Inguinal Hernias and Hydroceles
History
The term hernia comes from the Greek ‘hernios’, meaning offshoot or bud. Inguinal hernias were described in the Ebers Papyrus in 1552 BC,1 and were found in Egyptian mummies. Celsus is thought to have performed hernia repairs in 50 AD. Galen (b. 129 AD) described the processus vaginalis, defined hernias as a rupture of the peritoneum, and advised surgical repair.2 Ambrose Paré advocated repair of inguinal hernias in childhood in the 16th century, and condemned concomitant castration.1 In 1807, Cooper identified the transversalis fascia and the ligament associated with his name. In 1817, Cloquet observed that the processus vaginalis is often patent at birth and described femoral hernias.3,4 Marcy reported high ligation of the hernia sac in 1871.5 In 1877, von Czerny first described narrowing the inguinal canal and tightening the external inguinal ring,6,7 followed by Bassini’s description of internal inguinal ring tightening and reinforcement of the posterior canal in 1887.8 Gross reported a 0.45% recurrence rate in a large series of inguinal hernia repairs (3,874 children) in 1953.9
Incidence
Approximately 1–5% of all children will develop an inguinal hernia and a positive family history is found in about 10%.10 There is an increased incidence in twins, more frequently in male twins.11 In a series of 6,361 pediatric herniorrhaphies performed by a single surgeon, the male-to-female ratio was 5 to 1, and right-sided hernias were twice as common as those on the left.12 The mean age at diagnosis was 3.3 years.
The incidence of an inguinal hernia varies directly with the degree of prematurity. The overall incidence of inguinal hernia in premature infants is estimated to be 10–30%, whereas term newborns have a rate of 3–5%.13–15 The incidence by gender is closer to 1 : 1 in premature infants. Comorbidities such as chronic lung disease associated with prematurity may play a role in the development of an inguinal hernia.
Associations
Cystic Fibrosis
Patients with cystic fibrosis have an increased risk of an inguinal hernia, with an incidence as high as 15%.16 This heightened risk may be due to elevated intra-abdominal pressure from respiratory symptoms, but developmental and/or embryologic factors may also play a role because the risk of a hernia is also increased in unaffected siblings and parents.
Hydrocephalus
Ventriculoperitoneal shunts (VPS) are associated with an increased incidence of an inguinal hernia as well as increased chance of bilaterality, incarceration, and recurrence.17 In a series of 430 children who underwent placement of a VPS, 15% developed a hernia.18 Bilaterality was common; it occurred in nearly 50% in boys and approximately 25% of girls. Hydroceles occurred in another 6%. A large series of children with shunts found that inguinal hernias were more likely to develop in neonates than in older children and were more common in boys than in girls. The average time from placement of a VPS to hernia repair was approximately one year.19
Peritoneal Dialysis
As with VPS, patients on long-term peritoneal dialysis also have an increased risk of an inguinal hernia. Some authors even advocate searching for a patent processus during insertion of the dialysis catheter (either radiographically or via laparoscopy), and closing the sac at that time.20
Embryology and Anatomy
The inguinal canal is a six-sided cylinder. The cephalad opening is the internal inguinal ring and the caudal border is the external inguinal ring. The cephalad aspect is bordered by the internal oblique, transversus abdominis, and medial external oblique fibers. The floor is formed by the transversalis fascia and the ‘conjoint tendon.’ The anterior roof is created primarily by the aponeurosis of the external oblique. The inferior wall is composed by the inguinal ligament, lacunar ligament (medial third), and iliopubic tract (lateral third). Contents include the ilioinguinal nerve (exiting through the external inguinal ring) and in males, the spermatic cord. In females, it also contains the round ligament.21
The processus vaginalis is a peritoneal diverticulum extending through the internal inguinal ring into the inguinal canal. It can be seen by 3 months of fetal life.22 The somatic base of this diverticulum is the transversalis portion of the endoabdominal fascia. The gonads form on the anteromedial nephrogenic ridges in the retroperitoneum during the 5th week of gestation. The gonads are attached to the scrotum by the gubernaculum in the male and to the labia via the round ligament in the female. Gonadal descent begins by 3 months’ gestation, and the testis reaches the internal inguinal ring by about 7 months. Descent of the testis is thought to be directed in the abdominal phase by insulin-like 3 protein, a product of the Leydig cells, and directed in the second phase by androgens and release of calcitonin gene-related peptide (CGRP) from the genitofemoral nerve (via fetal androgen release).23,24 CGRP appears to mediate closure of the patent processus vaginalis (PPV), although this process is not completely understood.24
The testis begins to descend down the canal by the seventh month of fetal life preceded and guided by the processus vaginalis.22,24,25 The processus, which is located anterior and medial to the cord structures, gradually obliterates, and the scrotal portion forms the tunica vaginalis. The female anlage of the processus vaginalis is the canal of Nuck, a structure that leads to the labia majora. This also closes by about 7 months of fetal life, and ovarian descent is arrested in the pelvis.22 The precise incidence of PPV in newborns is unknown and depends on gender and gestational age. It is estimated to be 40–60%, but may be lower or higher.26 However, at autopsy, only 5% of adults have a PPV.22 PPVs can still close after birth, but this is felt less likely to occur with increasing age. It is failure of the PPV to close that results in an indirect inguinal hernia. As mentioned, the factors driving PPV closure are incompletely understood. Intra-abdominal pressure probably plays a role because disorders with increased abdominal pressure/fluid (e.g., ascites, VPS) are associated with an increased incidence of indirect inguinal hernias and an increased incidence of bilaterality.17 Indirect inguinal hernias are more common on the right. The various clinical findings related to the processus vaginalis are illustrated in Figure 50-1.
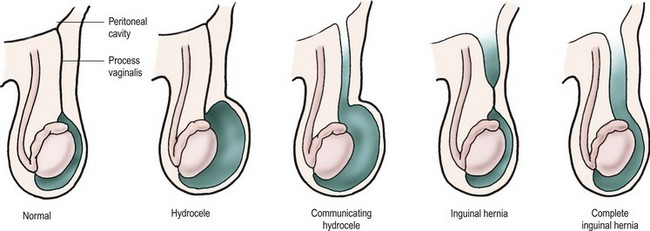
FIGURE 50-1 From left, configurations of hydrocele and hernia in relation to patency of the processus vaginalis.
The layers of the abdominal wall contribute to the layers of the testis and spermatic cord as the gonad descends. The internal spermatic fascia is a continuation of the transversalis fascia, the cremaster muscle derives from the internal oblique, and the external spermatic fascia originates from the external oblique aponeurosis. The processus vaginalis envelops the testis as the visceral and parietal layers of the tunica vaginalis.22
Clinical Presentation
A common occurrence is a normal examination in combination with a suggestive history. Cell phone picture documentation by the parents has become commonplace. A good history is acceptable as an indication for operation. False-negative explorations are rare. In the previously mentioned series of 6,361 hernia repairs by a single surgeon (definitive inguinal hernia on examination was the indication for operation), there was only one false-negative exploration (0.02%).12
Children are often referred for inguinal pain in the absence of any history of bulging or swelling, often with a normal physical examination. Other sources such as musculoskeletal strain, gastrointestinal, or genitourinary abnormalities should be excluded before operative intervention. Diagnostic transumbilical laparoscopy is useful in a small subset with equivocal examinations or persistent symptoms and no other apparent cause.
Ancillary findings such as a ‘silk glove sign’ (feeling the thickened peritoneum of the patent processus as the cord is palpated) or examination under anesthesia are of variable reliability.27,28 Radiologic diagnostic aids are not generally necessary or helpful. Ultrasonography (US) can be used to identify a PPV indirectly via widening of the internal inguinal ring (more than 4–5 mm is positive), but the technique is highly operator dependent and not widely used in children.29,30 It is not generally necessary to restrict an asymptomatic child’s activities until repair is scheduled, but prompt repair may decrease interim incarceration, particularly in the very young.
Hydrocele
The management of asymptomatic hydroceles in infants is somewhat controversial. There is general agreement that a noncommunicating, asymptomatic hydrocele in an infant should simply be followed. One recent study found that 89% of 121 infants who were followed resolved by 1 year of age.31 The duration of observation varies by surgeon, with most recommending operation by one or two years of age if the hydrocele fails to resolve or if a clinical hernia is apparent.20,32
Expectant management has been extended by some authors to infants with communicating hydroceles (and therefore PPVs). In a 2010 report, in 110 infant boys with an apparent communicating hydrocele, 63% had complete resolution without operation by a mean age of nearly one year.33 Interim incarceration did not occur in this series.
Many authors recommend operation for an infant with a giant hydrocele, although the definition is subjective and variable. Most surgeons also repair hydroceles of the cord.34
Excision of the hydrocele sac is not necessary. The fluid should be evacuated, and the distal sac is opened widely. Large or thick sacs may be everted behind the cord (Bottle procedure).35
Hydroceles in adolescents are often a complication of varicocelectomy. A de novo hydrocele in this age group may represent an inguinal hernia or simply an idiopathic hydrocele. A thorough history and physical examination to exclude communication hernia should be performed. Also, an ultrasound (particularly if the testis is not palpable, since a reactive hydrocele accompanies about 15% of testicular tumors) should be obtained. A trans-scrotal hydrocelectomy is appropriate in adolescents in the absence of signs of hernia or tumor. Otherwise an inguinal approach is best.36 Transumbilical diagnostic laparoscopy for evaluation for a PPV is a good option in equivocal cases.
An abdominoscrotal hydrocele is an hourglass-shaped collection with both an inguinoscrotal and abdominal component. A combined inguinal and laparoscopic approach may be helpful.37
Incarceration
The incidence of hernia incarceration is variable and ranges from 12–17%.12,38,39 Younger age and prematurity are risk factors for incarceration.40 The mean age of patients with incarceration is significantly lower than that of those who undergo elective repair.12,41
Once an incarcerated hernia is reduced, a delay of 24 to 48 hours to allow resolution of edema is reasonable. Reliability of the family as well as clinical (very difficult reduction) and geographic considerations may dictate the need for admission and observation before definitive repair. Overall, 90–95% of incarcerated hernias can be successfully reduced.42 In one report, only 8% required emergency operation out of 743 incarcerated hernias.12 Two children required bowel resection.
Urgent operation is necessary if reduction fails. The hernia may reduce with induction of general anesthesia. If so, the hernia sac should be opened and inspected. The presence of enteric contents or bloody fluid mandates either open exploration via separate incision or La Roque maneuver (incision in the transversalis fascia through the same inguinal skin incision) or, more commonly, laparoscopic evaluation. It may be necessary to open the internal inguinal ring laterally to reduce the bowel. Some surgeons approach an incarcerated hernia by transumbilical laparoscopy to both reduce the hernia and evaluate the bowel.43,44 There is some evidence that the laparoscopic approach is associated with fewer complications.45
Intestinal injury requiring treatment is rare (1% to 2%), even with incarceration.12 The hernia sac is often quite edematous and friable, and repair of the hernia can be quite difficult. The risk of recurrence is significantly increased. We do not routinely employ laparoscopy looking for a contralateral PPV in patients with incarceration.
The testis on the incarcerated side is often edematous and somewhat cyanotic. Unless the gonad is frankly necrotic, it should be preserved. The parents of any boy with an incarcerated hernia should be counseled about the possibility of testicular loss or atrophy, but the incidence of this complication is only 2–3%.42,46 Incarceration of an ovary in a hernia sac may not always impair its blood supply, but most pediatric surgeons will promptly (but not emergently) repair the hernia in a girl even with an asymptomatic, nontender incarcerated ovary.47
Management
Anesthesia
There are no good data comparing regional to general anesthesia for pediatric inguinal hernia repair. A 2003 Cochrane meta-analysis of available data regarding this issue in premature infants concluded: ‘There is no reliable evidence from the trials reviewed concerning the effect of spinal as compared to general anesthesia on the incidence of postoperative apnea, bradycardia, or oxygen desaturation in ex-preterm infants undergoing herniorrhaphy.’48
Overnight hospitalization is not necessary after inguinal hernia repair for healthy children or term infants. However, the risk of postoperative apnea and bradycardia is increased in premature infants and overnight monitoring may be necessary. The postconceptual age (gestational age plus chronologic age) is commonly used to decide which infants require overnight admission. Several studies have addressed this issue.49,50 A review of 127 premature infants admitted after inguinal hernia repair found a incidence of apnea of about 5%, and attempted to identify factors associated with a higher risk (history of prior apnea, lower gestational age and birth weight, comorbidities).50 A less than 1% risk of postoperative apnea was found in former premature infants greater than 56 weeks postconceptual age in a comprehensive analysis of eight prospective studies.51 Sixty weeks postconceptual age is widely used as a cut-off for admission (although there is substantial institutional variability). However, a more recent retrospective review at our hospital demonstrated a low incidence of adverse events after 50 weeks postconceptual age.52
Timing
As premature infants have an increased incidence of an inguinal hernia, this is a common diagnosis in the neonatal intensive care unit. The incidence of bowel incarceration in premature infants is significantly increased (three times in one large series).12 Many institutions use 2 kg as a lower limit for repair in asymptomatic and otherwise relatively healthy newborns. We usually repair the hernia before discharge to avoid the need for readmission for herniorrhaphy and to decrease the risk of incarceration.46,53–55 However, this depends on other comorbidities. One recent study compared repair before discharge or as an outpatient, and found an increased length of hospitalization in the former group (largely from respiratory complications) and a low incidence of incarceration in the latter group.56 However, others have found that in-house repair is preferable due to a lower incarceration rate after discharge.57,58



