An ultrasound scan of an ovarian endometrioma. Note the homogenous appearance of the ovarian mass. Picture courtesy of Dr. Yasmin Tan, Women’s Health and Research Centre of Australia.
Endometriosis
This is an estrogen-dependent inflammatory disease that affects around 10% of reproductive-aged women. It is typified by the presence of endometrial-like tissue found outside the endometrial cavity, often associated with bleeding and inflammation (leading to scarring). The inflammation can irritate nerve fibers causing severe pain, as well as damaging adjacent structures such as the fallopian tubes (leading to blockage) or bowel (leading to pain and rectal bleeding). Infertility may result from the factors already mentioned or by adverse effects on uterine receptivity, sperm function or even the embryo itself. There may be a link with ovarian cancer.
Blood levels of CA-125 can be elevated in cases of endometriosis, but the only sure method of diagnosis is visual, usually by laparoscopy. The lesions are typically black or red but can be clear and are often seen on the pelvic peritoneum, especially the Pouch of Douglas, on the ovaries, bladder, bowel and uterine serosa. It can form (chocolate-) cysts within the ovary. There does appear to be a link between endometriosis and other autoimmune conditions [3].
The etiology of endometriosis still remains elusive. One theory speculates that endometrial cells implant on the peritoneum because of retrograde menstruation. Another theory suggests that the disease is the result of celomic metaplasia. It can be inherited in a polygenic way and it often runs in families. Endometriosis usually hyperexpresses ER; whereas PR is usually downregulated and within the lesions, aromatase is hyperexpressed also. Prostaglandins are produced in high amounts by the endometriotic tissue and so contribute to the pain and inflammation.
Simple ovarian cysts
These are benign tumors, greater than 4–5 cm in diameter with a simple thin capsule and no internal solid features. As in younger patients, if they are ovulation-type cysts, they typically resolve in a couple of months. Cystadenomas are increasingly common as women age and are seen in this age group. Most are of a serous or mucinous type. They do not resolve spontaneously, but rather increase in size over time and can become malignant.
Most serous tumors are partly cystic and solid. Inside the cystic component are papillary structures and grossly, they can resemble ovarian cancer but without invasion or atypia. Microscopically, there are small uni- or multi-locular cysts lined by a single layer of tall, columnar, ciliated cells resembling normal tubal epithelium, or cuboidal non-ciliated cells resembling ovarian surface epithelium. In a third of cases they are bilateral. Autoimplants may occur on the ovarian surface that appear identical to non-invasive desmoplastic (serous epithelium with or without Psammoma bodies within abundant fibrous or granulation-like tissue) changes that can occur outside the ovary (e.g. on the fallopian tube).
Mucinous cystadenomas can grow very large and mimic fibroids or even pregnancy. The epithelium is usually tall and columnar with basal nuclei and abundant intracellular mucin. Stroma may be fibrous. Calcifications and microscopic cyst rupture may be present. Around 5% are bilateral. If the tumor ruptures (spontaneously or at surgery), spill into the peritoneal cavity can result in pseudomyxoma peritonei (a gelatinous type of ascites).
Solid or complex ovarian cysts
Solid or complex ovarian cysts are fluid-filled structures with a diameter greater than 4–5 cm which also contain some internal solid tissue (Figure 14.2). A corpus luteum can appear as a complex ovarian cyst but will resolve over time. The differential diagnosis of a growing complex cyst includes a dermoid (teratoma; containing skin, neuronal tissue, teeth, hair etc.), ovarian cancer or an endometrioma (an ovarian cyst of endometriosis; a so-called “chocolate cyst”) [4].
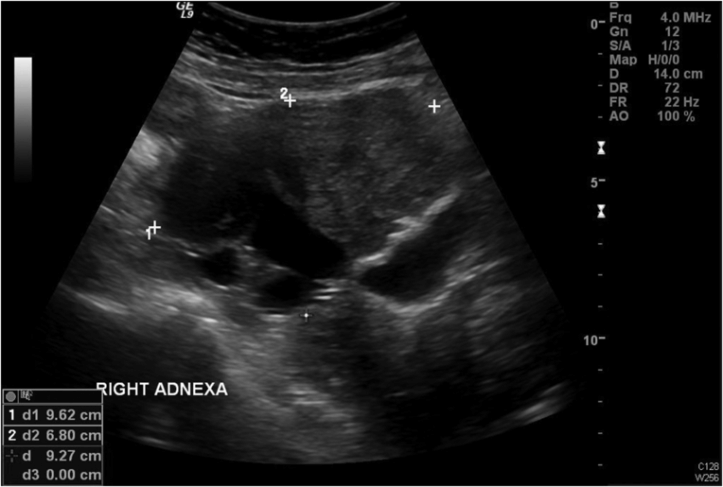
A complex ovarian mass as seen on ultrasound. Fluid shows up as “black” on ultrasound. This mass has both cystic and solid features. Picture courtesy of Dr. Yasmin Tan, Women’s Health and Research Centre of Australia.
Endometrial hyperplasia
This condition is characterized by proliferation and thickening of the endometrium. It is typically due to a prolonged, unopposed estrogen effect, which of course commonly occurs during the perimenopausal years. It is also linked to age over 40 years, tamoxifen usage, and postmenopausal bleeding. It may progress to a variety of degrees of atypia (mild, moderate or severe). In its simplest form, endometrial hyperplasia is microscopically characterized by mild stratification of the endometrium with round glands, perhaps with some cystic dilatation; mitotic figures are occasional and vessel uniform in distribution.
As the disease progresses to complex endometrial hyperplasia (with varying degrees of atypia), the microscopic features become more intricate. This may include rounding of nuclei and nucleoli formation, epithelial stratification, an increase in mitosis rate, cytomegaly and pleomorphism. Endometrial hyperplasia (with or without atypia) may progress to endometrial carcinoma and can often be reversed by moderately high doses of progestins. This usually presents with abnormal uterine bleeding, as will be discussed later.
Endometrial polyps
These are benign lesions that arise from the endometrium on a stalk fed by, often, a single blood vessel (Figure 14.3). They can range from a few millimeters to centimeters in size. They often present as abnormal uterine bleeding and are rarely premalignant (unlike bowel polyps).
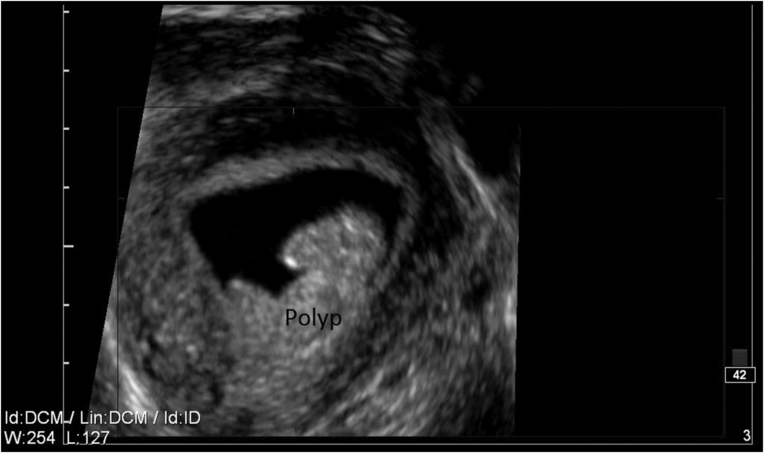
An endometrial polyp as seen on a sonohysterogram. Saline is instilled via the cervix to better define the uterine cavity. Picture courtesy of Dr. Yasmin Tan, Women’s Health and Research Centre of Australia.
Clinical scenarios and diagnostic options
Pelvic pain
Acute sudden and severe pelvic pain suggests either torsion of an ovarian cyst or perhaps rarely, in the setting of a perimenopausal woman, an ectopic pregnancy. Often the patient presents to the emergency ward and is in so much pain that she may be difficult to examine adequately.
Most of the time, pelvic pain is more chronic and often relates to the cycle. Patients with fibroids and/or adenomyosis may describe the pain as a dull ache, or feeling “bloated.” Pressure symptoms from the bladder or rectum may be present. Endometriosis often produces pain that is more sharp and crampy and relates to the menses. Premenstrual spotting or staining and pain with intercourse are also often present with endometriosis. Gynecologic pain is often also referred down the legs and around to the back.
Clinical examination might reveal a large, firm irregular mass arising out of the pelvis (fibroids or ovarian cancer). The presence of ascites would make the diagnosis of ovarian cancer much more likely. If endometriosis is present, often painful nodules can be palpated vaginally (in the Pouch of Douglas, behind the uterus) and in severe cases, the uterus might be fixed in position and non-mobile. Sometimes endometriotic nodules on the rectum can be palpated too.
Blood testing may demonstrate iron deficiency and anemia. Tumor markers such as CA-125 can be elevated in cases of endometriosis as well as some ovarian cancers.
Abdominal and transvaginal ultrasound is the single most useful test. In expert hands, high-quality ultrasound can usually easily diagnose fibroids and describe their location; adenomyosis is often, but not always picked up on ultrasound. In contrast, most patients with endometriosis have a normal scan, unless an endometrioma is present.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


