(1)
Department Obstetrics & Gynaecology, St George Hospital, Kogarah, New South Wales, Australia
Abstract
Uterovaginal prolapse is very common. The largest epidemiological study to date (n = 1,547 women interviewed, age 15–79) showed that 8.8 % had symptomatic prolapse and a further 23 % had undergone some form of prolapse surgery MacLennan et al. [12].
Uterovaginal prolapse is very common. The largest epidemiological study to date (n = 1,547 women interviewed, age 15–79) showed that 8.8 % had symptomatic prolapse and a further 23 % had undergone some form of prolapse surgery MacLennan et al. [12].
Nonsurgical Management Options
Until recently, the treatment of symptomatic prolapse has been considered to be largely surgical, with vaginal rings offered to those who were unfit for anesthetic. However, the median lifespan of women in developed countries is now around 83 years old, so many women live well into their 90s. Thus, prolapse is becoming more common. Patients with mild symptoms and mild–moderate prolapse often ask whether they “need” surgery.
There are few data to guide such patients. If a patient has mild asymptomatic prolapse, dealing with the precipitating factors (as per Chap. 1), along with a pelvic floor training program (Chap. 6), may be sufficient. One long-term study of women with asymptomatic rectocele who had no treatment over a median of 8 years found that only new-onset constipation provoked worsening of the prolapse on physical exam [26].
As regards the use of physiotherapy, a large RCT of pelvic floor training versus a leaflet about lifestyle advice has just been concluded in 447 women with mild to moderate prolapse (POPQ stage II 74 %, remainder stage III or stage I). In the active group (4–5 physiotherapy visits over 12 weeks) assessed at 6 months, there were highly significant improvements in symptom severity, quality of life test, and desire for further treatment, compared to control. The change in POPQ was moderate (p = 0.052) [8].
Use of Ring Pessary
In patients with symptomatic prolapse, who decline to have or are totally unfit for surgery, a vaginal ring pessary is very useful in selected cases. The main reasons for which patients are totally unfit for surgery are as follows:
Severe respiratory embarrassment, unable to lie flat without dyspnea
Transplant patients with pelvic kidney, on immunosuppressive drugs
Severe Alzheimer’s disease, unable to tolerate hospitalization
Morbid obesity, poor surgical access to the vagina
Unstable heart disease
Recurrent thromboembolic events, multiple previous stroke
Patients may decline surgery if they are an elderly sole caregiver for an ill husband or if they are sole caregiver for a disabled relative with no suitable respite care. Some women have had unpleasant surgical or anesthetic experiences and do not want another surgical episode. These reasons should be respected, especially if a ring pessary can be easily fitted.
Traditional vaginal ring pessaries (Portex) come in a range of sizes, from 56 to 100 mm diameter. Fitting a ring pessary is like assessing cervical dilatation in labor ward. Insert two fingers into the vagina, spread them apart, and mentally measure the vaginal diameter. In the United States and Australia, Gellhorn pessaries are used for large prolapse, and in the United Kingdom, shelf pessaries are also used. The ring pessary sits anteriorly behind the pubic bone and posteriorly rests on the perineal body.
Hence, if the perineum is very deficient, a ring pessary may not “sit” properly and be extruded during defecation. In a series of 100 patients with prolapse in the United States, 73 % could be fitted satisfactorily [Clemens et al. 5]. A deficient perineum with large introitus was often associated with failure.
In some cases, it may be possible to overcome this by fitting a “double ring,” using the largest ring possible in the upper vagina and the next smaller ring beneath it. This will not solve the problem if the patient has had multiple previous surgeries with scarring/thickening of the walls and vaginal shortening—such women are often very difficult to fit.
Topical vaginal estrogen cream (e.g., Ovestin) should be used three times weekly because the ring pessary is a foreign body which may increase desquamation of the vaginal epithelium, leading to a watery creamy discharge (see Fig. 10.1b). It is traditional to change the ring every 4–5 months, to inspect the vagina to ensure no major vaginal inflammation is occurring. In a snugly fitting pessary, when Ovestin is not used, or when the pessary is not changed regularly, there is a recognized incidence of vaginal bleeding. If this occurs, remove the ring, ask the patient to cleanse the vagina with salt baths twice daily for 5–7 days, and apply Ovestin nightly for 3 weeks. If there is an associated purulent discharge, metronidazole 400 mg TDS for 7 days will resolve this.

Figure 10.1
(a) Portex ring pessary sits anteriorly behind the pubic bone and posteriorly rests on the perineal body. (b) Information diagram about applying Ovestin cream for prolapse
A recent long-term study of 167 women using vaginal ring pessaries up to a median of 7 years showed that over time about 45 % of women may experience bleeding, infection, or both. These women were having the ring changed four monthly; we now teach women to self-insert and remove more often [18].
Surgery for Cystocele
The opening paragraph of the relevant chapter in a World Health Organization monograph on incontinence states that “experts and the majority of published literature suggest the anterior wall is probably the most challenging part of prolapse to cure” Brubaker et al. [4]. This is largely because there are few structures to “anchor” on to. Unlike repair of posterior wall prolapse, in which one can suture onto the sacrospinous ligament or the presacral ligament on the sacral promontory, the pubourethral/pubocervical fascia and paravesical fascia on the undersurface of the pubic rami may be thin and weak. The main surgical options for repair of the anterior wall (also known as the “anterior compartment”) comprise:
Anterior colporrhaphy with plication of the pubourethral and vaginal fascia
Anterior colporrhaphy with more vigorous plication of subpubic fascia
Paravaginal repair (either vaginal or abdominal approach)
Use of mesh to reinforce the anterior colporrhaphy
Anterior Colporrhaphy
The anterior colporrhaphy for cystocele is performed as follows (see Fig. 10.2a):
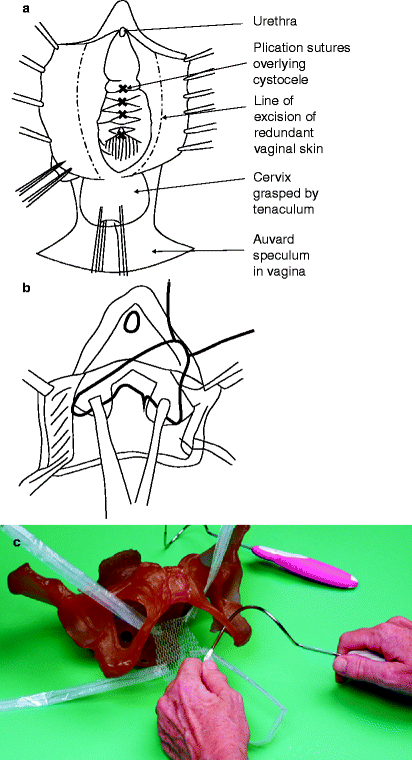

Figure 10.2
(a) Routine anterior colporrhaphy. (b) Ultralateral Anterior Colporrhaphy. (c) Four-cornered mesh kit
Inject local anesthetic with adrenaline into subcutaneous plane of anterior wall.
Dissect vaginal epithelium off the bladder and proximal urethra.
Plicate the paraurethral and paravesical tissue with a sagittal tier of horizontal mattress sutures, without tension.
Trim the redundant vaginal skin sparingly and close.
Insert pack and suprapubic catheter.
The Anterior Repair with Extensive Plication (Ultralateral Anterior Colporrhaphy)
The procedure starts with same dissection of vagina from bladder.
In this case, dissect well back into the pelvis; get under the pubic symphysis.
Place delayed absorbable vertical mattress sutures into the pubourethral/pubocervical or paravaginal fascia that borders the levator hiatus (underneath the pubic bone) to plicate this tissue across the midline under moderate tension, thus replacing the bladder into the abdominal cavity.
Closure with trimming of vaginal skin is identical to anterior colporrhaphy.
A similar procedure involving plication of the pubourethral “ligaments” has been recommended by Nichols [16]; see Fig. 10.2b.
The recurrence rate for cystocele after routine anterior colporrhaphy is up to 40 %. Few series of the more vigorous ultralateral approach have been published. The procedure remains popular because of minimal surgical morbidity.
Paravaginal Defect Repair
This has been the subject of several publications in the last two decades, but these often consider it as a treatment for stress incontinence, rather than for cystocele alone. Because effective operations are available for USI, but cystocele remains a difficult area, a long-term follow-up study of paravaginal repair for cystocele alone is needed.
The paravaginal defect can be repaired transabdominally or vaginally. Most gynecologists would be reluctant to perform an abdominal procedure for an isolated cystocele. If cystocele coexists with stress incontinence, then the colposuspension is highly curative of both. Therefore, transabdominal repair of paravaginal defect is not considered further (but see Shull [19]).
The vaginal approach to paravaginal defect repair is somewhat “challenging,” inasmuch as the obturator internus muscle must be identified first by palpation and then by inspection, so the white line of the arcus tendineus fasciae pelvis can be identified. This involves use of specialized illuminated retractors, to deflect the bladder into the midline. To date, no randomized controlled trials have evaluated the vaginal or abdominal paravaginal repair for cystocele.
Use of Mesh for Cystocele Repair
In 1997, Olsen et al. published a widely cited article showing that 30 % of women who had prolapse surgery in northwest USA ended up having repeat prolapse surgery at some stage [17]. If one reads the article carefully, actually many of the subjects had surgery for incontinence, not prolapse. Also, Olsen did not state whether the repeat surgery was necessarily for a recurrence of the same prolapse or a newly developed prolapse in a different vaginal area. Nevertheless, many gynecologist felt that prolapse surgery needed to be made more durable, and mesh was therefore increasingly employed.
Use of mesh to reinforce the anterior wall has been evaluated by several randomized controlled trials recently. The first compared simple anterior repair, versus anterior repair including use of polyglactin (Vicryl) mesh, versus “ultralateral” anterior repair, in 83 patients reviewed at 2 years. Results (using POPQ and symptom score) revealed that 30 % of the anterior repair group, 42 % of the repair plus mesh, and 46 % of the “ultralateral” repair patients achieved normal vaginal anatomy (POPQ stage 0 or 1). This definition of “cure” is quite strict. The authors pointed out that anterior colporrhaphy often simply does not replace the midpoint of the vagina to a level 3 cm above the introitus. Nevertheless, they concluded that the addition of mesh did not significantly improve cure rates [Weber et al. 25].
An Italian study of polypropylene (Prolene) mesh repair for cystocele in 32 women, at a mean follow-up of 1.5 years, found that dyspareunia was increased by 20 %; 6.5 % of women had mesh erosion. Despite a 94 % anatomical cure rate (using POPQ), the authors concluded that the use of Prolene mesh repair of prolapse should be abandoned because of associated morbidity [Milani et al. 14]. Use of Atrium polypropylene mesh in 64 women with cystocele in Australia yielded 4.7 % erosion rate and recurrence at 2 years in 10 % [Dwyer and O’Reilly 6]. All of these series varied in the selection of patients (primary versus previously failed prolapse surgeries). Over the decade of these published series, the implementation of mesh into a repair had changed. Initially, a square piece of mesh was sutured at the four corners into the vaginal vault, in the manner of Hung et al. [9]. However, a variety of mesh “kits” became available, which are anchored into the vagina by mesh arms protruding from the four corners of the central mesh (See Fig. 10.2c). A high-quality RCT Altman et al. [1] recently showed that in 389 women with primary prolapse, Gynecare Prolift Anterior mesh yielded an 82 % anatomic success compared to 47 % of those having native tissue repair.
These kits were much easier to insert than using the 4-corner suture method; thus, the use of mesh for cystocele repair became increasingly popular. Unfortunately, vaginal mesh is associated with a 5–15 % risk of mesh erosion with a weeping discharge and vaginal discomfort. About 5–10 % of patients experience dyspareunia/apareunia, and some are not able to sit down comfortably. Removal of mesh from the vagina can be quite difficult and sometimes requires more than one operation. In mid-2011, the Food and Drug Administration of the USA issued a product warning regarding the use of vaginal mesh. The FDA was concerned because there had been 1,503 adverse event reports from Jan 2008 to Dec 2010, which was a fivefold increase in such reports over the previous triennium. While this report was perhaps rather strongly worded (see Web site link: http://www.fda.gov), it would seem that use of mesh for primary prolapse repair needs careful discussion with the patient and informed consent must be meticulous. Several authors are currently endeavoring to develop a risk of recurrence score, based upon factors such as a large genital hiatus [23] or a complete avulsion of the subpubic attachment of the levator ani [15], which can be used to select primary prolapse in which mesh would confer a significant advantage. A recent editorial in International Urogynecology Journal, entitled “To mesh or not to mesh?,” gives an excellent summary of the current situation [21].
What Is the Value of Manchester Repair/Retention of a Nonprolapsed Uterus?
In a patient with a cystocele, in whom the cervix is bulky, protuberant, and somewhat elongated, without evidence of actual uterine descent, a Manchester repair may be useful. This comprises anterior colporrhaphy with amputation of the cervix as well as using sutures from the transverse cervical ligaments to enhance elevation of the upper vagina.
The Manchester repair (Fig. 10.3) was developed in the 1950s, at a time when anesthetic risks were greater than now. Thus, a simple procedure to remove an offending organ (the bulky protuberant cervix) without the prolonged anesthesia of a vaginal hysterectomy was attractive.
As anesthetic agents/morbidity improved, a concept evolved that if any part of the uterus/vagina was prolapsing, it should be removed/repaired. The extra time required for a vaginal hysterectomy was no longer an anesthetic issue.
In the last decade, greater scrutiny has been given to the concept of “If any part of the uterus prolapses, remove it all.” A gradually increasing perception of vault prolapse has pervaded the urogynecological community. Laparoscopic procedures to suspend the uterus from the presacral ligament in cases of prolapse (laparoscopic hysteropexy) have been the subject of sporadic reports. Because no large clinical trials have been reported, this procedure is not further discussed. Nevertheless, gynecologists have perhaps appreciated that women do not want their uterus removed unless the evidence proves this will give the best result. Inasmuch as we do not know how to predict vault prolapse, a “fallback” approach may be to leave the uterus intact unless it is truly prolapsed.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


