CHAPTER 2 Hysteroscopy
Historical Development
Bozzini was the first to look into the cavity of a hollow organ, the bladder, in 1805. This was achieved with long tubes assisted by external illumination. In 1853, Desormeaux made the first satisfactory endoscope, but it was Panteleoni who performed the first satisfactory hysteroscopy in 1869 (Panteleoni 1869). Inability to distend the uterus and lack of proper illumination caused poor visualization and lack of widespread acceptance until Lindemann renewed Rubin’s attempts to use carbon dioxide in the 1970s (Lindemann 1972).
Diagnostic Indications
Abnormal uterine bleeding
Menstrual abnormality is one of the most common reasons for referral to the gynaecological outpatient department. Menometrorrhagia, dysmenorrhoea and intermenstrual bleeding are the most common. After the menarche, such problems are usually dysfunctional, and hysteroscopy is rarely indicated. In postmenopausal patients, bleeding is a worrying symptom and warrants urgent attention as the patient is anxious to exclude endometrial cancer. Outpatient hysteroscopy offers rapid diagnosis and reassurance. In women of reproductive age, abnormal uterine bleeding can be associated with hormonal disturbances or pathology such as fibroids and polyps. Cervical polyps may be associated with endometrial polyps in 26.7% of patients (Stamatellos et al 2007), and exclusion of endometrial pathology is important.
Infertility
Hysteroscopy is a reliable method to detect and potentially treat submucous fibroids, endometrial polyps, intrauterine adhesions and endometritis. Congenital anomalies are infrequent but are associated with infertility. Abnormalities of the endometrium and organic intrauterine pathologies are important causes of failed in-vitro fertilization/embryo transfer cycles. A recent meta-analysis has shown potential benefits of performing pretreatment hysteroscopy in patients being referred for IVF (El-Toukhy et al 2008).
Recurrent miscarriage
The aetiology of recurrent miscarriage is poorly understood. Intrauterine pathology such as fibroids, polyps, Asherman’s syndrome or congenital abnormalities may be detected in up to 33% of infertile couples (Romano et al 1994). Implantation of an embryo over the septum or fibroid can also fail due to the poor blood supply in these parts of the uterus.
Chronic pelvic pain
Patients with chronic pain can pose a difficult challenge to the gynaecologist. Obstructive uterine anomaly may be associated in 40% of these patients (Schifrin et al 1973). Concomitant hysteroscopy may reveal polyps, fibroids, adhesions and septate uterus which may contribute to the patient’s symptoms.
Contraindications
Cervical stenosis
Patients who have a history of cervical surgery or difficult uterine entry in the past are at increased risk of cervical trauma, perforation and false passage during the performance of a hysteroscopy. Prostaglandins inserted 2 h before hysteroscopy may help to soften the cervix, allowing easy dilatation and entry into the uterine cavity, although the evidence is limited for their value in postmenopausal patients (Crane and Healey 2006).
Instrumentation
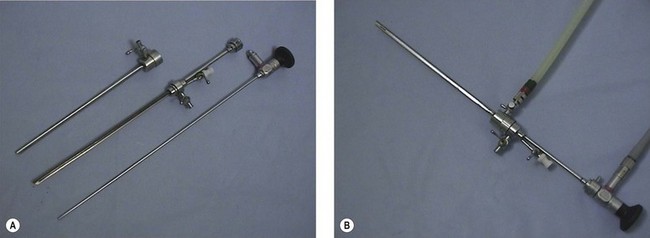
Inadequate instrumentation resulting in poor visualization and reduced safety is not only dangerous, but will potentially give erroneous results. Good-quality hysteroscopes, cameras and driver units are basic requirements for good visualization of the uterine cavity. Appropriate fluid monitoring systems and energy sources, such as laser or diathermy, are essential for operative hysteroscopy (Figure 2.1).
Before commencing any hysteroscopic procedure, one must ensure that the stack system (Figure 2.2) is fully functional. This includes the telescope, surgical instruments, camera drive unit, camera head, light source, light lead, monitor, electrosurgical generator and image recording equipment. If a simultaneous laparoscopy is required, two stack systems should be made available. It is essential to assemble the equipment to ensure that it works prior to use. The distension medium, suction machine, fluid measurement apparatus and connecting tubing must be checked. Fluid should be allowed to flow through the giving set to remove all air bubbles.
Optical systems
Rigid telescopes
Panoramic hysteroscopy is performed with the help of a distension medium, and is common practice. The majority of hysteroscopists use rigid telescopes. Improved technology has allowed significant reduction in the outer diameter (as low as 2.5 mm, including the inflow sheath). The outer diameter of operative hysteroscopes using monopolar electrosurgery may be up to 10 mm. This includes the telescope with the working element, inflow sheath (inner) and outflow sheath (outer). Smaller-diameter operative scopes (Figure 2.3) measure 5 mm and consist of a 5 French operative channel communicating with the outflow sheath. This allows instruments of 5F diameter (e.g. the bipolar electrode, laser fibre, mechanical scissors and grasper) down the operative channel. The telescopes may be forward view (0°) or oblique view (angled scopes 12, 30, 70 or 90°).
Hysteroscopic sheaths
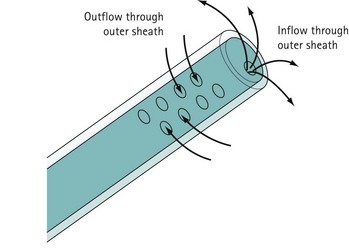
For diagnostic panoramic hysteroscopy, a single sheath for inflow is sufficient. This enables a smaller outer diameter and outpatient hysteroscopy without anaesthesia. Operative hysteroscopy needs separate sheaths for inflow and outflow. The inflow sheath carries the distension medium to the tip of the telescope, from where it is withdrawn via the outer sheath (Figure 2.4). Fluid circulates over the tip of the telescope to maintain a clear view.
Camera and stack system
The hysteroscopic image is visualized on a monitor with the help of a camera connected to a camera drive unit. The image clarity of a single chip camera is perfectly adequate. Special weighted cameras are available and facilitate orientation, but the same camera as is used for laparoscopy is equally appropriate. It is important to ‘white balance’ the camera system to ensure that the colours are displayed correctly (see Figure 2.2).
Technique of Diagnostic Hysteroscopy
Clinical anatomy of the uterus
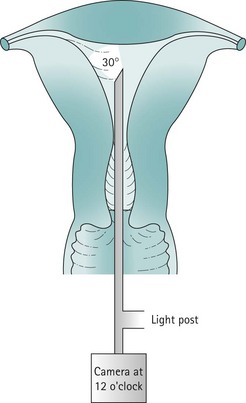
The uterine cavity is flat in its anterior/posterior dimension as, when not distended, the anterior and posterior endometrial surfaces are apposed to each other. The cervical canal is essentially round, but once the cavity is entered, the lateral dimension widens until it is at its broadest at the fundus. The cavity extends laterally towards the tubal ostia (see Figure 2.1). The fundal dome may be flat, concave or convex on its interior surface. Indeed, a proportion of uteri have a pronounced fundal convexity, a so-called ‘arcuate deformity’.
Procedure
Inpatient/general anaesthetic
Potential problems include blood accumulating in the cavity and obscuring the view. This can occur during diagnosis when the seal between the hysteroscope and the cervix is very tight, preventing outflow of distension medium. The passage of a dilator 1 mm greater than the outer diameter of the hysteroscope allows flow and clearance of the contaminating blood. If a large polyp is present in the endometrial cavity, it is possible to inspect the cavity without realizing that the polyp is there because the polyp fills the cavity and the scope has been passed beyond the tip of the polyp before inspection begins (Figure 2.5). Suspicion should be aroused if the endometrial surfaces are different in colour, and careful inspection of the panoramic view of the cavity during insertion and removal of the scope will avoid missing a large polyp as the tip of it will be seen. A thorough examination of the uterine cavity should allow inspection of both tubal ostia. A record of this in the operation notes demonstrates that the operator has obtained a good view.
Hysteroscopy in outpatients
Provision of hysteroscopy combined with transvaginal ultrasound in outpatients provides a very efficient method of assessing women with pre- and postmenopausal bleeding. As mentioned above, many patients who require a hysteroscopy pose significant risk for general anaesthesia, and are best managed under local or no anaesthetic (Valli et al 1998). In addition, there are many advantages to the patient, including shorter time at the hospital and more rapid return to normal activity. There are also cost advantages to the hospital, as an outpatient clinic is clearly a less expensive environment than an operating theatre (Marsh et al 2004).
Choice of equipment
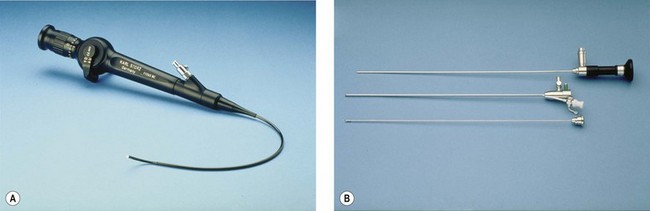
In reality, there are disadvantages and advantages of every system. Rigid autoclavable scopes tend to be of larger diameter, necessitating cervical dilatation in a proportion of cases. The advantage is clarity of view and a fore/oblique view allowing easier examination of the cornua. Flexible scopes are delicate and more expensive, but allow steering through the cervical canal and angulation to allow full inspection of the uterine cavity without any anaesthesia (Kremer et al 1998) (Figure 2.6A). Fibreoptic rigid scopes can be of very small diameter (1.2 mm in a 2.5-mm diagnostic sheath (Figure 2.6B). They are 0° so viewing of the cornua is more difficult, but they are relatively easy to pass through stenosed postmenopausal cervices, leading to a very low failure rate. The Versascope system provides a disposable sheath which can be distended by the passage of a 5 French instrument. This means that a change of sheath is not required to convert a diagnostic procedure into an operative procedure.
Hysteroscopy technique
In the outpatient setting, the authors favour a Cusco’s speculum to bring the cervix into view prior to introduction of the hysteroscope. This allows manipulation of the position of the cervix with the speculum as the scope is inserted to straighten the cervical canal. Rarely, it is necessary to grasp the cervix with a single-toothed tenaculum. Others use no speculum at all when performing vaginoscopy, and locate the cervix before inserting the hysteroscope into it. With the patient awake and the gynaecologist concentrating on passing the scope through a difficult cervix, the role of the attending nurse is paramount (Prather and Wolfe 1995
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree