CHAPTER 79
Increased Intracranial Pressure
Hanalise V. Huff, MD, MPH, and Kenneth R. Huff, MD
CASE STUDY
A 7-year-old boy has a 2-week history of recurrent vomiting. No fever, abdominal pain, or diarrhea has accompanied the vomiting; the vomiting has no particular relationship to meals; and the boy’s appetite has decreased only slightly. The vomiting has gradually increased in frequency and is occurring every night. The day before this visit there were 4 episodes. The boy’s parents have noticed that their son is generally less active; he spends more time playing on the floor of his room and does not want to ride his bicycle or play with neigh-borhood friends. Some unsteadiness in the boy’s gait has manifested in the past few days. His parents attribute this to weakness from the vomiting.
The child’s vital signs are normal except for a blood pressure of 130/80 mm Hg. Although the boy is somewhat pale and uncomfortable, he does not appear to be in acute distress. His abdominal examination is unremarkable. His speech is grammatically correct but sparse and hesitant, and he seems inattentive. On lateral and upward gaze the boy has coarse nystagmus, and upward gaze is somewhat limited. Dysconjugate left gaze is apparent, with slight failure of left eye abduction. The left eye does not blink as much as the right eye. Fundal examination discloses elevated discs with indistinct margins. No upper extremity weakness is evident. The right foot is slightly weaker than the left, ankle tone is bilaterally increased, and 3 to 4 beats of clonus on the right and bilateral positive Babinski reflexes are present. Some tremor occurs in both arms with finger-to-nose testing. The boy walks with shuffling, small steps; his gait has a slight lurching character; and he veers to the right.
Questions
1. What clinical situations are associated with increased intracranial pressure?
2. What is the pathophysiological process leading to increased intracranial pressure?
3. What studies are used to evaluate the child with increased intracranial pressure?
4. What measures are used to treat the child with increased intracranial pressure?
The signs and symptoms of increased intracranial pressure (ICP) often signal a serious, potentially brain damaging intracranial process that may require surgical or intensive care intervention depending on the underlying cause. Recognizing signs and symptoms early often results in a determination of the underlying cause, after which management can resolve the secondarily increased ICP problem. Increased ICP can also be a critical care issue by itself even if the specific etiologic diagnosis is indeterminate. A growing number of effective medical and surgical treatments for increased ICP are available. It is critical to have an understanding about when to initiate them because they can be lifesaving.
Epidemiology
A wide variety of clinical situations, including both acute and subacute processes that occur at all ages, in both sexes, and among all ethnic groups, are commonly responsible for increased ICP. Only a few can be mentioned herein. Traumatic brain injury (TBI) is a leading cause of increased ICP. Fifty-two percent of infants with TBI may be victims of nonaccidental trauma; older children may be stricken pedestrians or bicycle riders, occupants of crashed motor vehicles, or victims of falls or sports injuries; and children of any age may suffer gunshot wounding. Traumatic brain injury is the leading source of trauma mortality and morbidity in children, most often as the result of brain-damaging increases in ICP.
Brain tumors are the most common solid neoplasms in children and frequently result in subacutely increased ICP by direct mass effect or blockage of cerebrospinal fluid (CSF) flow. Diagnosis may sometimes be suggested by neurocutaneous signs or other evidence of tumor suppressor gene mutation. Ischemic brain damage resulting from a difficult delivery at birth, a near drowning incident, or a major intracranial arterial or venous vessel thrombosis is also a significant etiologic contributor to increased ICP. Other causes of brain swelling, such as lead intoxication and liver failure in Reye syndrome, have become less common etiologies.
Idiopathic intracranial hypertension (IIHP, also called benign intracranial hypertension or pseudotumor cerebri), which has an overall prevalence of 1 to 2 per 100,000 but is 20 times higher in adolescent girls with obesity, may ultimately be found as the cause for the increased ICP. It often occurs spontaneously but may also follow the use of high doses of vitamin A, growth hormone, or tetracycline, or it may occur after withdrawal of steroid therapy. Similar physiology is seen with thrombosis of a venous sinus caused by a clotting diathesis or complicated otitis media or mastoiditis.
Clinical Presentation
The child with increased ICP may present with a history of recurrent vomiting, lethargy, and new headaches of increasing frequency or severity (ie, crescendo headaches) or that awaken the child from sleep. The physician must be acutely aware of the clinical situation. A prior history of trauma, ischemia, meningitis, hypertension, or vasculitis; presence of a CSF shunt; or a concomitant history of intoxication or metabolic aberration (ie, carbon monoxide, hyperammonemia, or diabetic ketoacidosis) may also be suspicious for increased ICP in the child with compatible examination findings. Neonates with intraventricular hemorrhage or myelomeningocele or other major central nervous system malformations are prone to hydrocephalus. Children with cyanotic congenital heart disease are prone to cerebral abscesses, and children with sickle cell disease can present with stroke or hemorrhage, resulting in increased ICP. In endemic areas of the world, cerebral malaria and intraventricular cysticercosis are frequent causes of increased ICP. The pertinent physical findings may include elevated optic disc, failure of upward gaze, hypertonicity of the extremities, and either depressed alertness or inattention or severely altered mental status. More localized findings on neurologic examination may also point to a lesion indicative of a space-occupying intracranial mass, which could contribute to increased ICP (Box 79.1).
Pathophysiology
The problem of increased ICP can be understood in terms of the Monro-Kellie doctrine, which applies to the rigid cranial compartment and pressure-volume relationships of the contents. This doctrine is conceptually useful even though not always quantitatively predictive because of the variable compliance of the child’s skull and dural membranes, particularly in the first 2 years after birth before most of the cranial sutures are fused. The skull and dura mater form a relatively rigid compartment; any increase in 1 of the 3 intracranial volume components—brain parenchyma, CSF, and blood— must occur at the expense of 1 or both of the other 2. Decreased volume results in increased pressure in an inverse relationship; the rise becomes much steeper, however, when initial compliance factors are overwhelmed. Irreversible damage to brain tissue occurs primarily as a result of pressure of the other components overtaking the arterial blood pressure and not allowing adequate tissue perfusion. In younger children, nonfused sutures allow more compliance if volume increases are relatively slow, but this factor is less true for acute volume increases. Additionally, pressure gradients exist across compartments or sites of CSF flow obstruction, or even around lesions within brain parenchyma, which results in focal findings in addition to those caused by global ICP or perfusion changes.
Box 79.1. Diagnosis of Increased Intracranial Pressure
•Loss of appetite, nausea, vomiting, headache, or lethargy
•Inattention, decreased ability to arouse
•Full fontanelle, increased head circumference
•Papilledema, upward gaze paresis
•Increased tone, positive Babinski reflex
•Focal signs and history compatible with an intracranial mass
•Mass lesion, cerebral edema, occluded major vessel, or enlarged ventricles on an imaging study
•Elevated cerebrospinal fluid pressure in the lumbar intrathecal or intracranial space as measured using a manometer
Changes in any of the 3 components comprising the intracranial volume may result in increased ICP in several ways. First, the brain parenchyma component may be directly increased by mass lesions, such as neoplasms, abscesses, or hemorrhages. Vasogenic edema may increase the brain parenchyma volume because of vascular leakage due to cytokines. Brain edema may also result from cytotoxic damage, cell death, and necrosis, producing increased interstitial oncotic pressure from released proteins and ions, and cellular inflammatory and repair processes. The immediate cause may be mediated by cellular insults, including hypoxemia; intermediary metabolic toxins, including neuronal excitotoxins; and depletion of energy substrates that are consequential to major vessel occlusion, contusional trauma or traumatic diffuse axonal injury, anoxia from cardiac arrest, hypertensive encephalopathy, encephalitic infection, or external metabolic poisoning. Edema with head trauma is known to be worse in children than in adults and may be a combination of vasogenic and cytotoxic edema and may be related to neurogenic inflammatory release of substance P and calcitonin gene-related peptide at the molecular level close to vessels.
Second, the pressure of the CSF volume component (ventricles or subarachnoid spaces) may increase in the setting of hydrocephalus. Hydrocephalus can result in 2 ways: from a discrepancy in the rate of formation of CSF relative to absorption and from an obstruction between the point of formation in the lateral ventricles and the sites of absorption at the arachnoid granulations. An obstruction can occur with a congenital malformation; a parenchymal or intraventricular mass, such as a cyst or neoplasm; CSF inflammatory cells from meningitis, ventriculitis, or hemorrhage; subarachnoid protein or debris; displaced brain parenchyma from mass effect; or overgrowth of dural tissue. The small passageways connecting the ventricular system, the foramen of Monro, and the aqueduct of Sylvius; the exits of the ventricular system, the foramen of Magendie, and the foramen of Luschka; and the cisterns surrounding the brain stem are particularly vulnerable points of obstruction. Another type of brain edema, interstitial edema, is characterized by periventricular tran-sudation of CSF into the adjacent white matter and generally occurs in the patient with acute or subacute hydrocephalus.
Third, ICP may rise because the intravascular volume component may increase. One process that leads to this increase is venous outflow obstruction, such as with a dural sinus thrombosis. Many patients initially diagnosed as having IIHP are subsequently found to have a diagnosis of transverse sinus stenosis or thrombosis. Other processes that raise jugular venous pressure may also increase ICP. Additionally, the intracranial arterial vascular volume is affected by partial pressure of carbon dioxide. It not only increases with hypercapnia and inadequate ventilation but also decreases with hypocapnia, which occurs with compensatory central neurogenic hyperventilation or iatrogenic reduction of ICP by mechanical hyperventilation.
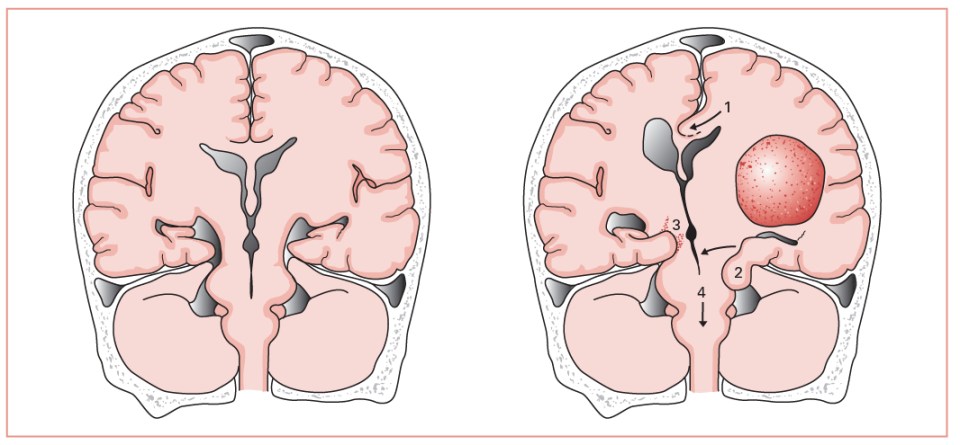
Figure 79.1. Left, Illustration of a normal brain. Right, Illustration of the anatomy of several potential herniation syndromes caused by intracranial compartment pressure gradients related to a mass in a cerebral hemisphere. 1, Transfalcine herniation. 2, Uncal herniation. 3, Contralateral tentorial-midbrain damage. 4, Central herniation and foramen magnum pressure cone. These syndromes often result in further brain ischemia and additional increases in intracranial pressure.
Because the physiology is dynamic, it has proven useful to quantitate ICP for management purposes. Intracranial pressure is often measured as centimeters of water (cm H2O), whereas blood pressure is noted as millimeters of mercury (mm Hg). Normal ICP levels are somewhat lower in the neonatal and infantile period, at approximately 6 cm H2O (5 mm Hg), but in adolescents, pressures above 25 cm H2O (18 mm Hg) are abnormal and may produce symptoms. Although it is possible to have normal cognitive function at an ICP of 52 cm H2O (40 mm Hg), this assumes an adequate perfusion pressure. Perfusion pressure is the mean arterial pressure (MAP) less the ICP. The ICP becomes clinically significant when the perfusion pressure is compromised, which may occur when the ICP is 78 cm H2O (60 mm Hg) below the MAP, which might translate to an ICP as low as 20 mm Hg if the MAP is 80 mm Hg. It can become dangerous when the ICP is only 52 cm H2O (40 mm Hg) below the MAP, which translates to an ICP of 40 mm Hg if the MAP is 80 mm Hg. Decreased perfusion produces swollen, damaged tissue, which increases the brain parenchymal compartment volume and further exacerbates the pressure-volume problem in a cascading fashion. Total loss of brain perfusion occurs when the rise in ICP overtakes and becomes equal to the MAP.
As ICP increases, brain perfusion pressure may be maintained transiently by a spontaneous increase in MAP, a response referred to as the Cushing response (ie, hypertension along with bradycardia and bradypnea). Although the relationship may not be universally reliable, when it is present with other suggestive clinical circumstances, the rise in systemic pressure can be a useful clinical sign of increased ICP. Normally, changes in arterial cerebrovascular resistance meet changes in perfusion pressure to maintain constant cerebral blood flow, a process called autoregulation. This process is frequently compromised after head trauma or asphyxia, however, and is shifted with chronic hypertension.
Acute or subacute changes in pressure within an intracranial compartment may produce a pressure gradient across compartments that may precipitate brain herniation syndrome (Figure 79.1). An ominous heralding sign of transtentorial herniation of the uncus of the temporal lobe is loss of the pupillary light reflex caused by entrapment of cranial nerve III. This herniation often results in irreversible brain stem damage as well as infarcts and additional secondary edema, which can end with brain death. Focally increased posterior fossa pressure may result in a pressure cone downward through the foramen magnum, compressing medullary centers, sequentially extinguishing cranial nerve functions, producing decerebrate posturing, and finally causing apnea and brain death. A marginally compensated system could be decompensated by an ill-advised lumbar puncture when the spinal compartment pressure is acutely decreased, thereby increasing the pressure gradient across the foramen magnum and producing herniation.
Differential Diagnosis
Complicated migraine, seizures, and metabolic derangements are common problems that sometimes have a clinical presentation similar to increased ICP because they may present with headache and altered mental status. A characteristic prodrome or the “pounding” nature of the pain may help separate migraine from increased ICP. At the initial headache presentation or when only a short headache history is present, the complicated migraine diagnosis may be one of exclusion. If the child displays focal neurologic signs with some of the general symptoms of increased ICP, an imaging study to rule out a space-occupying lesion and confirm the safety of a lumbar puncture as well as a subsequent measurement of normal pressure by lumbar puncture manometry may be necessary to support the diagnosis of complicated migraine rather than increased ICP.
In the child who is only partially responsive, the task of distinguishing a seizing or postictal state from a condition that may be producing increased ICP is sometimes difficult. Findings suggestive of a seizure include rhythmic, clonic movements or sudden myoclonic jerks; rapid or variable changes of tone or posturing that are different from the decerebrate posturing that may accompany a process producing increased ICP; abrupt, fluctuating changes of autonomic function (eg, heart rate, blood pressure, pupillary size); saliva production without swallowing; and a history of prior seizures. Sometimes, however, only direct electroencephalography (EEG) monitoring with ICP monitoring can distinguish ongoing electrographic “subclinical” seizure activity from increased ICP as the cause of the change in level of responsiveness.
In some instances, diffuse brain dysfunction from a toxic or metabolic etiology mimics increased ICP. Such toxic or metabolic causes include medication toxicity, electrolyte or blood chemistry imbalance, and systemic infection. With toxic or metabolic disorders, inattention is often accompanied by an acute confusional state with disorientation, incoherence, and sometimes agitation. In contrast, with subacutely increased ICP, inattention frequently is accompanied by slowness of thought, perseveration, decreased mental activity, and impaired gait.
Evaluation
History
A thorough neurologic history should be obtained (Box 79.2). Headache, nausea and vomiting, drowsiness, personality change, declining school performance, and changes in visual acuity and obscurations are important historical factors. The headache history may be one of crescendoing in frequency or intensity, consistent localization, and only a few days’ or weeks’ duration. The headache may awaken the patient from sleep or be worsened by cough, micturition, defecation, or other Valsalva-like maneuvers. A “thunderclap” headache may be indicative of an intracranial hemorrhage. Rapid progression of symptoms generally motivates concern.
Box 79.2. What to Ask
Increased Intracranial Pressure
•How long has the child been vomiting, and when does the vomiting occur?
•Do headaches awaken the child?
•Does the child have weakness or change in gait?
•Does the child have a recent history of trauma?
•Has there been a progressive decline in activity level or loss of developmental skills?
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


