Fig. 31.1.
(a) Meckel’s diverticulum, (b) omphalomesenteric fistula, (c) omphalomesenteric cyst, and (d) remnant vitelline artery/fibrous cord.
Pathophysiology
Meckel’s diverticulum is located on the antimesenteric border of the ileum and contains all of the layers of the intestinal wall making it a true diverticulum. It is located within 20–100 cm (average 50 cm) proximal to the ileocecal valve [2, 8, 11]. The blood supply to the MD is the remnant vitelline artery, which now arises from a terminal branch of the superior mesenteric artery (SMA) [10]. The rules of 2s are frequently used to describe MD [7, 12].
Rules of 2s
2 % of the population.
Twice as common in males.
2 % are symptomatic.
Usually 2 cm in diameter.
Usually 2 in. long.
Located within 2 ft of the ileocecal valve.
As the cells of the omphalomesenteric duct are pluripotent, the MD may contain ectopic mucosa. It has been estimated that 12–50 % of MD contain heterotopic mucosa, most commonly gastric mucosa followed by pancreatic tissue [4, 13–15]. Less commonly, the diverticulum may contain colonic tissue, Brunner’s glands, hepatobiliary tissue, and small bowel malignancies such as carcinoid, gastrointestinal stromal tumors, lymphoma, leiomyosarcoma, adenocarcinoma, and intraductal papillary mucinous neoplasm [5, 16].
Preoperative Evaluation
History
Most MD are asymptomatic and are usually found incidentally when undergoing surgical exploration for other intra-abdominal processes. The most common presentations for symptomatic MD are bleeding, intestinal obstruction, and inflammation.
Episodic painless hematochezia occurs when MD contains ectopic gastric mucosa that secretes hydrochloric acid. This can cause ulceration on the adjacent ileum, leading to bleeding and perforation. Meckel’s diverticulum is the most common etiology of intestinal bleeding in children. Bleeding can occur in MD containing malignancies, but this has been described predominantly in adults [17, 18].
Obstruction related to MD can be caused by intussusception, volvulus, or internal abdominal wall herniation. It is the second most common presentation of MD in children, but the most common presentation in adults. Intussusception can occur when the MD inverts into the lumen of the ileum, acting as a lead point. Volvulus most commonly occurs when a loop of small bowel twists around a fibrous band remnant or an incompletely obliterated omphalomesenteric duct connecting the small bowel to the umbilicus. Loops of bowel can also be obstructed through an internal herniation of bowel around a mesodiverticular band, which is the remnant vitelline artery from the diverticulum to the mesentery [13, 17]. Littre’s hernia is the presence of a Meckel’s diverticulum in a hernia sac, most commonly in an inguinal (50 % of cases), umbilical, or femoral hernia [7, 19].
Meckel’s diverticulitis most commonly occurs secondary to an enterolith in the lumen, similar to the pathophysiology of acute appendicitis, and most commonly occurs in adult patients. Inflammation of the MD can also occur from peptic ulceration of the ileal mucosa or from a foreign body in the diverticular orifice (i.e., gallstones or ingested foreign objects such as bones or toothpicks) [17, 20].
If the omphalomesenteric duct is completely patent or if the distal portion is patent, then umbilical drainage can also be a presenting symptom.
Exam
In most studies, the most common presenting symptom of MD in children under the age of 5 years is episodic painless hematochezia. This bleeding is acute and can be massive, often requiring blood transfusions. The stool is often dark red or “currant jelly.” These bleeding ulcers can also perforate and cause peritonitis. Obstruction is the second most common presentation and is more common in adults. These patients present with crampy abdominal pain, nausea, bilious emesis, and obstipation. Patients presenting with diverticulitis have signs and symptoms that are similar to those of acute appendicitis [4, 13].
Laboratory Testing
If the patient is presenting with rectal bleeding, a hemoglobin, hematocrit, and type and cross are required. A white blood cell count is also helpful to delineate an inflammatory process such as diverticulitis. If the patient is presenting with obstructive symptoms, a metabolic panel is indicated to evaluate for electrolyte abnormalities.
Imaging
Abdominal X-Ray
Conventional X-rays may occasionally show an enterolith, evidence of a bowel obstruction, or a gas-/fluid-filled diverticulum. If a patient has a perforation, then free air could optimally be seen on upright chest X-ray or at left lateral decubitus film. Barium studies may show a blind-ending pouch in the distal ileum, but it has low sensitivity secondary to poor filling of the diverticulum from stenosis of the neck, obstructing intestinal contents, contraction of the tunica muscularis, and/or overlapping of small bowel [21].
Ultrasound
High-resolution ultrasonography has been utilized in diagnosing MD. It classically would show a fluid-filled structure in the mid-lower abdomen with a connection to a peristaltic small bowel loop [22]. During episodes of diverticulitis, it would have an appearance similar to appendicitis.
Computed Tomography (CT)
CT is currently the most widely used imaging modality to evaluate for abdominal pain, obstructive symptoms, or potential inflammation. In a study by Kawamoto et al., MD was detected in up to 47.5 % of all patients and 57 % of symptomatic patients [23]. It is the best modality for Meckel’s diverticulitis and obstruction. An adjunctive imaging modality is CT enterography, where a large volume of oral contrast is ingested to achieve adequate small bowel distention to evaluate the small bowel [21].
Meckel’s Scan
This is the most accurate diagnostic study (sensitivity and specificity >90 %) for detecting MD containing gastric mucosa. It is performed using technetium-99 m, which is taken up by the mucin-secreting cells of the ectopic gastric mucosa. It is a commonly used diagnostic test for rectal bleeding since it is noninvasive with a low radiation burden [21, 24].
Angiography
This is another useful imaging modality if the patient presents with active large volume bleeding. Meckel’s diverticulum can be diagnosed by seeing a blush at the site of the persistent vitelline artery arising from the distal SMA. This has less sensitivity as it is only diagnostic if there is bleeding of at least >0.5 ml/min [21, 25]. This modality also allows the option of angiographic embolization that may control bleeding until operative resection.
Other Tests
Double Balloon Enteroscopy
This is a less commonly used modality for diagnosis. It is performed by transoral or transanal endoscopy. This diagnostic tool has also been described as an aid in surgical resection, where the endoscopic light is directed to the abdominal wall indicating the location of the diverticulum and allowing resection through a single small umbilical incision [9, 17, 26]. This modality has largely been replaced by laparoscopic evaluation.
Laparoscopy
Surgical Indications
There is clear consensus that surgical resection is indicated for all symptomatic MD; however, there is controversy regarding resection of incidentally found MD. In a retrospective study of MD, Cullen et al. described a 6.4 % cumulative lifetime risk of developing complications and found surgical resection of incidental MD had less morbidity compared to surgical resection of complicated MD; therefore, they recommend surgical resection of all MD [29]. Alternatively, Soltero et al. recommended against prophylactic resection as they found a 9 % risk of morbidity after MD resection in these patients and calculated that 800 asymptomatic diverticula had to be removed to prevent one death [3]. Between these two extreme positions, other studies, such as Park et al., found that certain characteristics may be predictive for the development of complications and recommended diverticulectomy of incidental diverticula that meet any of the following four criteria: (1) male sex, (2) age less than 50 years, (3) diverticular length greater than 2 cm, and (4) abnormal features of the diverticula such as thickened tissue or a palpable mass [14]. Other studies have also recommended prophylactic resection if there is a narrow diverticular base [30]. At this time, most pediatric surgeons will perform a resection of an incidentally identified MD if it does not significantly increase the risk of the primary procedure.
Technique
Special Considerations
If a patient is presenting with obstruction, then either laparotomy or laparoscopy (performed by a surgeon experienced in laparoscopic small bowel resection and lysis of adhesions) is the procedure of choice. If a Littre’s hernia is present, then a MD resection should be performed first followed by herniorrhaphy [7, 31]. There are various new surgical techniques that are being published, such as hand-assisted laparoscopic resection and single-site surgery; however, these are beyond the scope of this chapter and will not be discussed further [32–34]. Instead, this chapter will focus on laparoscopic MD resection.
Anatomy
As described above, a MD is found on the antimesenteric border of the ileum within 2 ft of the ileocecal valve. It receives its own blood supply from the mesentery of the ileum, usually from the remnant vitelline artery.
Instruments
30° laparoscope, 10 or 12 mm trocar through the umbilicus for the laparoscope and subsequent endoscopic stapler, two additional 5 mm trocars as working ports, atraumatic bowel graspers, a laparoscopic electrocautery or other energy devices, or vascular clip can all prove useful for this procedure.
Steps
Standard general anesthesia and preoperative antibiotics are utilized. A preoperative time-out should be performed. For patients with hematochezia, blood products should be readily available. A Foley catheter should be placed for decompression of the bladder if the patient has not voided immediately before entering the OR suite. A chlorhexidine/alcohol skin preparation should be performed, and the patient should be draped to expose the entire abdomen.
The first step is entrance into the abdomen. We recommend entrance using the Veress needle technique or the Hassan technique using a 10–12 mm trocar through the umbilicus into the peritoneum under direct visualization and subsequent insufflation.
Insufflation is achieved using carbon dioxide pneumoperitoneum at 8–12 mmHg.
A 30° laparoscope should then be introduced through the umbilical port.
Two additional 5 mm trocars should then be inserted through the left lower quadrant and suprapubic region, similar to sites used for an appendectomy (Fig. 31.2).
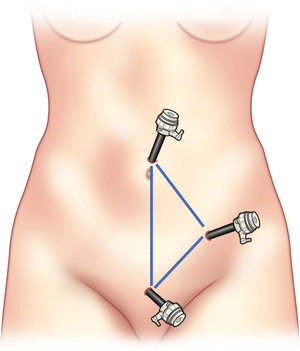
Fig. 31.2.
Trocar locations.
The cecum should first be identified, and this will be facilitated by moving the patient into a left lateral decubitus and Trendelenburg’s position.
A systematic exploration of the small intestine from the terminal ileum to the jejunum should then be performed in a retrograde fashion using blunt bowel graspers.
Once located, the diverticulum will need to be released from the mesentery by dividing the feeding vessel using an energy device or vascular clip.
The MD should then be grasped and delivered through the umbilical port site under direct visualization by placement of the laparoscope through one of the other working ports. The umbilical incision may need to be extended.
A segmental resection of the diverticulum and adjacent ileum should be performed if the patient is presenting with bleeding to ensure removal of all ectopic mucosa and the bleeding ileal ulcer (Fig. 31.3).
Re-approximation of the small bowel may be performed using a hand-sewn anastomosis or a side-to-side functional end-to-end stapled anastomosis.




