Anesthetic Considerations for Pediatric Surgical Conditions
Risks of Anesthesia
Quantifying the risk of pediatric anesthesia is difficult due to the difficulty in determining whether complications are attributable to the anesthetic, and if so, to what degree. The risk of cardiac arrest for children undergoing anesthesia was estimated in the 1990s to be 1 : 10,000.1,2 However, these studies did not take patient co-morbidity or the surgical condition into consideration. The risk of a healthy child suffering cardiac arrest during myringotomy tube placement is significantly less than the likelihood of a child with complex cardiac disease arresting during a complex cardiac repair.3
A recent review of cardiac arrests in anesthetized children compared 193 events from 1998–2004 to 150 events from 1994–1997.4 A reduction in medication-caused arrests from 37% to 18% was identified, and was attributed to the decline in halothane use (that causes myocardial depression) and the advent of using sevoflurane (that is not associated with myocardial depression). There was also a reduction in unrecognized esophageal intubation as a cause of arrest, due in large part to the advent of end-tidal carbon dioxide (ETCO2) monitoring, pulse oximetry, and an increased awareness of the problem.
Recent large single center reports yield a current estimate of anesthesia-related mortality of 1 : 250,000 in healthy children. To put this into perspective for parents, the risk of a motor vehicle collision on the way to the hospital or surgery center is greater than the risk of death under anesthesia. However, risks of mortality and morbidity are increased in neonates and infants less than one year of age, those who are ASA (American Society of Anesthesiologists) status 3 or greater, and those who require emergency surgery.5
Preoperative Anesthesia Evaluation
All patients presenting for operations under anesthesia benefit greatly from a thorough preanesthetic/preoperative assessment and targeted preparation to optimize any coexisting medical conditions. The ASA physical status (PS) score is a means of communicating the condition of the patient. The PS is not intended to represent operative risk and serves primarily as a common means of communication among care providers (Table 3-1). Any child with an ASA classification of 3 or greater should be seen by an anesthesiologist prior to the day of surgery. This may be modified in cases of hardship due to the distance from the surgical venue or when the patient is well known to the anesthesia service, and the child’s health is unchanged. Finally, outstanding and unresolved medical issues may be significant enough to warrant cancellation of the procedure for optimization of anesthesia and/or further diagnostic workup.
TABLE 3-1
ASA Physical Status Classification
| ASA Classification | Patient Status |
| 1 | A normal healthy patient |
| 2 | A patient with mild systemic disease |
| 3 | A patient with severe systemic disease |
| 4 | A patient with severe systemic disease that is a constant threat to life |
| 5 | A moribund patient who is not expected to survive without the operation |
| 6 | A declared brain-dead patient whose organs are being removed for donor purposes |
| E | An emergency modifier for any ASA classification when failure to immediately correct a medical condition poses risk to life or organ viability |
Criteria for Ambulatory Surgery
Ambulatory surgery comprises 70% or more of the caseload in most pediatric centers. Multiple factors should be considered when evaluating whether a child is suitable for outpatient surgery. Some states regulate the minimum age allowed in an ambulatory surgical center. For example, the minimum age in Pennsylvania is six months. In most cases, the child should be free of severe systemic disease (ASA PS 1 or 2). Other factors that may determine the suitability of a child for outpatient surgery are family and social dynamics. Some institutions utilize a telephone screening evaluation process to determine whether a patient can have their full anesthesia history and physical on the day of surgery rather than being evaluated in a preoperative evaluation clinic prior to surgery.6
Well-controlled systemic illnesses do not necessarily preclude outpatient surgery, but any concerns must be addressed in advance in a cooperative fashion between the surgical and anesthesia services. If a child has a moderate degree of impairment, but the disease is stable and the surgical procedure is of minimal insult, outpatient surgery may be acceptable.
General Principles
In addition to the physical examination, the essential elements of the preoperative assessment in all patients are listed in Box 3-1. Patients and parents may be anxious about recurrence of adverse perianesthetic events such as those listed, and they should be reassured that efforts will be made to prevent these events.
Miscellaneous Conditions
Malignant Hyperthermia Susceptibility
MH is an inherited disorder of skeletal muscle calcium channels, triggered in affected individuals by exposure to inhalational anesthetic agents (e.g., isoflurane, desflurane, sevoflurane), succinylcholine, or both in combination, resulting in an elevation of intracellular calcium. The incidence of an MH crisis is 1 : 15,000 general anesthetics in children. Fifty per cent of patients who have an MH episode have undergone a prior general anesthetic without complication. The resulting MH crisis is characterized by hypermetabolism (fever, hypercarbia, acidosis), electrolyte derangement (hyperkalemia), arrhythmias, and skeletal muscle damage (elevated creatine phosphokinase [CPK]). This constellation of events may lead to death if unrecognized and/or untreated. Dantrolene, which reduces the release of calcium from muscle sarcoplasmic reticulum, when given early in the course of an MH crisis, has significantly improved patient outcomes. With early and appropriate treatment, the mortality is now less than 10%. Current suggested therapy can be remembered using the mnemonic ‘Some Hot Dude Better GIve Iced Fluids Fast” and is summarized in Box 3-2.7 It should be noted that dantrolene must be prepared at the time of use by dissolving in sterile water. It is notoriously difficult to get into solution and the surgeon may be asked to help with this process.
Patients traditionally thought to be MH susceptible are those with a spectrum of muscle diseases listed in Box 3-3. However, many patients who develop MH have a normal history and physical examination. In the past, patients with mitochondrial disorders were thought to be at risk. Anesthetic gases appear safe in this population, but succinylcholine should still be avoided as some patients may have rhabdomyolysis (elevated CPK, hyperkalemia, myoglobinuria) with hyperkalemia without having MH.
Preoperative Fasting Guidelines
Research performed at our institution has demonstrated that intake of clear liquids (i.e., liquids that print can be read through, such as clear apple juice or Pedialyte) up until two hours prior to the induction of anesthesia does not increase the volume or acidity of gastric contents.8 Our policy is to recommend clear liquids until two hours prior to the patient’s scheduled arrival time. Breast milk is allowed up to three hours before arrival for infants up to 12 months of age. Infant formula is allowed until four hours before arrival in infants less than 6 months old, and until six hours before arrival in babies 6–12 months old. All other liquids (including milk), solid food, candy, and gum are not allowed less than eight hours before induction of anesthesia. Although these are the guidelines for our institution, the surgeon should be aware that NPO (nil per os) guidelines are variable and institutionally dependent.
Laboratory Testing
Although serum electrolytes are not routinely screened, electrolytes may be helpful in patients on diuretics. Preoperative glucose should be monitored in insulin-dependent diabetic patients, and also in any patient who has been receiving parenteral nutrition or intravenous (IV) fluids with a dextrose concentration greater than 5% prior to surgery.
Clinical Scenarios and High Risk Populations
Upper Respiratory Tract Infection
One of the most common questions confronting an anesthesiologist is whether to cancel a procedure because of an upper respiratory infection (URI). It is not uncommon for some patients to spend much of their childhood catching, suffering from, or recovering from a URI, with the highest frequency occurring in children under age 6 who attend day care or preschool.9 Patients with a current or recent URI undergoing general anesthesia are theoretically at increased risk for perioperative respiratory complications, including laryngospasm, bronchospasm, and hypoxia, with the youngest patients (<2 years of age) being at greatest risk.10,11 However, anesthetic management may also be tailored to reduce stimulation of a potentially hyper-reactive airway. In addition, cancellation of a procedure imposes an emotional and/or economic burden on patients and families, physicians, and operating rooms. Unless the patient is acutely ill, it is often acceptable to proceed with the anesthetic. Patients with high fever, wheezing, or productive cough may actually have a lower respiratory tract infection and the planned procedure is more likely to be cancelled. Our approach is to discuss the urgency of the scheduled operation with the surgeon, and then to review the risks and benefits of proceeding versus rescheduling with the parents, taking into consideration the possibility that the child may have another URI at the time of the rescheduled procedure. Allowing the parents to participate in the decision-making process (when appropriate) usually leads to mutual satisfaction among all involved parties.
The Former Preterm Infant
Postanesthetic Apnea
The risk of apnea is increased in ex-premature infants because of immaturity of the central and peripheral chemoreceptors with blunted responses to hypoxia and hypercapnia, even without the additional burden of anesthetic/opioid-induced respiratory depression. In addition, anesthetic agents decrease muscle tone in the upper airway, chest wall, and diaphragm, thereby further depressing the ventilatory response to hypoxia and hypercapnia. In the immediate neonatal period, immaturity of the diaphragmatic musculature causes early fatigability, which may also contribute to apnea.12 Although postanesthetic apnea may be brief and resolve either spontaneously or with minor stimulation, in ex-premature infants even brief apnea may result in significant hypoxia. Although most apneic episodes occur within the first two hours after anesthesia, apnea can be seen up to 18 hours postoperatively.
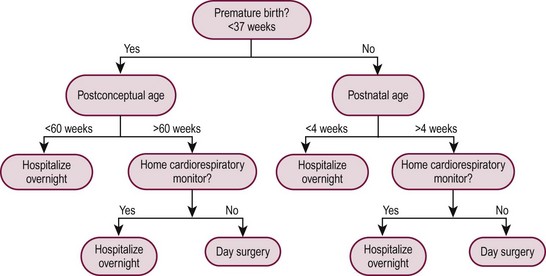
This increased risk of apnea affects the postanesthetic care of infants born prematurely, mandating that those at risk be admitted for cardiorespiratory monitoring. Despite numerous studies on this issue, the postnatal age at which this increased risk of apnea disappears is still being debated. The results of a meta-analysis of pertinent studies indicated that a significant reduction occurred in the incidence of apnea at 52 to 54 weeks’ postconceptual age.13 A hematocrit less than 30% was identified as an independent risk factor, and it was recommended that ex-premature infants with this degree of anemia be hospitalized postoperatively for observation regardless of the postconceptual age. However, conclusions drawn from this meta-analysis have been challenged. Moreover, the sample size of this study may not have been large enough to draw valid conclusions.14
Although the risk of apnea can be decreased with regional anesthesia and/or caffeine, our practice is to admit all at-risk patients (those with a postconceptual age of younger than 60 weeks), regardless of the anesthetic technique used, to monitored, high-surveillance inpatient units for 23 hours after anesthesia and operation. Similarly, infants born at term must be at least 1 month of age to be candidates for outpatient surgery because postanesthetic apnea has been reported in full-term infants up to 44 weeks postconceptual age.13 Figure 3-1 shows an algorithm useful for decision making regarding eligibility for day surgery in young infants.
Anterior Mediastinal Mass
It has long been recognized that the anesthetic management of the child with an anterior mediastinal mass is very challenging and fraught with the risk of sudden airway and cardiovascular collapse. Signs and symptoms of positional airway compression and cardiovascular dysfunction may, or may not, be present. However, the absence of signs and symptoms does not preclude the possibility of life-threatening collapse of the airway or cardiovascular obstruction upon induction of anesthesia. Patients presenting with anterior mediastinal masses (e.g., lymphoma) are at particularly high risk of airway compromise and cardiovascular collapse with the induction of general anesthesia due to compression of the trachea or great vessels when intrinsic muscle tone is lost and spontaneous respiration ceases.15–17 When this occurs, there may not be airway compromise, but rather obstruction of vascular inflow to the right atrium and/or outflow tract obstruction from the right or left ventricle.
Positioning the child is an important part of the anesthetic plan for these patients. The sitting position favors gravitational pull of the tumor toward the abdomen rather than allowing the tumor to fall posteriorly onto the airway and major vessels as occurs in the supine position. However, the sitting position makes intubation challenging. Thus, positioning the symptomatic child in the lateral decubitus position is recommended. Turning the child lateral or prone, or lifting the sternum, have been shown to alleviate acute deterioration in ventilation or cardiovascular collapse secondary to tumor compression.18,19 In any patient with an increased potential for such obstruction, provision should be made for the availability of a rigid bronchoscope, the ability to move the operating room table to effect position changes, and the ability to institute cardiopulmonary bypass or extracorporeal membrane oxygenation (ECMO). Compression of greater than 50% of the cross-sectional area of the trachea on CT imaging has been suggested to identify a population at risk of airway collapse during induction of general anesthesia.20
When possible, percutaneous biopsy of the mass using local anesthesia with or without judicious doses of sedative medication is often ideal and poses the least risk to the patient. In patients who have additional tissue sites from which a biopsy can be obtained (e.g., cervical, axillary, or inguinal lymph nodes), it may be safer to proceed with the patient in a semi-sitting position using local anesthesia and carefully titrating sedation so that spontaneous ventilation is preserved. Recently, ketamine and dexmedetomidine have been shown to provide good sedation with preservation of airway patency and spontaneous respiration in this setting.21 If progression to general anesthesia is required and airway and/or vascular compression exists, standby ECMO capability is strongly recommended.
The inherent conflict between the need to obtain an accurate and timely tissue diagnosis and the very real concern regarding the safe conduct of the anesthetic requires an open dialogue between the anesthesiologist, surgeon, and oncologist to reach an agreement on strategies to achieve these goals. Many experts recommend the development and utilization of an algorithm for anesthetic management of the child with an anterior mediastinal mass (Fig. 3-2). The algorithm addresses assessment of signs and symptoms, evaluation of cardiopulmonary compromise, and treatment options.18,22,23
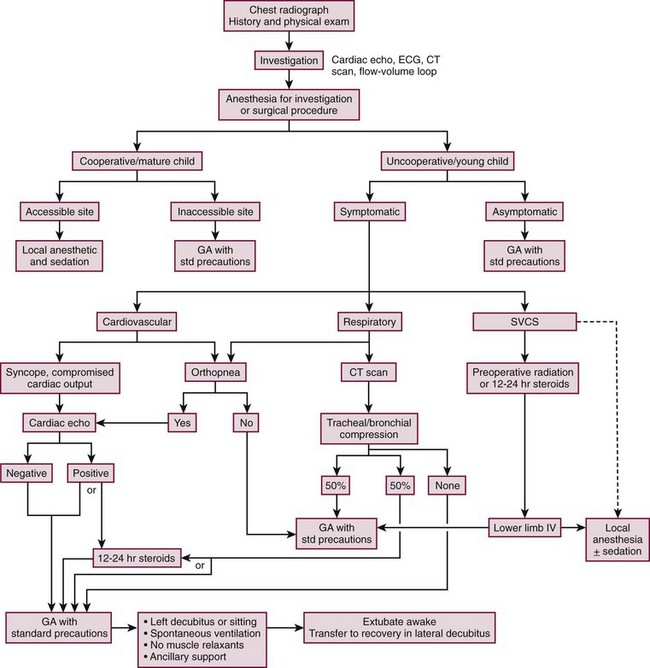
FIGURE 3-2 This algorithm describes management of the patient with a large anterior mediastinal mass. GA, general anesthesia. SVCS, superior vena cava syndrome. (Adapted from Cheung S, Lerman J. Mediastinal masses and anesthesia in children. In: Riazi J, editor. The Difficult Pediatric Airway. Anesthesiol Clin North Am 1998;16:893–910.)
Patients with Congenital Heart Disease
Each year in the U.S., nearly 32,000 children are born with CHD. Extracardiac anomalies are seen in up to 30% of infants with CHD,24,25 and may necessitate operative intervention in the neonatal period prior to repair or palliation of the cardiac lesion. Although physiologically well-compensated patients may undergo noncardiac surgery with minimal risk, certain patient groups have been identified as high risk: children less than 1 year of age, especially premature infants; patients with severe cyanosis, poorly compensated congestive failure or pulmonary hypertension; patients requiring emergency surgery and patients with multiple coexisting diseases.26
Endocarditis Prophylaxis
The most recent American Heart Association (AHA) guidelines for perioperative antibiotic prophylaxis emphasize evidence-based practice. Current opinion reflects the view that endocarditis is more likely to result from frequent exposure to bacteremias occurring as a consequence of activities of daily living than those due to dental, gastrointestinal, or genitourinary tract procedures.27–30 Except for the conditions listed in Box 3-4, the AHA no longer recommends routine antibiotic prophylaxis for any other form of CHD. For a more comprehensive discussion, the reader is referred to the original publications.31,32