Location
Associated symptoms
Orbital
Diplopia with cranial nerve palsy
Proptosis
Gingival
Gingival hypertrophy
Gum bleeding
Parotid gland
Facial nerve palsy [19]
Nasal cavity
Epistaxis, nasal airway obstruction
Temporal bone
Middle ear
Otitis media, Bell’s palsy [22]
Differential Diagnosis
Hematologic malignancies should be included in the differential diagnosis of head and neck tumors in children, particularly when systemic symptoms are present (Table 27.2).
Table 27.2
Differential diagnosis
Presentation | Inflammatory | Infectious | Neoplastic |
|---|---|---|---|
Systemic symptoms | Rheumatoid arthritis | Tuberculosis EBV Cytomegalovirus (CMV) | Nasopharyngeal carcinoma Small round blue cell tumors (Ewing sarcoma, neuroblastoma, rhabdomyosarcoma) Rhabdoid tumor |
Head and neck mass | Cat scratch disease EBV CMV Tuberculosis Mycobacterium Bacterial lymphadenitis | Nasopharyngeal carcinoma Small round blue cell tumors (Ewing sarcoma, neuroblastoma, rhabdomyosarcoma) Rhabdoid tumor | |
Skin Lesions | Lipoma | Langerhans cell histiocytosis Melanoma Neuroblastoma |
Diagnosis and Evaluation
Physical Examination
Patients with hematologic malignancies can present with hemodynamic instability including tachycardia and hypotension often secondary to infection. Patients are immunosuppressed and infections should be treated with aggressive resuscitation and broad-spectrum antibiotics.
Assess for signs of respiratory distress including increased respiratory rate, stridor, wheezing, and retractions which can result from cervicothoracic masses.
Nasal cavity and oropharynx should be assessed for lesions including mucosal ulceration and granulocytic sarcomas.
Patients presenting with the endemic form of Burkitt lymphoma may present with large masses in the craniofacial bones, most typically the jaw or maxillary bones.
Skin should also be assessed for granulocytic sarcomas as well as for signs of anemia, i.e., pallor and thrombocytopenia including petechiae and excessive bruising.
The patient should have a complete lymph node exam and abdominal exam to assess for lymphadenopathy and hepatosplenomegaly.
Laboratory Data
Standard laboratory studies include a complete blood count (CBC) with differential, coagulation studies (PT/PTT), and type and cross for blood bank. The presence of alterations in the CBC, even in the presence of enlarged lymph nodes, suggests bone marrow involvement and should lead to a bone marrow aspirate and biopsy as diagnostic procedures.
A lumbar puncture for evaluation of cerebrospinal fluid (CSF) cytology is performed for staging and therapeutic purpose in acute leukemia and also in some cases of NHL, such as Burkitt lymphoma and LBL. In the setting of a diagnosis of leukemia, intrathecal chemotherapy is administered at the time of the diagnostic procedure, for treatment of the CNS and to avoid contamination of the CSF by leukemic cells.
Tumor lysis labs including potassium, calcium, phosphate, uric acid, creatinine, and LDH should be checked especially in patients with rapidly dividing tumors and large masses, as is the case with some of the NHLs, and in patients with leukemia with high presenting white blood cell count.
Imaging Evaluation
While most head and neck masses will not cause significant respiratory distress, mediastinal masses can. A chest radiograph (posteroanterior and lateral), specifically looking for a mediastinal mass, should be obtained on all patients that present with leukemia or lymphoma prior to any sedation.
Evaluation with ultrasound can be considered for cervical masses or skin lesions, in the absence of signs of leukemia or lymphoma in the peripheral blood.
Lymphoma staging generally includes a positron emission tomography (PET) scan to assess for other sites of disease in addition to ultrasound, CT, or MRI of lymphadenopathy or masses (Fig. 27.1).
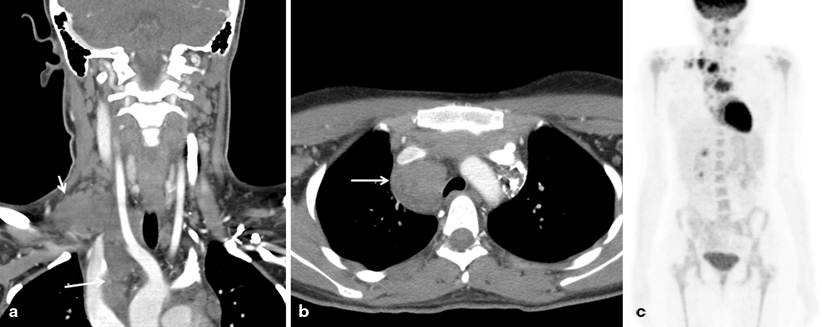
Fig. 27.1
Hodgkin lymphoma (HL). Sixteen-year-old girl with right neck and supraclavicular mass. CT scan (a and b) shows a nodal conglomerate extending from neck into the mediastinum (arrows). PET scan (c) shows increased fludeoxyglucose (FDG) avidity in the right supraclavicular region, anterior mediastinum, and right paratracheal region. Final diagnosis was stage IIA HL
Pathology
When leukemia is suspected, bone marrow aspirates and biopsy (usually from the posterior iliac crest) should be performed. Samples should be obtained for morphologic evaluation as well as for flow cytometry, cytogenetic studies, and molecular studies as indicated. In patients with AML presenting with chloroma, a biopsy of the lesion may be diagnostic (Fig. 27.2). Bone marrow studies are also performed in the staging work-up of HL and NHL.
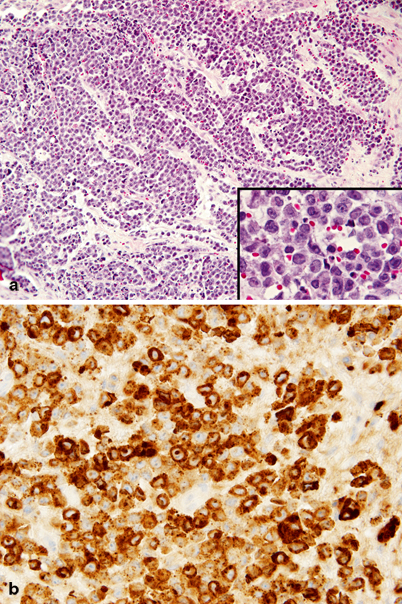
Fig. 27.2
Chloroma (myeloblastic sarcoma). a Profuse soft tissue infiltrate of medium-sized tumor cells. Inset: tumor cells at higher magnification showing relatively abundant cytoplasm, high nuclear cytoplasmic ratio, and reniform, clefted nuclei with prominent eosinophilic single nucleoli. b Tumor cells show diffuse strong cytoplasmic immunoreactivity for myeloperoxidase
In cases of lymphoma where a biopsy of a lymph node is required, an excisional biopsy is preferred over a needle biopsy, since it is important to evaluate the entire architecture of the lymph node for a proper diagnosis.
Both T-cell and B-cell ALL contain lymphoblasts, small- to medium-size uniform cells with little cytoplasm. Flow cytometry and antibody staining confirm the diagnosis and help distinguish between the two types of ALL. Lymphoblasts are almost always deoxynucleotidyl transferase (TdT) and CD34 positive. B-cell markers include CD19, CD20, CD22, CD79a, and PAX5. T-cell markers are CD2, CD3, CD5, and CD7 [23].
Classical HL is divided into four histologic subtypes: nodular sclerosis, mixed cellularity, lymphocyte rich, and lymphocyte depleted (Fig. 27.3). Nodular sclerosis HL is the most common subtype. As its name suggests, it is characterized by nodules and fibrous bands (sclerosis) separating the nodules. The classic Reed–Sternberg cells, large binucleated cells with abundant cytoplasm, and prominent eosinophilic nucleoli accompanied by a mixed inflammatory background including eosinophils, are rare. Multilobated nuclei in an abundant pale cytoplasm, aptly named lacunar cells, are more common. Mixed cellularity, the second most common subtype, also demonstrates a nodular growth pattern but without sclerosis. Reed–Sternberg cells are more common. The lymphocyte rich subtype is distinguished from the two above by a background infiltrate consisting predominantly of B lymphocytes, as opposed to T lymphocytes, and eosinophils, histiocytes and neutrophils. The lymphocyte depleted subtype has very few inflammatory cells but large numbers of Reed–Sternberg cells in a fibrotic background [23].
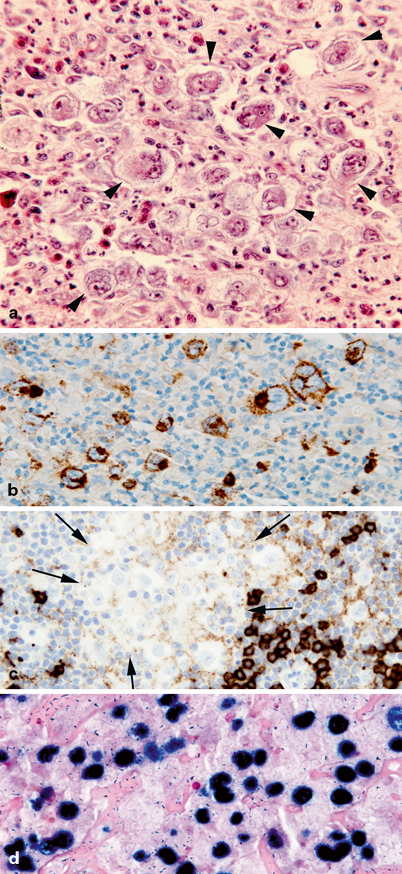
Fig. 27.3
Hodgkin lymphoma (HL). a Numerous large pleomorphic tumor cells with the characteristics of Hodgkin and Reed–Sternberg cells (arrowheads) are surrounded by a nonneoplastic mixed inflammatory background including numerous eosinophils. b Membranous and Golgi immunoreactivity for CD15 is present in large, neoplastic cells. c Large, neoplastic cells (between arrows) are negative for the B-cell marker CD20. Scattered nonneoplastic B lymphocytes (dark staining cells) are also present. d EBV EBER in-situ hybridization showing strong nuclear reactivity (dark blue nuclei) in all large, neoplastic cells
Nodular lymphocyte-predominant Hodgkin lymphoma (NLPHL) is a rare variant of classical HL distinguished by an atypical Reed–Sternberg cell called the lymphocytic and histiocytic (L&H) cell or the “popcorn cell” because of its appearance. While Reed–Sternberg cells are typically CD30 and CD15 positive and CD45 and CD20 negative, L&H cells are the opposite, CD30 and CD15 negative and CD45 and CD20 positive [23].
As mentioned earlier, NHL consists of a heterogeneous population of diseases. LBL pathology is very similar to that of lymphoblastic leukemia (Fig. 27.4). Three other common NHLs in children with distinct pathology are Burkitt lymphoma, large B-cell lymphoma, and anaplastic large cell lymphoma.
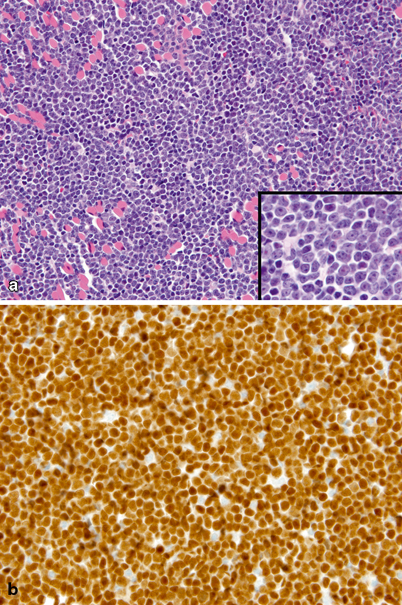
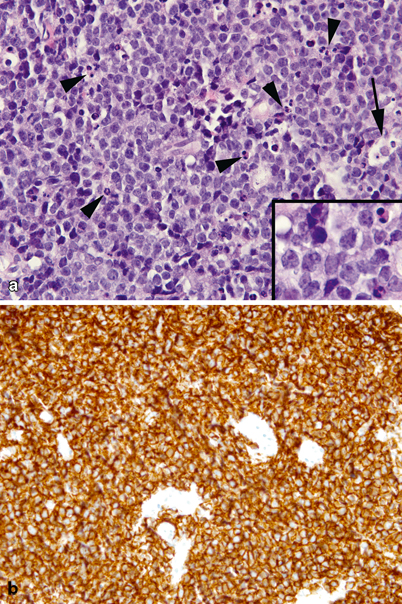
Fig. 27.4
Lymphoblastic lymphoma. a Diffuse, proliferation of monotonous small-sized tumor cells infiltrating skeletal muscle. Inset: small- to medium-sized tumor cells at higher magnification showing scant cytoplasm, irregular nuclear envelopes, finely dispersed chromatin, and small nucleoli. Muscle cells are the large eosinophilic cells interspersed throughout. b Diffuse, strong nuclear immunoreactivity for terminal TdT
Burkitt lymphoma is the most common mature B-cell neoplasm in children. The tumor is comprised of a homogeneous population of medium-size cells with multiple prominent nucleoli and lipid-filled basophilic cytoplasm, intermixed with macrophages full of debris from turnover of rapidly dividing and apoptotic tumor cells. Together they create the classic starry-sky appearance (Fig. 27.5). The tumor cells express B-cell antigens such as CD19 and CD20 and lack immature markers including TdT and CD34. Ninety percent will have a MYC gene translocation.