FIGURE 17-1 Fetal bladder volume measurements together with fetal heart rate (FHR) variation recorded in relation to 1F or 2F behavior states. State 1F fetal heart rate has a narrow bandwidth consistent with quiet sleep. State 2F heart rate shows wide oscillation of the baseline consistent with active sleep. (Modified from Oosterhof, 1993.)
An important determinant of fetal activity appears to be sleep-awake cycles, which are independent of the maternal sleep-awake state. Sleep cyclicity has been described as varying from approximately 20 minutes to as much as 75 minutes. Timor-Tritsch and associates (1978) reported that the mean length of the quiet or inactive state for term fetuses was 23 minutes. Patrick and associates (1982) measured gross fetal body movements with real-time sonography for 24-hour periods in 31 normal pregnancies and found the longest period of inactivity to be 75 minutes. Amnionic fluid volume is another important determinant of fetal activity. Sherer and colleagues (1996) assessed the number of fetal movements in 465 pregnancies during biophysical profile testing in relation to amnionic fluid volume. They observed decreased fetal activity with diminished amnionic volumes and suggested that a restricted uterine space might physically limit fetal movements.
Sadovsky and coworkers (1979b) studied fetal movements in 120 normal pregnancies and classified the movements into three categories according to both maternal perceptions and independent recordings using piezoelectric sensors. Weak, strong, and rolling movements were described, and their relative contributions to total weekly movements throughout the last half of pregnancy were quantified. As pregnancy advances, weak movements decrease and are superseded by more vigorous movements, which increase for several weeks and then subside at term. Presumably, declining amnionic fluid and space account for diminishing activity at term. Figure 17-2 shows fetal movements during the last half of gestation in 127 pregnancies with normal outcomes. The mean number of weekly movements calculated from 12-hour daily recording periods increased from approximately 200 at 20 weeks to a maximum of 575 movements at 32 weeks. Fetal movements then declined to an average of 282 at 40 weeks. Normal weekly maternal counts of fetal movements ranged between 50 and 950, with large daily variations that included counts as low as 4 to 10 per 12-hour period in normal pregnancies.
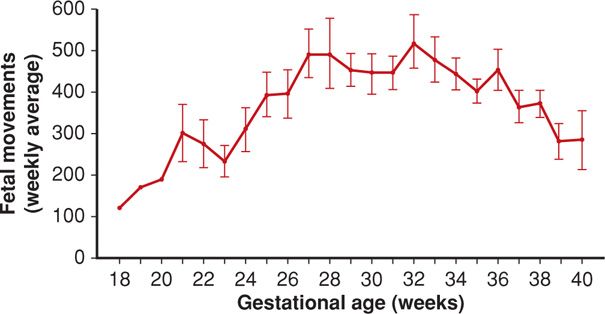
FIGURE 17-2 Graph depicts averages of fetal movements counted during 12-hour periods (mean ± SEM). (Data from Sadovsky, 1979a.)
 Clinical Application
Clinical Application
Diminished fetal activity may be a harbinger of impending fetal death (Sadovsky, 1973). Because of this, various methods have been described to quantify movement of the fetus to forecast its well-being. Methods include use of a tocodynamometer, visualization with sonography, and maternal subjective perceptions. Most, but not all, investigators have reported excellent correlation between maternally perceived fetal motion and movements documented by instrumentation. For example, Rayburn (1980) found that 80 percent of all movements observed during sonographic monitoring were perceived by the mother. In contrast, however, Johnson and colleagues (1992) reported that beyond 36 weeks, mothers perceived only 16 percent of fetal body movements. Fetal motions lasting more than 20 seconds were more likely to be identified than shorter episodes. Although several fetal-movement counting protocols have been used, neither the optimal number of movements nor the ideal duration for counting them has been defined. For example, in one method, perception of 10 fetal movements in up to 2 hours is considered normal (Moore, 1989). In another, women are instructed to count fetal movements for 1 hour a day, and the count is accepted as reassuring if it equals or exceeds a previously established baseline count (Neldam, 1983).
Commonly, women may present in the third trimester complaining of subjectively reduced fetal movement. Harrington and associates (1998) reported that 7 percent of nearly 6800 women presented with a complaint of decreased fetal movement. Fetal heart rate monitoring tests were employed if sonographic scans for fetal growth or Doppler velocimetry were abnormal. Pregnancy outcomes for women who complained of decreased fetal movement were not significantly different from those for women without this complaint. Nonetheless, the authors recommended evaluation to reassure the mother.
Grant and coworkers (1989) performed an unparalleled investigation of maternally perceived fetal movements and pregnancy outcome. More than 68,000 pregnancies were randomly assigned between 28 and 32 weeks. Women in the fetal movement arm of the study were instructed by specially employed midwives to record the time needed to feel 10 movements each day. This required an average of 2.7 hours each day. Women in the control group were informally asked about movements during prenatal visits. Reports of decreased fetal motion were evaluated with tests of fetal well-being. Antepartum death rates for otherwise normal singleton fetuses were similar in the two study groups. Despite the counting policy, most stillborn fetuses were dead by the time the mothers reported for medical attention. Importantly, rather than concluding that maternal perceptions of fetal activity were meaningless, these investigators concluded that informal maternal perceptions were as valuable as formally recorded fetal movement.
The most recent randomized study of fetal movements was performed in Norway by Saastad and colleagues (2011). A total of 1076 women were randomly assigned to standardized fetal movement counting from gestational week 28 or to no counting. Growth-restricted fetuses were identified before birth significantly more often when fetal movement counting was used. There was also a significant reduction (0.4 versus 2.3 percent) in 1-minute Apgar scores ≤ 3 when counting was used. Also, Warrander and associates (2012) performed the first study of placental pathology in pregnancies complicated by decreased fetal movements. Decreased movement was associated with a variety of placental abnormalities including infarction.
FETAL BREATHING
After decades of uncertainty as to whether the fetus normally breathes, Dawes and coworkers (1972) showed small inward and outward flows of tracheal fluid in fetal sheep, indicating thoracic movement. These chest wall movements differed from those following birth in that they were discontinuous. Another interesting feature of fetal respiration was paradoxical chest wall movement, which is depicted in Figure 17-3. In the newborn or adult, the opposite occurs. One interpretation of the paradoxical respiratory motion might be coughing to clear amnionic fluid debris. Although the physiological basis for the breathing reflex is not completely understood, such exchange of amnionic fluid appears to be essential for normal lung development.
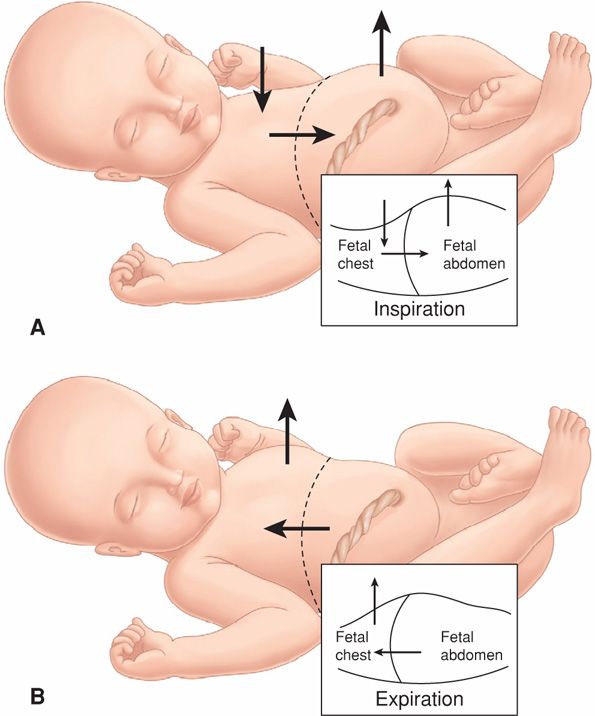
FIGURE 17-3 Paradoxical chest movement with fetal respiration. During inspiration (A), the chest wall paradoxically collapses and the abdomen protrudes, whereas during expiration (B), the chest wall expands. (Adapted from Johnson, 1988.)
Dawes (1974) identified two types of respiratory movements. The first are gasps or sighs, which occurred at a frequency of 1 to 4 per minute. The second, irregular bursts of breathing, occurred at rates up to 240 cycles per minute. These latter rapid respiratory movements were associated with rapid eye movements–REM. Badalian and associates (1993) studied the maturation of normal fetal breathing using color flow and spectral Doppler analysis of nasal fluid flow as an index of lung function. They suggested that fetal respiratory rate decreased in conjunction with increased respiratory volume at 33 to 36 weeks and coincidental with lung maturation.
Many investigators have examined fetal breathing movements using sonography to determine whether chest wall movements might reflect fetal health. Several variables in addition to hypoxia were found to affect fetal respiratory movements. These included hypoglycemia, sound stimuli, cigarette smoking, amniocentesis, impending preterm labor, gestational age, the fetal heart rate itself, and labor—during which it is normal for respiration to cease.
Because fetal breathing movements are episodic, interpretation of fetal health when respirations are absent may be tenuous. Patrick and associates (1980) performed continuous 24-hour observation using sonography to characterize fetal breathing patterns during the last 10 weeks of pregnancy. There was a total of 1224 hours of fetal observation in 51 pregnancies. Figure 17-4 displays the percentages of time spent breathing near term. Clearly, there is diurnal variation, because breathing substantively diminishes during the night. In addition, breathing activity increases somewhat following maternal meals. Total absence of breathing was observed in some of these normal fetuses for up to 122 minutes, indicating that fetal evaluation to diagnose absent respiratory motion may require long periods of observation.
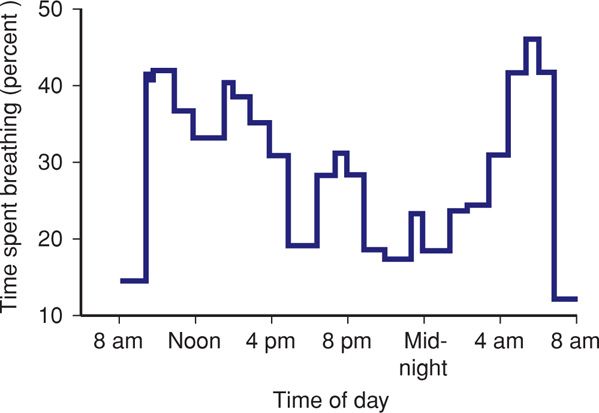
FIGURE 17-4 The percentage of time spent breathing by 11 fetuses at 38 to 39 weeks demonstrated a significant increase in fetal breathing activity after breakfast. Breathing activity diminished during the day and reached its minimum between 8 PM and midnight hours. There was a significant increase in the percentage of time spent breathing between 4 and 7 AM, when mothers were asleep. (Adapted from Patrick, 1980.)
The potential for breathing activity to be an important marker of fetal health is unfulfilled because of the multiplicity of factors that normally affect breathing. Most clinical applications have included assessment of other fetal biophysical indices, such as heart rate. As will be discussed, fetal breathing has become a component of the biophysical profile.
CONTRACTION STRESS TESTING
As amnionic fluid pressure increases with uterine contractions, myometrial pressure exceeds collapsing pressure for vessels coursing through uterine muscle. This ultimately decreases blood flow to the intervillous space. Brief periods of impaired oxygen exchange result, and if uteroplacental pathology is present, these elicit late fetal heart rate decelerations (Chap. 24, p. 483). Contractions also may produce a pattern of variable decelerations as a result of cord compression, suggesting oligohydramnios, which is often a concomitant of placental insufficiency.
Ray and colleagues (1972) used this concept in 66 complicated pregnancies and developed the oxytocin challenge test, which was later called the contraction stress test. Intravenous oxytocin was used to stimulate contractions, and the fetal heart rate response was recorded. The criterion for a positive test result, that is, an abnormal result, was uniform repetitive late fetal heart rate decelerations. These reflected the uterine contraction waveform and had an onset at or beyond the contraction acme. Such late decelerations could be the result of uteroplacental insufficiency. The tests were generally repeated on a weekly basis, and the investigators concluded that negative contraction stress test results, that is, normal results, forecasted fetal health. One disadvantage cited was that the average contraction stress test required 90 minutes to complete.
To perform the test, the fetal heart rate and uterine contractions are recorded simultaneously with an external monitor. If at least three spontaneous contractions of 40 seconds or longer are present in 10 minutes, no uterine stimulation is necessary (American College of Obstetricians and Gynecologists, 2012a). Contractions are induced with either oxytocin or nipple stimulation if there are fewer than three in 10 minutes. For oxytocin use, a dilute intravenous infusion is initiated at a rate of 0.5 mU/min and doubled every 20 minutes until a satisfactory contraction pattern is established (Freeman, 1975). The results of the contraction stress test are interpreted according to the criteria shown in Table 17-1.
TABLE 17-1. Criteria for Interpretation of the Contraction Stress Test
Negative: no late or significant variable decelerations
Positive: late decelerations following 50% or more of contractions (even if the contraction frequency is fewer than three in 10 minutes)
Equivocal-suspicious: intermittent late decelerations or significant variable decelerations
Equivocal-hyperstimulatory: fetal heart rate decelerations that occur in the presence of contractions more frequent than every 2 minutes or lasting longer than 90 seconds
Unsatisfactory: fewer than three contractions in 10 minutes or an uninterpretable tracing
Nipple stimulation to induce uterine contractions is usually successful for contraction stress testing (Huddleston, 1984). One method recommended by the American College of Obstetricians and Gynecologists (2012a) involves a woman rubbing one nipple through her clothing for 2 minutes or until a contraction begins. This 2-minute nipple stimulation ideally will induce a pattern of three contractions per 10 minutes. If not, after a 5-minute interval, she is instructed to retry nipple stimulation to achieve the desired pattern. If this is unsuccessful, then dilute oxytocin may be used. Advantages include reduced cost and shortened testing times. Some have reported unpredictable uterine hyperstimulation and fetal distress, whereas others did not find excessive activity to be harmful (Frager, 1987; Schellpfeffer, 1985).
NONSTRESS TESTS
Freeman (1975) and Lee and colleagues (1975) introduced the nonstress test to describe fetal heart rate acceleration in response to fetal movement as a sign of fetal health. This test involved the use of Doppler-detected fetal heart rate acceleration coincident with fetal movements perceived by the mother. By the end of the 1970s, the nonstress test had become the primary method of testing fetal health. The nonstress test was easier to perform, and normal results were used to further discriminate false-positive contraction stress tests. Simplistically, the nonstress test is primarily a test of fetal condition, and it differs from the contraction stress test, which is a test of uteroplacental function. Currently, nonstress testing is the most widely used primary testing method for assessment of fetal well-being and has also been incorporated into the biophysical profile testing system subsequently discussed.
 Fetal Heart Rate Acceleration
Fetal Heart Rate Acceleration
There are autonomic influences mediated by sympathetic or parasympathetic impulses from brainstem centers that normally increase or decrease the fetal heart rate. Beat-to-beat variability is also under the control of the autonomic nervous system (Matsuura, 1996). Consequently, pathological loss of acceleration may be seen in conjunction with significantly decreased beat-to-beat variability of the fetal heart rate (Chap. 24, p. 479). Loss of such reactivity, however, is most commonly associated with sleep cycles as discussed on page 335. It also may be caused by central depression from medications or cigarette smoking (Jansson, 2005).
The nonstress test is based on the hypothesis that the heart rate of a fetus that is not acidemic as a result of hypoxia or neurological depression will temporarily accelerate in response to fetal movement. Fetal movements during testing are identified by maternal perception and recorded. As hypoxia develops, these fetal heart rate accelerations diminish (Smith, 1988).
Gestational age influences acceleration or reactivity of the fetal heart rate. Pillai and James (1990b) studied the development of fetal heart rate acceleration patterns during normal pregnancy. The percentage of body movements that is accompanied by accelerations and the amplitude of these accelerations increase with gestational age (Fig. 17-5). Guinn and colleagues (1998) studied nonstress test results between 25 and 28 weeks in 188 normal fetuses. Only 70 percent of these normal fetuses demonstrated the required 15 beats per minute (bpm) or more of heart rate acceleration. Lesser degrees of acceleration, that is, 10 bpm, occurred in 90 percent of the fetuses.
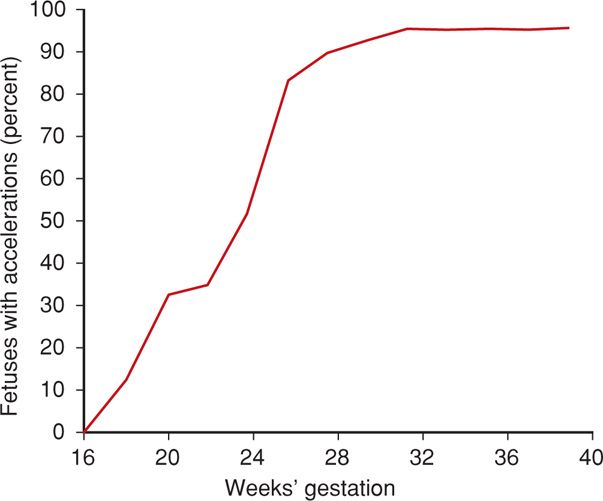
FIGURE 17-5 Percentage of fetuses with at least one acceleration of 15 bpm sustained for 15 seconds concurrent with fetal movement. (Adapted from Pillai, 1990b.)
The National Institute of Child Health and Human Development Fetal Monitoring Workshop defined normal acceleration based on gestational age (Macones, 2008). In fetuses at or beyond 32 weeks, the acceleration acme is 15 bpm or more above the baseline rate, and the acceleration lasts 15 seconds or longer but less than 2 minutes. Before 32 weeks, accelerations are defined as having an acme that is 10 bpm or more above baseline for 10 seconds or longer. Cousins and associates (2012) compared the Workshop criteria recommended before 32 weeks, that is, 10 bpm/10 seconds, with standard 15 bpm/15 seconds criteria in a randomized trial of 143 women. They found no differences in perinatal outcomes.
 Normal Nonstress Tests
Normal Nonstress Tests
There have been many different definitions of normal nonstress test results. They vary as to the number, amplitude, and duration of acceleration, as well as the test duration. The definition currently recommended by the American College of Obstetricians and Gynecologists and the American Academy of Pediatrics (2012) is two or more accelerations that peak at 15 bpm or more above baseline, each lasting 15 seconds or more, and all occurring within 20 minutes of beginning the test (Fig. 17-6). It was also recommended that accelerations with or without fetal movements be accepted, and that a 40-minute or longer tracing—to account for fetal sleep cycles—should be performed before concluding that there was insufficient fetal reactivity. Miller and coworkers (1996b) reviewed outcomes in fetuses with nonstress tests considered as nonreactive because there was only one acceleration. They concluded that one acceleration was just as reliable as two in predicting healthy fetal status.
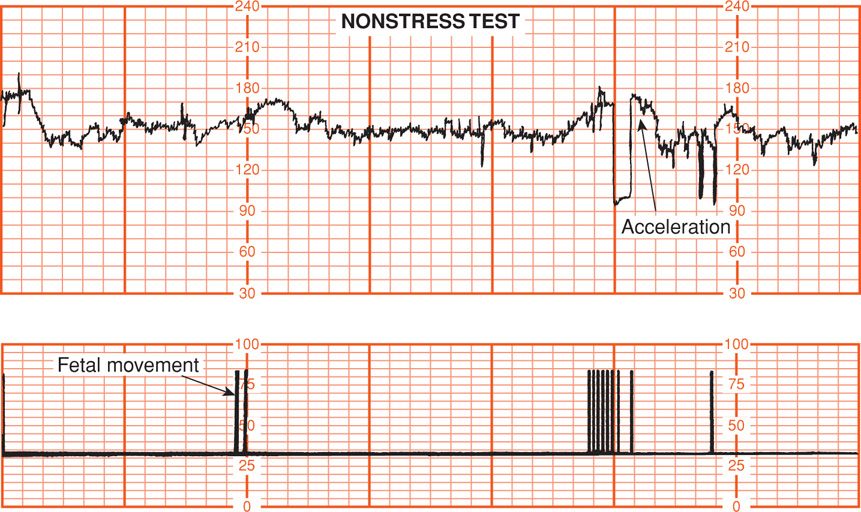
FIGURE 17-6 Reactive nonstress test. In the upper panel, notice the increase of fetal heart rate by more than 15 beats/min for longer than 15 seconds following fetal movements, which are indicated by the vertical marks (lower panel).
Although a normal number and amplitude of accelerations seems to reflect fetal well-being, their absence does not invariably predict fetal compromise. Indeed, some investigators have reported 90-percent or higher false-positive rates (Devoe, 1986). Because healthy fetuses may not move for periods of up to 75 minutes, some have considered that a longer duration of nonstress testing might increase the positive-predictive value of an abnormal, that is, nonreactive, test (Brown, 1981). In this scheme, either the test became reactive during a period up to 80 minutes or the test remained nonreactive for 120 minutes, which indicated that the fetus was very ill.
Not only are there many different definitions of normal nonstress test results, but the reproducibility of interpretations is problematic. For example, Hage (1985) mailed five nonstress tests, blinded to specific patient clinical data, to a national sample of obstetricians for their interpretations. He concluded that although nonstress testing is popular, the reliability of test interpretation needs improvement.
 Abnormal Nonstress Tests
Abnormal Nonstress Tests
Based on the foregoing, an abnormal nonstress test is not always malignant. The example shown in Figure 17-7, however, was ominous. Such a pattern can also be seen with a sleeping fetus. Also, an abnormal test can revert to normal as the fetal condition changes, such as the example shown in Figure 17-8. Importantly, a normal nonstress test can become abnormal if the fetal condition deteriorates. There are abnormal patterns that reliably forecast severe fetal jeopardy. Devoe and coworkers (1985) concluded that nonstress tests that were nonreactive for 90 minutes were almost invariably—93 percent—associated with significant perinatal pathology. Hammacher and coworkers (1968) described tracings with what they termed a silent oscillatory pattern that he considered dangerous. This pattern consisted of a fetal heart rate baseline that oscillated less than 5 bpm and presumably indicated absent acceleration and beat-to-beat variability.
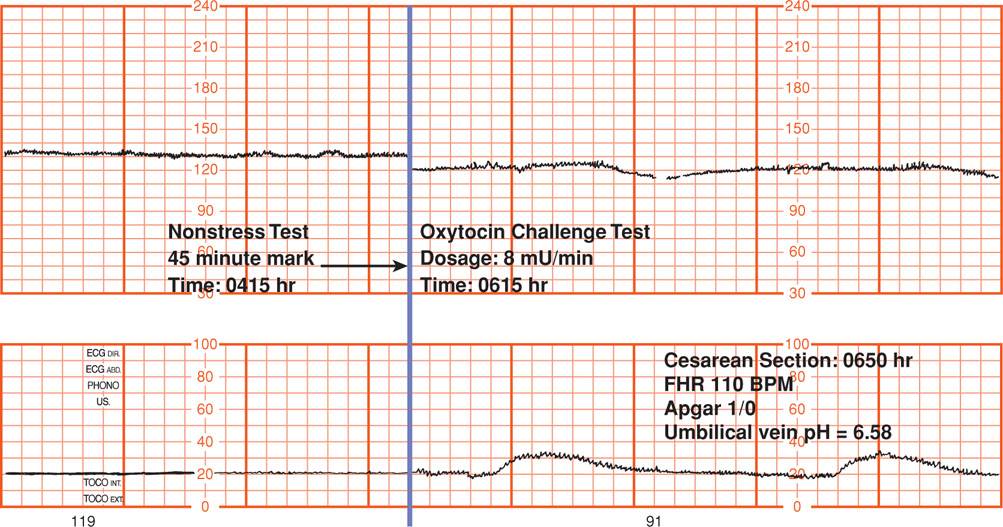
FIGURE 17-7 Nonreactive nonstress test (left side of tracing) followed by contraction stress test showing mild, late decelerations (right side of tracing). Cesarean delivery was performed, and the severely acidemic fetus could not be resuscitated.
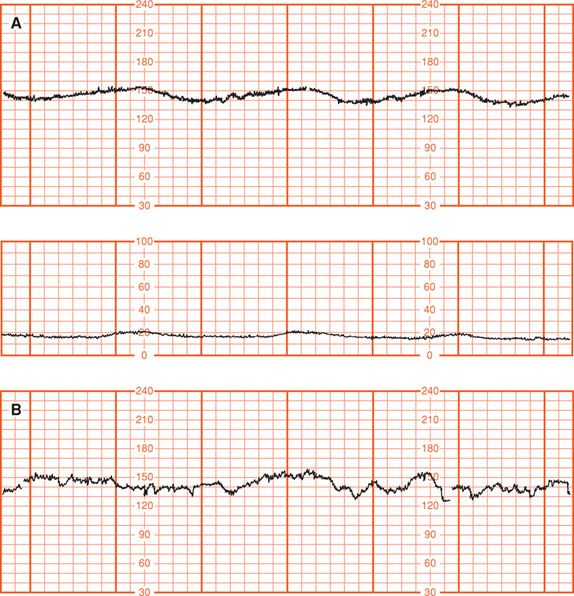
FIGURE 17-8 Two antepartum fetal heart rate (FHR) tracings in a 28-week pregnant woman with diabetic ketoacidosis. A. FHR tracing (upper panel) and accompanying contraction tracing (second panel). Tracing, obtained during maternal and fetal acidemia, shows absence of accelerations, diminished variability, and late decelerations with weak spontaneous contractions. B. FHR tracing shows return of normal accelerations and variability of the fetal heart rate following correction of maternal acidemia.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


