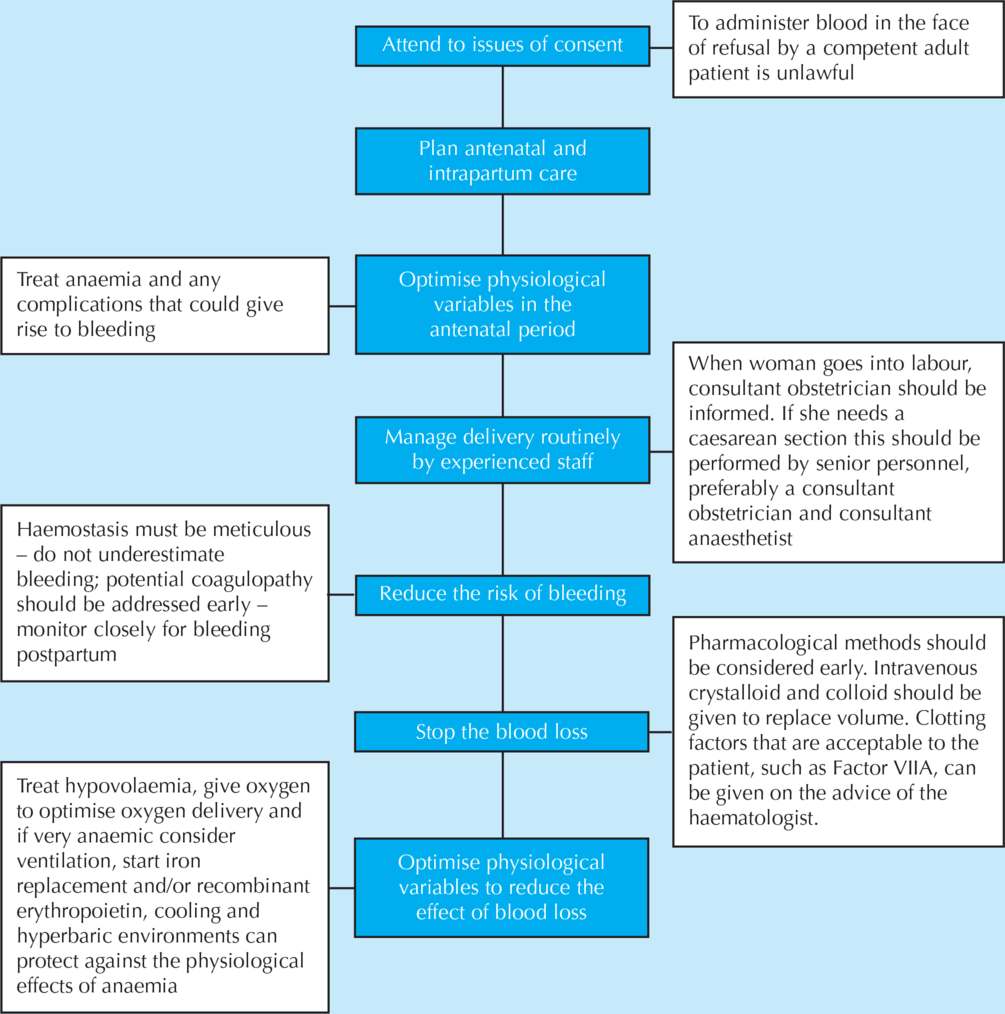
Guidelines for the management of major haemorrhage
The recommendations of the Royal College of Obstetricians and Gynaecologists (RCOG) Greentop Guideline 52, Prevention and Management of Postpartum Haemorrhage, updated in 2011, form the basis of this chapter.2
Most obstetric units in the UK now have a ‘Major Obstetric Haemorrhage’ procedure, as recommended by CMACE and endorsed by the RCOG. This now forms part of the requirements of the Clinical Negligence Scheme for Trusts (CNST). This protocol should be readily available on the delivery suite, including details of senior staff to be contacted in obstetrics, anaesthetics and haematology. Its use should also be subject to local audit. Blood loss of greater than 1000 ml or the recognition of maternal signs of shock in the absence of large visible loss should prompt the initiation of the protocol set out in the major haemorrhage guideline.
Practical management
The cornerstones in the management of major obstetric haemorrhage are early recognition, the restoration of the circulating blood volume and oxygen-carrying capacity, and the prevention of further loss. Failure to maintain adequate tissue perfusion leads to loss of vital organ function, and care becomes increasingly more complex.
Recognition of haemorrhage
Not all haemorrhage is revealed and large volumes of blood can be lost from the circulation while remaining concealed in the peritoneal cavity, the uterus, the retroperitoneal space or the ischiorectal fossa. A high index of suspicion should be maintained while assessing patients with abdominal pain, unusual levels of perineal discomfort post delivery, fainting and any signs of shock as listed below. The classes of increasing severity of blood loss are described in detail in Chapter 5 on shock.
Maternal signs of shock
These include (see also Chapter 5):
tachycardia (on occasions a normal pulse rate or bradycardia can be seen – beware the patient on beta-blockers)
tachypnoea
poor peripheral perfusion
confusion or unresponsiveness
oliguria
hypotension
requiring more than 2 units of colloid to maintain blood pressure
unexplained metabolic acidosis.
Management
Management involves various elements, all of which need to be undertaken simultaneously:
communication and documentation
resuscitation
fluid replacement
arresting the bleeding and obstetric intervention
monitoring and investigation
anaesthetic management.
Communication (‘call for help’) and documentation
Institute the following:
call senior midwife, obstetric and anaesthetic registrars
inform consultant obstetrician and anaesthetist
inform blood bank technician and consultant haematologist (most units will have an agreed form of words, e.g. ‘Major obstetric haemorrhage’ to which the laboratory will respond)
call porter for delivery of specimens and blood products
allocate one member of resuscitation team as team leader and one to record events, fluids, drugs and vital signs.
Ongoing written and verbal communication between senior staff is essential. This may involve the assistance of other specialists, such as intensive care specialists, vascular surgeons or radiologists.
Resuscitation
Assess airway and breathing (see Chapter 8 for details). Conscious level and airway control usually improve rapidly once the circulating volume is restored. If the mother is shocked and there is no means of measuring saturations (or if saturations are reduced ≤95%), then administer high-flow oxygen.
Evaluate the circulation (see Chapters 5 [Shock] and 7 [Intravenous access and fluid administration] for details).
Correction of circulation within primary survey:
establish two 14-gauge intravenous lines
take 20 ml blood for diagnostic tests
commence 2 litres of warmed crystalloid intravenously
left lateral position should be adopted, to minimise the effects of aortocaval compression in an undelivered woman
lateral tilt with a wedge (or manual displacement of the uterus by an assistant) should continue to be used when obstetric procedures are in progress
stop any epidural infusion pending review by an anaesthetist; compensatory lower-limb vasoconstriction will be limited, so the effects of positioning may be more marked
head-down tilt can be used as a short-term measure to improve venous return, but this may compromise respiration.
Fluid, blood product, cell salvage and clotting factor replacement is all described in detail in Chapters 5 and 7 and is not repeated here.
Diagnostic tests
These include:
full blood count
coagulation screen including fibrinogen
cross match 4–6 units group specific if urgent
baseline renal function
blood gas.
Regular checks of the haematocrit, clotting studies and blood gases will help guide resuscitation.
Immediate bedside testing of haematocrit
This may guide the need for red blood cells, but if haemodilution has not yet occurred (as is the case with acute haemorrhage) then the result can be falsely reassuring. In addition, the Serious Hazards of Transfusion Scheme (SHOTS) report states that, ‘Blood gas machines must not be used for haemoglobin estimations on which to base a transfusion request. All point of care testing devices for haemoglobin estimation must be fully validated and both internal quality control and participation in external quality assurance schemes must be ensured.’ It is possible to overenthusiastically transfuse red blood cells, forgetting that these are concentrated red cells, not whole blood, and cannot be used as full replacement for loss on a volume-for-volume basis.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree