Hearing Loss and Cochlear Implantation in Children
Types of hearing impairment
Auditory neuropathy was first recognized as an entity in the 1990s, and was renamed ANSD in 2008 [1]. It is diagnosed when there is a discrepancy between cochlear and neural function. In ANSD, the outer hair cells function normally, but sound is not properly transmitted from the outer hair cells to the auditory cortex. This dysfunction is thought to be caused by desynchronized action potentials in the auditory nerve. Presentation varies widely. Children with ANSD may have hearing thresholds that range from normal to profound levels on audiometric testing; however, they usually exhibit disproportionately poor speech perception abilities for their degree of hearing loss. Some patients with ANSD have only very mild impairment, whereas others present as functionally deaf. Some have impairment that fluctuates in severity. Children with ANSD typically have difficulty hearing in noise. ANSD children may hear words but have difficulty understanding what is being said.
The cochlear implant
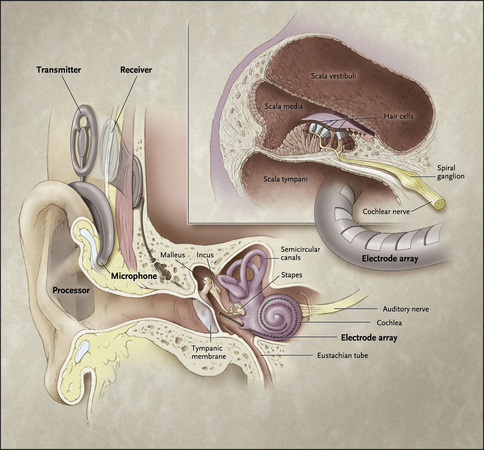
The device has internal and external parts (Figs. 1 and 2). The external parts include a microphone, speech processor, and transmitter. The microphone picks up sounds from the environment. The speech processor digitizes the sounds and then these signals are sent via the transmitter to the internal portion of the device, which is surgically implanted. The receiver, which is under the scalp, picks up the signals and delivers them to the electrode array that is inserted into the cochlea. The electrodes along the array directly stimulate the auditory nerve. The cochlea is tonotopically organized: high frequencies are picked up at the base of the cochlea, and low frequencies are detected at the apex of the snail-shaped organ.
Candidacy for implantation
All cochlear implant candidates should receive a trial of binaural amplification, fit appropriately, according to American Academy of Audiology pediatric hearing aid fitting guidelines. A 3- to 6-month trial of hearing aid use, in conjunction with intensive therapy, is typically necessary to evaluate benefit. To determine whether the hearing aids are providing enough useful auditory input to allow for communication and language acquisition, open-set aided speech recognition testing is performed in older children. In younger children, closed-set speech recognition tests may provide useful information. The FDA criteria vary slightly for different manufacturers’ devices, but speech discrimination scores of 50% to 60% or below with optimal amplification are currently required for candidacy. Receiving benefit from amplification no longer disqualifies one from receiving a cochlear implant, because most patients who are able to recognize approximately 50% of words using hearing aids have now been shown to generally have even higher speech recognition scores postimplantation, compared with their best preoperative scores [2,3]. For children with asymmetric hearing loss, the literature supports continued use of a hearing aid in the better-hearing, nonimplanted ear, with implantation of the worse-hearing ear. Greater residual hearing at implantation and shorter duration of deafness seem to be associated with improved language outcomes; conversely, poorer outcomes seem to be associated with prolonged use of hearing aids before implantation [4,5].
A situation in which the decision to implant is often expedited is when deafness occurs secondary to bacterial meningitis, particularly if Streptococcus pneumoniae is the pathogen. In these cases, because of the powerful inflammatory response triggered by the pneumococcal cell wall, rapid fibrosis and ossification of the cochlea may occur, often beginning within several weeks of the infection [6]. Ossification can lead to a more difficult surgery, incomplete electrode insertion, and poorer outcomes. For this reason, many advocate a more aggressive time line for surgery, including implantation under the age of 12 months if necessary. This recommendation is a matter of some debate, because hearing recovery in postmeningitic patients has been reported; for advocates of the watchful waiting approach, serial MRI scanning is performed, with urgent cochlear implantation for signs of ossification.
The age of implantation continues to decrease. Understanding of auditory development and brain plasticity has given physicians the impetus to implant children at a younger age, and earlier implantation has been correlated with improved communication outcomes [4,7–12]. A narrow window of time exists during development, known as a sensitive period, within which the central auditory cortex must be stimulated by sound if it is to develop normally. If the auditory cortex is deprived of stimulus during this period, it will reorganize to be used for other purposes. For example, studies using functional MRI have shown that in deaf individuals, areas of the auditory cortex were activated by visual stimuli; this was not seen in normal hearing controls [13]. Because of this reallocation of brain function, once the sensitive period has passed, auditory development becomes much more challenging.
Sharma and colleagues [13,15] looked to assess the effect of age at cochlear implantation on auditory development and found that the brain is maximally plastic for the first 3.5 years, and that the optimal time for implantation is within this period. Plasticity then declined, but was found to remain in varying degrees until approximately 7 years of age, which marked the end of the sensitive period. Some studies have shown that different language skills may have different sensitive periods. For example, one study concluded that there might be an earlier sensitive period for vocabulary-building skills than for speech perception skills [7].
Research has been directed toward further narrowing down the optimal age for implantation. No consensus has been reached, but most literature supports that younger is better [7,10,16]. The FDA currently approves cochlear implantation down to 12 months of age, but some centers have been implanting even younger infants.
As understanding of ANSD has grown, cochlear implants have also become recognized as a viable option for some children with this disorder. One might question the efficacy of a cochlear implant in patients with abnormal auditory nerve conduction, but it is postulated that the discrete electrical signals produced by a cochlear implant may be more effective at eliciting a synchronous neural response than acoustic stimuli. Several studies report that children with ANSD derive benefit from cochlear implantation, and some report outcomes comparable to those of implanted children with SNHL [17–20]. In 2008, a consensus panel recommended that cochlear implants should be considered in children with ANSD, regardless of audiometric threshold, if progress is not being made in language development, despite appropriate amplification.
However, children with ANSD are a heterogeneous group, and not all patients with ANSD should be implanted. Research has shown that for individuals with a cochlear nerve deficiency or hypoplastic cochlear nerve, outcomes are much poorer [21,22]. This does not mean that this group should automatically be excluded from consideration, but they should be carefully assessed for any auditory sensation, and expectations should be tempered.
There is also a chance that auditory function may improve in ANSD. This has been documented in several studies. In a 2002 study, 50% of children with ANSD and evidence of severe SNHL spontaneously improved, with a mean improvement time of 5.8 months. The children whose ANSD was attributed to hyperbilirubinemia had a greater tendency to improve. These children achieved a stable audiogram at a mean age of 18 months [23]. Thus, caution must be exercised before proceeding with implantation in infants and very young children who carry this diagnosis.
Cochlear implant programs in the past tended to exclude deaf children with other handicaps, but the number of children with other disabilities being implanted is increasing rapidly, and accounts for a large percentage of implant recipients at some centers. For most children, the goal of implantation is spoken language. For children with developmental delays or other disabilities, the goals of implantation may be different. Speech and language outcomes may be limited by cognitive deficits, but these children may still derive benefit from improved use of language, as well as sound awareness, and increased environmental and social connectedness. Communication by nonverbal modalities may still be supported by sound. Mere sound awareness can be useful in terms of social attachment, attention, and safety. Thus, these children should not automatically be excluded from implant candidacy [24,25].
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree