Chapter 43 Anatomy of the female genital tract
THE VULVA
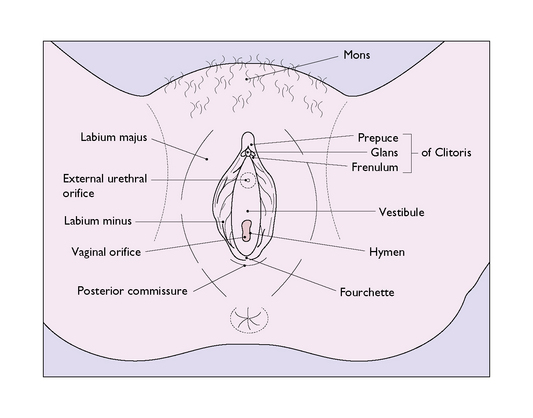
The labia majora are two large folds containing sebaceous and sweat glands embedded in adipose and connective tissue and covered by skin (Fig. 43.1). They form the lateral boundaries of the vulval cleft and are the homologues of the scrotum. Anteriorly, they unite in an adipose pad over the symphysis pubis to form the mons veneris. In the adult the mons is covered with hair, which terminates cephalically in a horizontal upper border. Hair also grows on the outer – but not the inner – surface of the labia majora. Posteriorly, the labia majora unite to form the posterior commissure. In childhood the labia majora contain little adipose tissue, and in older age the adipose tissue disappears. At the extremes of life, therefore, the labia majora are relatively small.
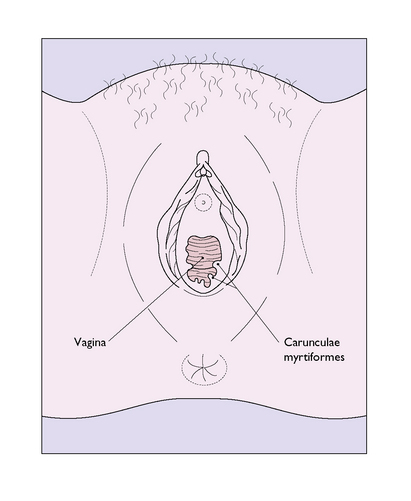
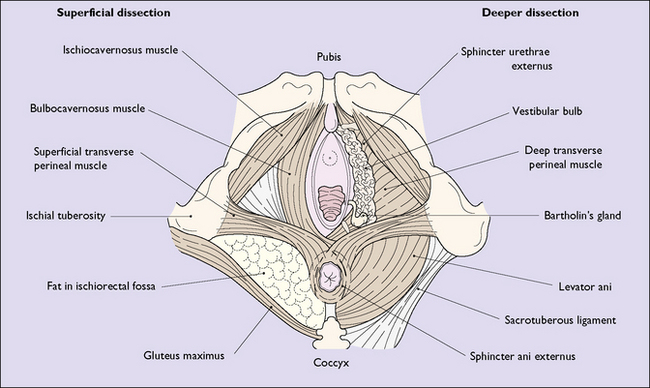
The hymen is a thin, incomplete membrane surrounding the vaginal orifice, and has one or more apertures in it that allow menstrual blood to escape. The apertures are of various shapes and sizes and the hymen varies considerably in elasticity, but is generally torn during a first coitus. An ‘intact’ hymen is considered a sign of virginity, but this is not reliable as in some cases coitus fails to cause a tear, and in others the hymen may be torn by digital interference. In attempting to make a decision regarding virginity, palpation to feel a circular ridge of hymenal tissue is more accurate than inspection. Although the hymen is relatively avascular, tearing at first coitus may be accompanied by a small amount of bleeding, which ceases rapidly. Childbirth causes a much greater tearing of the hymen, and after parturition only a few tags remain. These are known as carunculae myrtiformes (Fig. 43.2). Just lateral to the hymen, surrounding the vaginal orifice on each side and deep to the bulbocavernosus muscle (the sphincter vaginae), are two collections of erectile tissue – the vestibular bulbs. Embedded in the posterolateral parts of the bulbs on each side is Bartholin’s gland (Fig. 43.3), which is the homologue of Cowper’s gland in the male. The gland is pea-sized and not palpable unless infected. It is connected to the posterior part of the vestibule, between the hymen and the fourchette, by a duct some 2 cm in length. It is lined by columnar cells, which secrete a mucoid substance during sexual excitement.
Vascular and nerve supply
Arteries
The external genitalia are very vascular and are supplied by branches of the internal pudendal arteries, which originate from the internal iliac arteries, and by the external pudendal arteries, deriving from the femoral arteries (Fig. 8.23).
Lymphatics
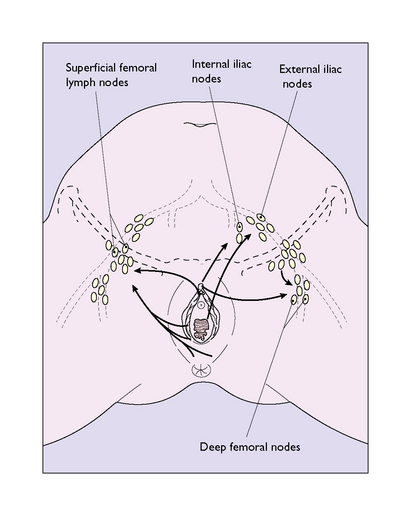
The lymphatics from the clitoral shaft (which interconnect with those of the glans) pass directly to internal iliac nodes in the pelvis (Fig. 43.4). The lymphatics anastomose with those of the opposite side, and consequently bilateral or contralateral involvement is not uncommon in malignant tumours of the vulva. The vulval lymphatics also anastomose with the lymphatics of the lower third of the vagina, which drain into the external iliac nodes.
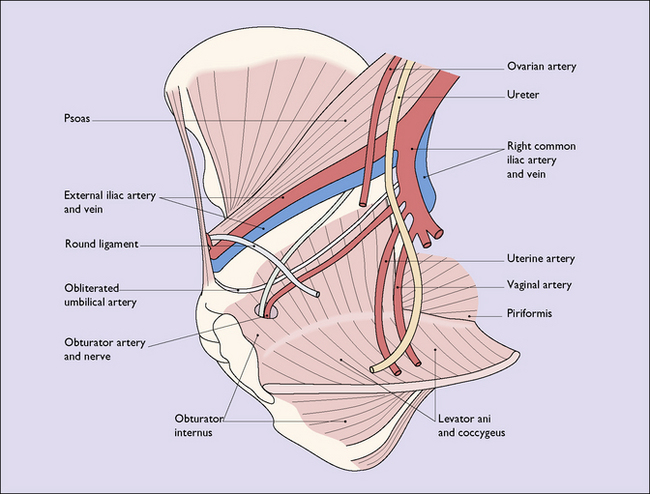
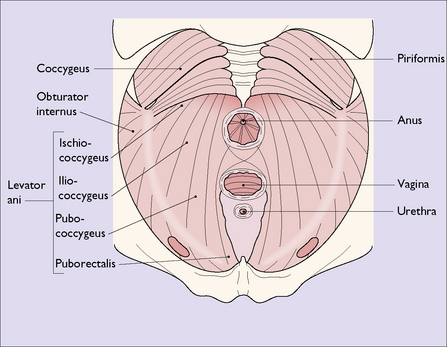
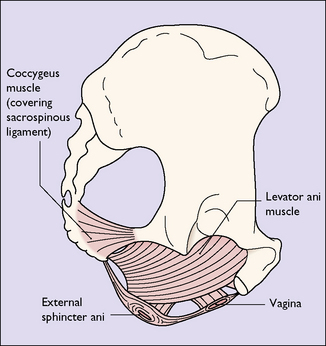
PELVIC FLOOR
The pelvic floor consists of the levator ani group of muscles, which arise on each side of the pelvis, (i) from the posterior surface of the pubis, (ii) from a condensation of fascia (the white line) which covers the obturator internus muscle, and (iii) from the pelvic aspect of the ischial spine (Fig. 43.5). The muscle has several designated parts, the pubococcygeus muscle, the levator ani muscle and the coccygeus muscle (Fig. 43.6). The fibres of these muscles slope downwards and forwards and interdigitate with muscle fibres of the opposite levator ani group to form a muscular sling through which the urethra, the vagina and the rectum pass. The muscles enclosed in fascia form the pelvic diaphragm (Fig. 43.7).
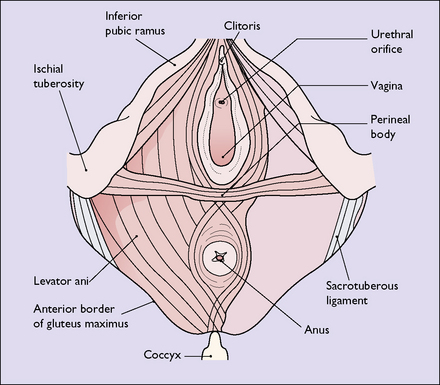
Perineum
The perineum (perineal body) is the tissue that lies distal to the pelvic diaphragm. It is pyramid shaped and is bounded superiorly by the lower surface of the pelvic diaphragm; laterally by the bones and ligaments of the pelvic outlet; and below by the vulva and the anus. It can be subdivided into the urogenital triangle anteriorly and the anal triangle posteriorly by the transverse perineal muscles (Fig. 43.8). The perineum contains a number of superficial muscles, is very vascular and is filled with fatty tissue. Its importance in childbirth is that it is frequently damaged as the fetus is born.
THE VAGINA
The vagina is a fibromuscular sheath extending upwards and backwards from the vestibule, at an angle of about 85° to the horizontal, and parallel with the plane of the pelvic brim when the woman is standing erect (Fig. 43.9). The walls of the vagina, as well as being muscular, contain a well-developed venous plexus. Normally the walls are in apposition, the vagina being a potential cavity and having an H-shape in cross-section in the middle third (Fig. 43.10). In the lower third the widest diameter of the vagina is the anteroposterior, but above this the widest diameter is the transverse. Knowledge of this is important when introducing a vaginal bivalve speculum. Because of the well-developed walls, the lining epithelium tends to be lifted into ridges, or rugae, which run in a circumferential manner from two longitudinal columns running sagittally the length of the anterior and posterior vaginal walls. The formation of rugae in this manner permits the great distension without damage of which the vagina is capable. The length of the anterior vaginal wall is about 7 cm, and its upper end is invaginated by the cervix. Because of this, the posterior vaginal wall, which ends blindly, is about 2 cm longer. The vaginal vault is divided into four areas, which are related to the projecting cervix. These are the shallow anterior fornix, the capacious posterior fornix and the shallow lateral fornices (Fig 43.10). Although the vagina varies considerably in length and width, its functional size is largely determined by the tone in its muscular wall and the contractions of the surrounding muscles. These are under voluntary control. Unless the vagina has been injured or shortened at operation, anatomical variations in size do not cause difficulty or pain (dyspareunia) during coitus; most cases of dyspareunia are psychosomatic in origin.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree