10.3 The dysmorphic child
• Syndrome. This is from the Greek ‘running together’ and refers to a cluster of physical and other features occurring in a consistent pattern, with an implied common specific cause that may be unknown. The word syndrome is often used loosely to describe any of the other diagnostic patterns described below.
• Association. This is a group of physical features that tends to occur together but the link is not consistent enough to allow the term syndrome to be used. An example is the VATER association (see below). The distinction between a syndrome and an association may be artificial and, increasingly, associations are being redefined as syndromes as their genetic basis is identified. For example, in 2004 CHARGE association (Coloboma, Heart defects, Atresia choanae, Retardation of growth and development, Genital and Ear anomalies) was identified as being due to mutations in the CHD7 gene on chromosome 8; with this, CHARGE association became CHARGE syndrome.
• Sequence. This refers to a group of abnormalities caused by a cascade of events beginning with one malformation. An example is the Potter sequence, which can result from any cause of severe oligohydramnios. For example, renal agenesis results in no fetal urine, leading to severe oligohydramnios, with the consequence of lung hypoplasia and intrauterine constraint, causing limb deformities, such as talipes, and a compressed facial appearance.
• Developmental field defect. This refers to a group of malformations caused by a harmful influence in a particular region of the embryo. Abnormalities of blood flow are thought to underlie many of these. An example is hemifacial microsomia with unilateral facial hypoplasia and ear anomalies relating to an abnormality in development of first and second branchial arch structures.
Why is it necessary to recognize an underlying diagnosis? Some important reasons are:
• avoiding unnecessary investigations
• providing information about prognosis for doctors and family
• recognizing and treating complications that need to be looked for prospectively
• determining the pattern of inheritance and recurrence risk
• enabling support from other families. Individual syndromes are rare and parents become the experts in day-to-day management of the child and can share this with other families.
Are there any pitfalls in making a diagnosis? Some areas for consideration are:
• The diagnosis must be correct: the diagnosis may be based on clinical assessment alone with no confirmatory tests available; diagnosis must not be undertaken lightly as it can be difficult to remove or alter a diagnosis once it has been made, with harmful consequences for the child and family.
• Parents do not wish their child to be labelled, especially if the child is young and they do not yet perceive any problems themselves.
• Syndromes are individually rare but collectively common.
• Accurate syndrome diagnosis is important to give information about prognosis, associated problems, recurrence risk and support groups for families.
• Diagnoses are made by thorough clinical assessment followed by use of aids such as computerized syndrome databases.
• Even an experienced clinical geneticist may not make a syndrome diagnosis in a child with dysmorphic features.
• It is better to make no syndrome diagnosis than to make an incorrect diagnosis.
How to assess the dysmorphic child
History
Special points in the medical history include:
Growth and development, behaviour, sleep patterns.
Examination
• height, weight, head circumference and shape, body proportions (e.g. height to arm span ratio, upper and lower body segment length) and any asymmetry
• always plot measurements on standard normal charts. Charts are available for many different body parts (e.g. hand measurements, foot and ear length). Special charts are available for certain disorders (e.g. Down syndrome) and various bone dysplasias (e.g. achondroplasia)
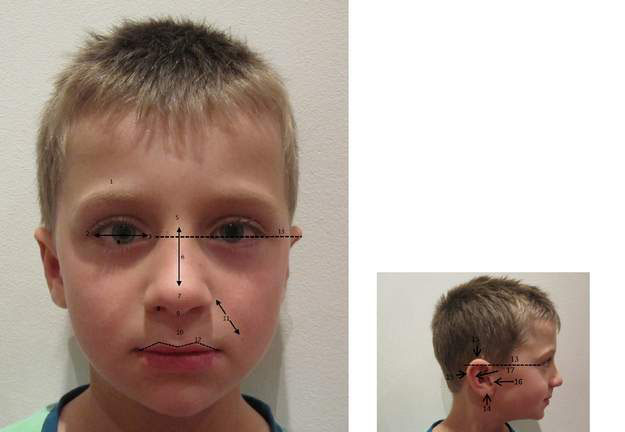
• facial features (Fig. 10.3.1) – careful assessment of facial anatomy often provides clues to the diagnosis
• chest shape and spinal curvature
• proportions of the limbs, muscle bulk and tone, joint contractures and mobility
• structure of hands and feet (shape, length, number of digits, dermal ridges, nails)
• skin pigmentary or vascular markings
• any birth defects? (e.g. cleft palate)
• auscultate the chest for any cardiac murmurs
A photograph (with parental consent) is useful for:
When examining the family of a child with unusual facial features:
Putting it all together
• Check textbooks of syndromes (e.g. Smith’s Recognizable Patterns of Human Malformation) by trying to match the most important dysmorphic features and comparing the photographs with those of the patient.
• It’s not easy! A skilled dysmorphologist makes a syndrome diagnosis in a dysmorphic child in about 30% of cases referred by an experienced paediatrician. If an overall diagnosis is not made, the recognized problems must still be managed.
• Review in 3–5 years’ time is often valuable and may allow a diagnosis to be made as the features of the syndrome evolve over time and new information or laboratory techniques become available.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree