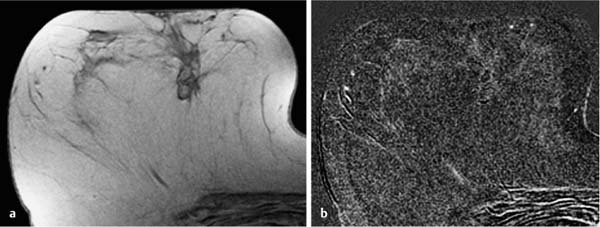
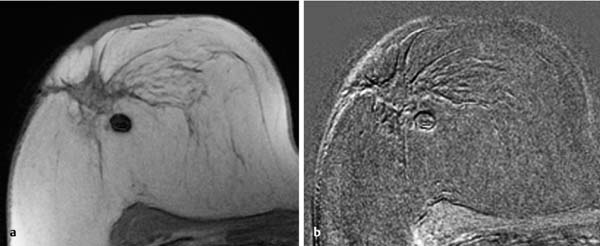
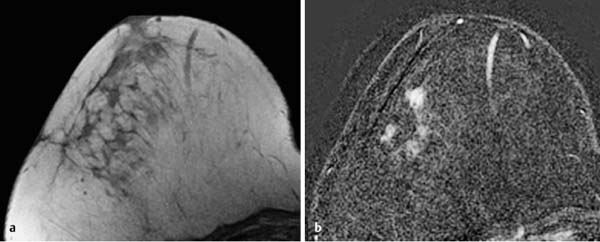
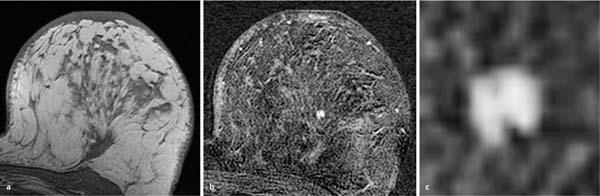
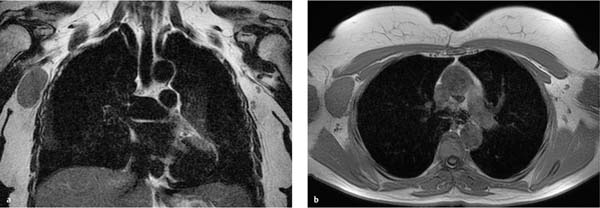
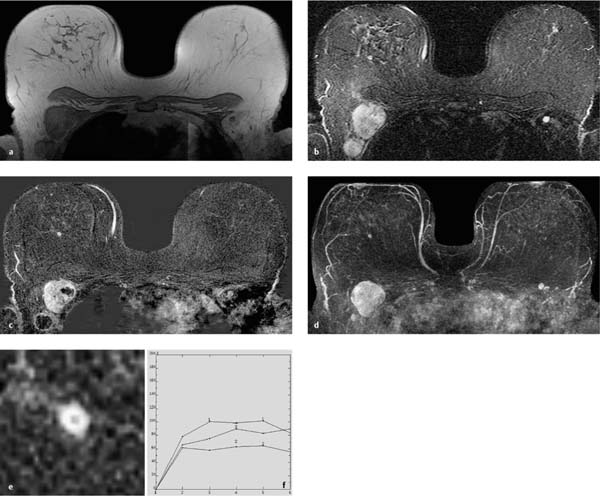
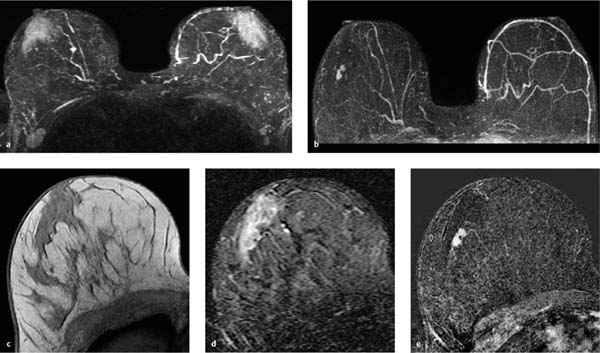
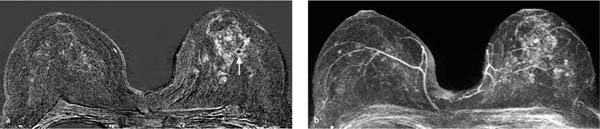
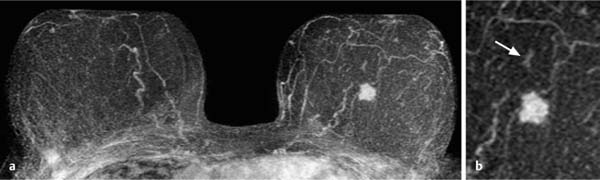
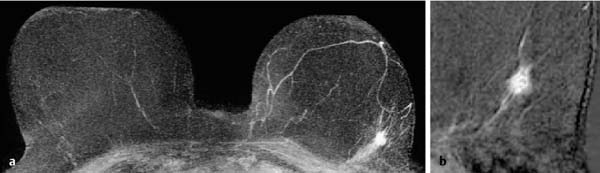
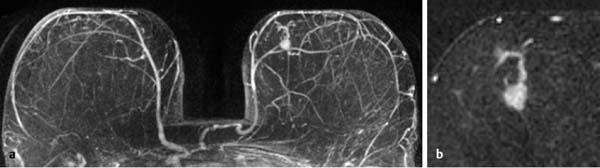
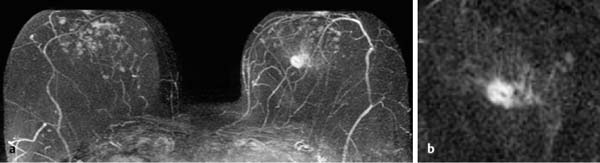
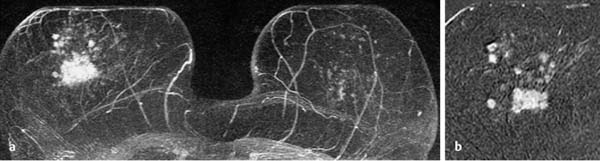
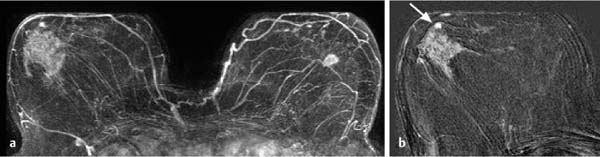
17 Indications for Breast MRI The expedient use of breast MRI requires several aspects to be taken into consideration: Clinical examination, mammography, and breast ultrasonography cannot always reliably differentiate between a postoperative scar and breast cancer, or between a postoperative scar after breastconserving therapy (BCT) and a local recurrence. Vascularization. MRI makes possible the differentiation between nonvascularized scar tissue and a strongly vascularized malignancy, and is therefore the ideal examination technique for this indication. If breast MRI shows no enhancement in the scar tissue area after surgery, the presence of a carcinoma or recurrence can be reliably excluded (Figs. 17.1, 17.2). If localized enhancing areas are present in the scar area, then the presence of a malignancy must be considered (Figs. 17.3, 17.4). The dynamic signal characteristics are usually not relevant in this context as carcinoma tissue within a scar often displays an unspecific TIC. Apparently tumor angiogenesis of a carcinoma within fibrotic scar tissue is often increased to a lesser degree than that of carcinomas at other locations. It must be noted, however, that not all enhancing areas within a scar are necessarily an indication of malignancy or recurrence. Occasionally reactive hyperemia may be present. The recommended interval between the end of radiation therapy after breast-conserving therapy and a breast MRI examination is 12 months. Fig. 17.1a, b Postoperative scar after diagnostic excision. a T1w precontrast slice image: architectural distortion and several hyperintense fat necroses in the surgical region. b Subtraction slice image: no enhancement in the scar region. No pathological findings during the follow-up period. Fig. 17.2a, b Postoperative scar after breast-conserving surgery. a T1w precontrast slice image: typical architectural distortion in the region of surgery. Susceptibility artifact due to clip marker. b Subtraction slice image: no enhancement in the scar region rules out recurrence reliably. No pathological findings during the followup period. Fig. 17.3a, b Recurrence in the surgical region after breast-conserving surgery. a T1w precontrast slice image: several, nonlipomatous, masslike lesions in the surgical region after breast-conserving therapy without radiation therapy (rejected by the patient). b Subtraction slice image: nodular enhancement within these lesions with discrete enhancement of surrounding tissue areas. Histology: bifocal invasive ductal breast carcinoma (= recurrence with surrounding EIC). Fig. 17.4a–c Recurrence in the surgical region after breast-conserving surgery. a T1w precontrast slice image: typical architectural distortion in the surgical region after breast-conserving therapy. Skin thickening due to radiation therapy. b Subtraction slice image: focal enhancement in the ventral aspect of the scar area shows endotumoral dark septation, not typical of malignancy. c Zoomed partial view of the hypervascularized lesion. Histology: recurrence of invasive lobular breast carcinoma. Importantly, breast MRI allows the reliable exclusion of breast cancer recurrence when there is no enhancement in the scar region. False-negative breast MRI findings are practically unknown. Scheduling. There are no concrete recommendations for the scheduling of breast MRI examinations after BCT. From our experience, however, it seems expedient to routinely perform breast MRI one year after the end of adjuvant radiation therapy and at 2-year intervals thereafter. New possible therapeutic strategies. Due to cumulative side-effects, it is not possible to perform a second course of radiotherapy in a woman who has had BCS with adjuvant radiation therapy. The detection of an ipsilateral breast cancer recurrence therefore generally results in mastectomy. However, the great reliability of high-quality high-resolution (HR) breast MRI examinations in excluding breast cancer makes it conceivable that this strategy, which originated in the era of x-ray mammography and ultrasonography, might change in the future. It is imaginable, for example, that a second BCS could be performed when a metachronous, ipsilateral breast cancer recurrence has been detected in a woman who has already undergone earlier BCS with adjuvant radiation therapy. This could be the case when the recurrence is a solitary finding without signs of extensive intraductal component in a breast with high breast MRI transparency (MRI density I, Fig. 17.5). Such speculation has not yet been substantiated by scientifically acquired data. Fig. 17.5 A theoretical constellation of findings that could make possible a second BCS for breast cancer recurrence after BCS + radiation therapy. New hypervascularized mass lesion with a diameter of 6 mm in the scar area of the right breast, 8 years after BCS with radiation therapy. High transparency (MRI density I) without further suspicious findings. Histology: IDC, pT1b. The term CUP syndrome is used to denote the presence of cancer cells, i.e., metastases, without knowledge of the primary tumor site from which these originate. A typical constellation leading to diagnostic breast examinations is the detection of an axillary lymph node metastasis (locoregional metastasis) without prior identification of the primary tumor site (e.g., in the breast, Fig. 17.6). In a broader context, it is sometimes reasonable to search for the primary tumor site in the breast when metastatic spread into other body regions (hematogenous metastasis) has occurred. Diagnostic strategy. X-ray mammography is the diagnostic imaging method of choice in such a setting. Ultrasonography of the breast is a useful complementary method depending on parenchymal density. Dynamic MR mammography is indicated when both these diagnostic techniques have failed to identify an intramammary primary tumor (Figs. 17.7, 17.8). Fig. 17.6a, b Carcinoma of unknown primary (CUP) with axillary lymph node metastasis. Enlarged right axillary lymph node with histologically verified adenocarcinoma cells (US-guided core biopsy). At this time there were no clinical or mammographic signs of breast cancer making this a CUP. a T1w precontrast coronal slice image: upper thoracic aperture. b T1w precontrast axial slice image. a T1w precontrast slice image: several pathologically enlarged right axillary lymph nodes. b IR T2w slice image. c Corresponding subtraction slice image: hypervascularized focus with a diameter of 4 mm and little central perfusion (rim sign). d Subtraction MIP. e Zoomed subtraction slice image: better visualization of rimenhancement and poorly defined borders. f TIC: unspecific. Histology: invasive ductal breast carcinoma (pT1a) as the primary tumor with extensive locoregional lymph node metastases. a T2w MIP: pathologically enlarged right axillary lymph node. b Subtraction MIP: solitary barbell-shaped enhancement in the lateral aspect of the right breast. c T1w precontrast slice image (right breast): unremarkable findings. d IR T2w slice image (right breast): unremarkable findings. e Corresponding subtraction slice image: good visualization of primary tumor. Histology: bifocal invasive ductal breast carcinoma. One of the most important indications for use of breast MRI preoperatively. Of all breast imaging techniques, breast MRI has the greatest sensitivity for the detection of breast cancer. It is for this reason that MR mammography is especially suited to give additional preoperative information about the tumor extent, the presence of an extensive intraductal component (EIC), possible multifocality or multicentricity, and the presence of contralateral breast cancer. Numerous studies convincingly document the importance of preoperative MRI staging for the effective planning of the appropriate stage-dependent treatment strategy. Studies comparing the preoperative estimation of tumor size by breast imaging techniques (mammography, ultrasonography, MRI) with the histological tumor measurements show that MR mammography is superior to the other imaging modalities (Fig. 17.9). Current data show no significant difference in the size estimated by breast MRI and that measured in the histological specimen. In contrast, x-ray mammography and ultrasonography significantly underestimate the tumor size. An extensive intraductal component (EIC) is present when the intraductal component surrounding an invasive ductal carcinoma constitutes 25% or more of the tumor. An EIC is the primary cause of breast cancer local recurrence in the scar area after BCT. Breast MRI is superior to x-ray mammography and breast ultrasonography in the preoperative detection of EIC. Criteria and procedures. The principal characteristic in breast MRI indicating the presence of an EIC is a contrast-enhancing area with linear (Figs. 17.10, 17.11), linear-branching (Fig. 17.12), or dendritic (Fig. 17.13) configuration adjacent to the primary tumor. The additional presence of small round or ovoid enhancing foci is often an indication of the presence of microinvasive tumor (Fig. 17.14). The signal analysis is usually not diagnostically helpful in such nonmasslike enhancing lesions. In the presence of the morphological changes described above, therefore, an unsuspicious time to signal intensity curve (TIC) cannot rule out an EIC. An extensive intraductal tumor that contains areas indicative of invasive carcinoma should not be designated as an EIC, but rather as a ductal carcinoma in situ (DCIS) with signs of incipient localized invasion (Fig. 17.15). The percutaneous biopsy of such lesions should therefore preferably target areas most suspicious for invasive tumor so that, if it is confirmed, appropriate therapeutic procedures can be initiated (e.g., sentinel lymph node biopsy). Preoperative confirmation of breast cancer infiltration into the pectoral muscle is significant because it occasionally leads to a change in the indicated surgical procedure to include a fasciotomy or myotomy of the affected muscle areas. Here also, breast MRI is the favored imaging technique because it allows overlap-free visualization of the pectoral muscle in the entire retroparenchymal space. Fig. 17.9a, b Tumor extent in breast MRI. Patient with regionally distributed, pleomorphic microcalcifications in the upper outer quadrant of the left breast. Histology revealed DCIS after stereotactic VAB. a Subtraction slice image: linear/dendritic enhancement pattern in both upper breast quadrants of the left breast. Biopsy cavity (arrow). b Subtraction MIP: improved visualization of tumor extent. Procedure: percutaneous histological confirmation of DCIS in the left upper inner quadrant resulted in primary mastectomy of the left breast. Fig. 17.10a, b Intraductal component in breast MRI. a Subtraction MIP: invasive ductal breast carcinoma in the central aspect of the left breast. b Zoomed partial view of subtraction slice image: discrete linear enhancement (4 mm long) ventrally of the index tumor as the imaging correlate of an intraductal component. Histological confirmation. Fig. 17.11a, b EIC in breast MRI. a Subtraction MIP: invasive ductal breast carcinoma in the lateral aspect of the left breast. b Zoomed partial view of subtraction slice image: linear enhancement ventrally and dorsally of the index tumor as the imaging correlate of EIC. Histological confirmation. Fig. 17.12a, b EIC in breast MRI. a Subtraction MIP: invasive ductal breast carcinoma in the craniomedial aspect of the left breast. b Zoomed partial view of the subtraction slice image: microlobulated index carcinoma with ventral linear-branching enhancement as the imaging correlate of EIC. Histological confirmation. Fig. 17.13a, b EIC in breast MRI. a Subtraction MIP: invasive ductal breast carcinoma in the central aspect of the left breast. b Zoomed partial view of the subtraction slice image: peritumoral dendritic enhancement of the index tumor as the imaging correlate of EIC. Histological confirmation. Fig. 17.14a, b EIC in breast MRI. a Subtraction MIP: large invasive ductal breast carcinoma in the central aspect of the right breast. b Zoomed partial view of the subtraction slice image: multiple peritumoral hypervascularized foci as the imaging correlate of EIC with microinvasive tumor components at several locations. Histological confirmation. a Subtraction MIP: regional enhancement in the upper outer quadrant of the right breast without correlative findings on mammography or breast ultrasound. Mass lesion correlating with mammographically and sonographically detected breast carcinoma in the left breast. b Subtraction slice image (right breast): nonmasslike enhancement with small, localized area of significantly stronger enhancement (arrow) as the correlate of area of invasive tumor. Histology: extensive area of high-grade DCIS with small invasive component (IDC pT1b) in the right breast. IDC (pT1c) in the left breast.
 Widely accepted indications for the performance of breast MRI include the differentiation between scar tissue and local recurrence in women after breast-conserving therapy, and the search for the primary tumor site in women with cancer of unknown primary origin (CUP).
Widely accepted indications for the performance of breast MRI include the differentiation between scar tissue and local recurrence in women after breast-conserving therapy, and the search for the primary tumor site in women with cancer of unknown primary origin (CUP).
 Recent studies have shown that it is expedient to perform breast MRI for preoperative local staging of patients with an invasive lobular carcinoma and for early detection in women with a highly increased breast cancer risk profile. Some national guidelines (e.g., German, American Cancer Society [ACS]) currently include specific recommendations for the use of breast MRI.
Recent studies have shown that it is expedient to perform breast MRI for preoperative local staging of patients with an invasive lobular carcinoma and for early detection in women with a highly increased breast cancer risk profile. Some national guidelines (e.g., German, American Cancer Society [ACS]) currently include specific recommendations for the use of breast MRI.
 Current study data have shown that breast MRI yields significant additional information when performed in the framework of pretherapeutic breast cancer staging, for monitoring during neoadjuvant chemotherapy, and in women with breast implants.
Current study data have shown that breast MRI yields significant additional information when performed in the framework of pretherapeutic breast cancer staging, for monitoring during neoadjuvant chemotherapy, and in women with breast implants.
 The most common use of breast MRI in the diagnostic breast clinic is as a problem solver in cases where ambiguous findings are still present after the diagnostic work-up with clinical examination and/or mammography, and/or breast when ultrasound has been completed.
The most common use of breast MRI in the diagnostic breast clinic is as a problem solver in cases where ambiguous findings are still present after the diagnostic work-up with clinical examination and/or mammography, and/or breast when ultrasound has been completed.
 The significant value of the information provided by breast MRI, especially in comparison with that provided by other breast imaging modalities, makes it an examination that should be considered in the framework of the diagnostic work-up of an ambiguous breast finding and early breast cancer detection. As a consequence, the application of modified examination protocols (e.g., Göttingen Optipack) and the primary use of breast MRI in early breast cancer detection (MRI screening) may provide an alternative to the diagnostic breast examination.
The significant value of the information provided by breast MRI, especially in comparison with that provided by other breast imaging modalities, makes it an examination that should be considered in the framework of the diagnostic work-up of an ambiguous breast finding and early breast cancer detection. As a consequence, the application of modified examination protocols (e.g., Göttingen Optipack) and the primary use of breast MRI in early breast cancer detection (MRI screening) may provide an alternative to the diagnostic breast examination.
Differentiation between Scar and Local Recurrence after Breast-Conserving Therapy
 The recommended interval between a diagnostic excision and a breast MRI examination is 6 months.
The recommended interval between a diagnostic excision and a breast MRI examination is 6 months.
Note: there is great interindividual variability of tissue enhancement after radiation therapy.
Carcinoma of Unknown Primary
 The most common sites of metastatic spread from breast cancer are:
The most common sites of metastatic spread from breast cancer are:
 Lymphogenous locoregional spread: primarily to the axillary, clavicular, and parasternal lymph nodes
Lymphogenous locoregional spread: primarily to the axillary, clavicular, and parasternal lymph nodes
 Hematogenous spread: to bone, lung, liver, and brain.
Hematogenous spread: to bone, lung, liver, and brain.
 When searching for the primary tumor of a locoregional lymph node metastasis, every ipsilateral ambiguous breast MRI finding must be considered a potential primary lesion and be submitted to a diagnostic work-up.
When searching for the primary tumor of a locoregional lymph node metastasis, every ipsilateral ambiguous breast MRI finding must be considered a potential primary lesion and be submitted to a diagnostic work-up.
Preoperative Local Staging
 The purpose of preoperative local MRI staging in breast cancer patients is assessment of:
The purpose of preoperative local MRI staging in breast cancer patients is assessment of:
 Tumor size
Tumor size
 EIC
EIC
 Multifocality
Multifocality
 Multicentricity
Multicentricity
 Contralateral tumor
Contralateral tumor
 Lymph node involvement
Lymph node involvement
Tumor Size
Extensive Intraductal Component
 EIC: Intraductal tumor in the periphery of an invasive breast carcinoma.
EIC: Intraductal tumor in the periphery of an invasive breast carcinoma.
Infiltration of the Pectoral Muscle
Indications for Breast MRI
Only gold members can continue reading. Log In or Register to continue

Full access? Get Clinical Tree