Carrie E. Frederick
Thomas G. Stovall
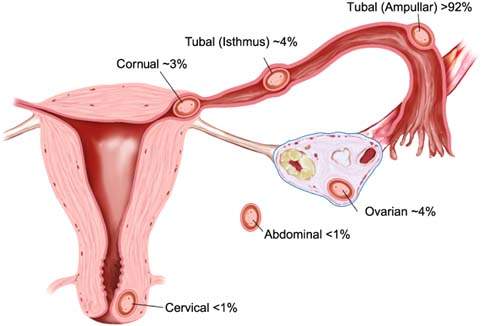
An abnormal gestation can be either intrauterine or extrauterine. Extrauterine or ectopic pregnancy occurs when the fertilized ovum becomes implanted in tissue other than the endometrium. Although 70% of ectopic gestations are located in the ampullary segment of the fallopian tube, such pregnancies may also occur in other sites (Fig. 20.1) (1). Abnormal intrauterine pregnancy often results in pregnancy loss early in gestation. Such losses can be related to a number of factors such as age, previous pregnancy loss, and maternal smoking (Table 20.1). With both abnormal intrauterine and extrauterine gestation, early recognition is key to diagnosis and management.
Table 20.1 Potential Causes of Spontaneous Pregnancy Loss
| Increased maternal age |
| Previous spontaneous abortion |
| Maternal smoking |
| Maternal systemic disease (diabetes mellitus, infection, thrombophilia, etc.) |
| Maternal alcohol consumption (moderate to high) |
| Increasing gravidity |
| Amphetamine use |
| Chromosomal or other embryologic abnormalities |
| Anembryonic gestation |
| Uterine anomalies |
| Intrauterine device in place |
| Placental anomalies |
| Severe maternal trauma |
| Extremes of maternal weight |
Figure 20.1 Common sites of ectopic pregnancy. (Adapted from Seeber BE. Suspected ectopic pregnancy. Obstet Gynecol 2006;107:399–413.)
Abnormal Intrauterine Pregnancy
Spontaneous Abortion
Spontaneous abortion is a pathologic process resulting in unintentional termination of the pregnancy prior to 20 weeks’ gestation. About 8% to 20% of known pregnancies terminate in spontaneous abortion (2,3). About 80% of spontaneous pregnancy losses occur in the first trimester; the incidence decreases with each gestational week (4–6). In women who had one prior spontaneous abortion, the rate of spontaneous abortion in a subsequent pregnancy ranges from 13% to 20%; in women who had three consecutive losses, the rate is 33% (7). Patients should be reassured that, in most cases, spontaneous abortion does not recur. In women less than 36 years of age, when fetal cardiac activity is confirmed by ultrasound, the risk of spontaneous abortion is less than 4.5%. For women older than 36, the risk of spontaneous abortion rises to 10%, and above 40 years may approach 30% (8). Risk factors for spontaneous abortion include increasing maternal age, closely spaced pregnancies (less than 3 to 6 months apart), history of previous spontaneous abortion, maternal diabetes, and maternal smoking during pregnancy (9–13).
With pelvic ultrasound, spontaneous abortion can be differentiated into various categories, based on examination findings and ultrasound findings. Missed abortion is defined as a nonviable intrauterine pregnancy in the presence of a closed cervix and little or no abdominal cramping or vaginal bleeding and can be subdivided into anembryonic gestation and embryonic demise. Anembryonic gestation is a pregnancy where the embryo failed to develop and is confirmed when the mean gestational sac diameter measured by transvaginal ultrasound is greater than 20 mm and no embryonic pole is present. When an embryo is present with crown-rump length greater than 5 mm and no cardiac activity, this is classified as embryonic demise, and the pregnancy is nonviable (14).
Threatened Abortion
Threatened abortion is defined as vaginal bleeding before 20 weeks of gestation. It occurs in at least 20% of all pregnancies (15). The distinction from missed or inevitable abortion requires ultrasound documentation of an intrauterine embryo or fetus with cardiac activity. The bleeding is usually light and may be associated with mild lower abdominal or cramping pain. The differential diagnosis in these patients includes consideration of possible cervical polyps, vaginitis, cervical carcinoma, gestational trophoblastic disease, ectopic pregnancy, trauma, and foreign body. On physical examination, the abdomen usually is not tender, and the cervix is closed. Bleeding can be seen coming from the os, and usually there is no cervical motion or adnexal tenderness.
In the vast majority of cases, threatened abortion does not result in a pregnancy loss, but may be associated with poor outcomes later in pregnancy. In a study of 347 patients with a first-trimester pregnancy documented by ultrasonography, the overall rate of pregnancy loss was 6.1% to 4.2% in patients without bleeding and 12.4% in patients with bleeding (4). In a review of over 800 women presenting with first trimester vaginal bleeding or abdominal pain, nearly 14% with bleeding had spontaneous abortion compared with 2.5% in patients without bleeding (5). There is no effective therapy for a threatened intrauterine pregnancy. Bed rest and progesterone treatment, although often advocated, are not effective (16–18). Women with first trimester vaginal bleeding who do go on to have continuing pregnancies have nearly three times the risk of preterm birth between 28 and 31 weeks as women without bleeding, and a 50% higher likelihood of preterm birth between 32 to 36 weeks (19). First trimester bleeding may predict higher risk for intrauterine growth restriction, preterm premature rupture of membranes, and placental abruption (20). Bacterial vaginosis, if present, should be treated, as this is associated with increased risk for spontaneous abortion (21).
Inevitable Abortion
With an inevitable abortion, the volume of bleeding is often greater than with other types of abortion, and the cervical os is open and effaced, but no tissue has passed. Most patients have crampy lower abdominal pain, and some have cervical motion or adnexal tenderness. When it is certain that the pregnancy is not viable because the cervical os is dilated or excessive bleeding is present, the patient should be offered medical or surgical management. Blood type and Rh determination and a complete blood count should be obtained if there is any concern about the amount of bleeding. Rh0(D) immune globulin (RhoGAM) should be given if the patient’s blood is Rh negative (22). It is acceptable to give a dose of 50 μg [MB2]until 12 completed weeks; if this dose is not available, the standard 300 μg dose may be given.
Incomplete Abortion
An incomplete abortion is a partial expulsion of the pregnancy tissue. Although most patients have vaginal bleeding, only some have passed tissue. Lower abdominal cramping is invariably present, and the pain may be described as resembling labor. On physical examination, the cervix is dilated and effaced, and bleeding is present. Often, clots are admixed with products of conception. If the bleeding is profuse, the patient should be examined promptly for tissue protruding from the cervical os; removal of this tissue with a ring forceps may reduce the bleeding. A vasovagal bradycardia may occur and responds to removal of the tissue. A complete blood count, maternal blood type, and Rh determination should be obtained; Rh-negative patients should receive RhoGAM. If the patient is febrile, broad-spectrum antibiotic therapy should be administered.
Management of Spontaneous Abortion
In women with stable vital signs and mild vaginal bleeding, three management options exist: expectant management, medical treatment, and suction curettage. Despite a wide range (25% to 76%) of success cited in the literature, expectant management may remain a desirable option for a stable and carefully counseled patient (23,24). Medical management with 800 μg of misoprostol placed vaginally can be up to 84% effective in achieving complete abortion (25). For incomplete abortion, the misoprostol dose can be reduced to 600 μg orally or 400 μg sublingually, with efficacy greater than 90% (26). Suction curettage should be performed in women with excessive bleeding, unstable vital signs, or in whom reliable follow-up is a concern.
Ectopic Gestation
Incidence
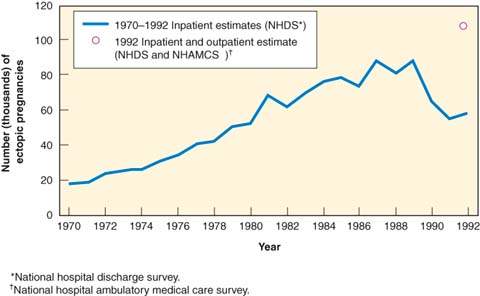
The most comprehensive data available on ectopic pregnancy rates were collected by the Centers for Disease Control and Prevention (CDC). The incidence of ectopic pregnancy increased significantly in the past century. In 1992, the latest year for which statistics are published, there were an estimated 108,800 ectopic pregnancies at a rate of 19.7 ectopic pregnancies per 1,000 reported pregnancies. This represents a sixfold increase compared with 1970 rates. The observed increase may represent an increase in detection and diagnosis resulting from more sensitive ultrasound technology, and a rise in sexually transmitted illnesses and assisted reproductive technologies (27). As Figure 20.2 demonstrates, while the absolute number of ectopic pregnancies continues to rise, the number of hospitalizations declined since the late 1980s, likely because of increasing outpatient management of this condition. Precise estimation of the true incidence of ectopic pregnancy is difficult, but the most recent estimate by the CDC is 2% of reported pregnancies (28). The data on demographic trends indicate that the highest rates occurred in women aged 35 to 44 years (27.2 per 1,000 reported pregnancies). When the data are analyzed by race, the risk for ectopic pregnancy among African Americans and other minorities (20.8 per 1,000) is 1.6 times greater than the risk among whites (13.4 per 1,000) (29). In 1992, 9% of all maternal deaths were attributable to ectopic pregnancy, down from 15% in 1988. The risk for death is higher for African Americans and other minorities than for whites. For all races, teenagers have the highest mortality rates, but the rate for African American and other minority teenagers is almost five times that of white teenagers (28,29). After an ectopic pregnancy, there is an 8% to 15% chance of recurrent ectopic pregnancy, with single-dose methotrexate conferring the lowest risk, while linear salpingostomy is associated with the highest risk (30). Many variables make accurate assessment of risk difficult (e.g., size and location of the ectopic pregnancy, status of the contralateral adnexa, treatment method, and history of infertility).
Figure 20.2 Estimation of the number of ectopic pregnancies (United[MB5]States, 1970–1992). (Adapted from MMWR 1995;44:46–48.)
Etiology and Risk Factors
Ectopic pregnancy results from various factors that interrupt the successful migration of the conceptus to the endometrium. The most important risk factors for ectopic pregnancy are a history of tubal surgery, including tubal ligation, prior ectopic pregnancy, in utero diethylstilbestrol (DES) exposure, and history of pelvic inflammatory disease (31,32). Intrauterine device (IUD) use and infertility are associated with increased risk for ectopic gestation, but these relationships are complex. Up to half of women with ectopic pregnancy will have no identifiable risk factors (33–35). Many other risk factors, including smoking and multiple lifetime sexual partners, are weakly associated with ectopic pregnancy (32).
Myoelectrical activity is responsible for propulsive activity in the fallopian tube (36). This activity facilitates movement of the sperm and ova toward each other and propels the zygote toward the uterine cavity. Estrogen increases smooth muscle activity, and progesterone decreases muscle tone. Aging results in progressive loss of myoelectrical activity along the fallopian tube, which may explain the increased incidence of tubal pregnancy in perimenopausal women (36). Hormonal control of the muscular activity in the fallopian tube may explain the increased incidence of tubal pregnancy associated with failures of the morning-after pill, minipill, progesterone-containing IUDs, and ovulation induction. There is no increase in the incidence of chromosomal abnormalities in ectopic pregnancies (37).
Tubal Surgery
As would be expected, factors that disrupt normal tubal anatomy are the primary etiology for ectopic pregnancy. Women with prior tubal surgery have a more than 20-fold increased risk of subsequent ectopic pregnancy (32). Tubal repair or reconstruction may be performed to correct an obstruction, lyse adhesions, or evacuate an unruptured ectopic pregnancy. Although it is clear that tubal surgery is associated with an increased risk for ectopic pregnancy, it is unclear whether the increased risk results from the surgical procedure or from the underlying problem. A four- to fivefold increased risk is associated with salpingostomy, neosalpingostomy, fimbroplasty, anastomosis, and lysis of complex peritubal and periovarian adhesions (38). After tubal surgery, the overall rate of ectopic pregnancy is 2% to 7%, and the viable intrauterine pregnancy rate is 50% (38).
Though tubal sterilization remains one of the most effective forms of contraception, failures do occur; when they do, they are more likely to result in ectopic gestation. The 10-year cumulative incidence of pregnancy after any form of tubal sterilization is 18.5 per 1,000 woman-years and the likelihood of sterilization failure does not decrease with time since the procedure (39). Despite a greater proportion of poststerilization failures resulting in ectopic pregnancy, the absolute rate of ectopic pregnancy is decreased after sterilization (40). Calculating cumulative lifetime risk for ectopic pregnancy according to method of contraception, sterilized women have a lower cumulative risk for ectopic pregnancy than IUD users or nonusers of contraception, and women using barrier methods or oral contraceptives have the lowest risk (40).
The 10-year cumulative incidence of tubal pregnancy after any sterilization procedure is 7.3 per 1,000 procedures (41). The risk depends on the sterilization technique and the woman’s age at the time of sterilization: postpartum partial salpingectomy and unipolar coagulation have the lowest rates of ectopic pregnancy (1.5 and 1.8 per 1,000 procedures), while bipolar coagulation techniques had the highest incidence (17.1 per 1,000 procedures). Spring clip and band application techniques have 10-year ectopic rates similar to the general incidence, 8.5 and 7.3 per 1,000 procedures, respectively (41). Women younger than 28 years at the time of sterilization are more likely to have a failure than women over 34 years.
Sterilization reversal increases risk for ectopic pregnancy. The exact risk depends on the method of sterilization, site of tubal occlusion, residual tube length, coexisting disease, and surgical technique. In general, the risk for reanastomosis of a cauterized tube is up to 17%, and it ranges from 6% to 9% for reversal of Pomeroy and 5% to 11% for reversal of ring procedures (42–47).
Prior Ectopic Pregnancy
A previous history of ectopic pregnancy is a risk factor for another occurrence. The likelihood of recurrence exists because of the factors that led to an initial ectopic implantation and may be affected by the type of treatment the patient received with the first episode. There is concern that conservation of the tube at the time of removal of an ectopic pregnancy would increase the risk for recurrent ectopic pregnancy (27,48). The rates for intrauterine pregnancy (40%) and ectopic pregnancy (15%, range 4% to 28%) are similar after tubal removal or conservation (49). In a series of 54 patients with conservative surgical procedures for management of ectopic pregnancy, the incidence of future ectopic pregnancy could be predicted by the status of the contralateral tube: normal (7%), abnormal (18%), or absent (25%) (50). In a later study of pregnancy outcomes of 200 patients treated with tubal conservation for ectopic pregnancy, preservation of the tube did not increase the incidence of repeat ectopic pregnancy, but it did improve overall fertility rates (51). The risk of recurrence after methotrexate treatment is similar to that encountered with salpingectomy (52,53). The risk of recurrent ectopic pregnancy after two prior episodes may be as high as 30% (54).
Pelvic Infection
The relationship of pelvic infection, tubal obstruction, and ectopic pregnancy is well documented. In a study of 2,500 women with suspected pelvic inflammatory disease (PID) who underwent diagnostic laparoscopy, the incidence of ectopic pregnancy in the subsequent pregnancy for those with laparoscopically confirmed disease was 9.1% compared with 1.4% in the women with normal laparoscopy (55). In a study of 415 women with laparoscopically proven PID, the incidence of tubal obstruction increased with successive episodes of PID: 13% after one episode, 35% after two, and 75% after three (56).
Chlamydia is an important pathogen causing tubal damage and subsequent tubal pregnancy. Chlamydia was cultured from 7% to 30% of patients with tubal pregnancy (57,58). A strong association between chlamydia infection and tubal pregnancy was shown with serologic tests for chlamydia (59–62). Conception is three times as likely to be tubal in women with anti–Chlamydia trachomatis titers higher than 1:64 than in those women whose titers were negative (63). The number of episodes of chlamydia is directly associated with risk for ectopic pregnancy. In a retrospective cohort study of 11,000 women, those with two chlamydial infections were more than twice as likely to develop ectopic pregnancy as those with one, and women with three or more were at greater than four times higher risk (64). Women at risk for chlamydia infections should be diligently tested, treated when infection is present, and counseled about the risk of ectopic pregnancy.
Diethylstilbestrol
Women exposed to DES in utero who subsequently conceive are at increased risk for ectopic pregnancy. In a review by Goldberg and Falcone, the pooled risk of ectopic pregnancy in DES-exposed women was nine times that of nonexposed women (65). Structural tubal abnormalities were found in DES-exposed women, including foreshortening, constrictions, and distortions (66). The Collaborative Diethylstilbestrol-Adenosis Project, which monitored 327 DES-exposed women, found that about 50% had uterine cavity abnormalities. In DES-exposed women, the risk for ectopic pregnancy was 13% in those who had uterine abnormalities compared with 4% in those who had a normal uterus. No specific type of defect was related to the risk for ectopic pregnancy (67).
Contraceptive Use
It is not surprising that by reducing the overall likelihood of pregnancy, contraceptive use reduces the risk of ectopic pregnancy. There is concern that because of the various mechanisms of action of contraceptives, if a pregnancy were to occur, it might be more likely to be ectopic. In a meta-analysis of 13 studies examining the relationship between contraception and the risk of ectopic pregnancy, there was no increased risk in users of oral contraceptives or barrier methods compared with pregnant controls (40). There is no demonstrated increased risk in users of depomedroxyprogesterone injections, emergency contraceptive pills, or etonogestrel implants (68–70).
Hormonal and copper-containing IUDs are highly effective at preventing both intrauterine and extrauterine pregnancies. Women who conceive with an IUD in place are more likely to have a tubal pregnancy than those not using contraceptives. For women using the levonorgestrel intrauterine device, half of pregnancies will be ectopic, and 1 in 16 pregnancies in women with a copper IUD in place will be ectopic (71). The baseline risk of ectopic pregnancy in women not contracepting is 1 in 50. In a meta-analysis of studies investigating risk of ectopic pregnancy in IUD users compared with nonpregnant controls, IUD use had a protective effect with the exception of one study that showed no effect of IUD use (40). In the same meta-analysis, when IUD users were compared with pregnant controls, IUD use was associated with a significantly increased risk for ectopic pregnancy; the odds ratios ranged from 4.2 to 45. A common odds ratio could not be calculated because of heterogeneity between studies. The study with the most precise point estimate was a multinational case-control study conducted by the World Health Organization involving more than 2,200 women, which found an odds ratio of 4.2 (95% confidence interval, 2.56.9) (72). This suggests that while the intrauterine device decreases the risk of pregnancy overall, if a failure does occur, the device is more successful at preventing intrauterine pregnancy than tubal pregnancy. Past IUD use may slightly increase the risk of ectopic pregnancy. It should be noted that many of the studies that demonstrated this finding were conducted in the 1970s and 1980s, when women may have been using the Dalkon Shield, an IUD strongly associated with PID and ectopic pregnancy (73). The IUDs on the market in recent years are not associated with PID after the immediate insertion period (74).
Other Causes
Prior Abdominal Surgery
Many patients with ectopic pregnancies have a history of previous abdominal surgery. The role of abdominal surgery in ectopic pregnancy is unclear. In one study, there appeared to be no increased risk for cesarean delivery, ovarian surgery, or removal of an unruptured appendix (75). Other studies showed that ovarian cystectomy or wedge resection increases the risk for ectopic pregnancy, presumably because of peritubal scarring (76,77). Although there is agreement that an increased risk for ectopic pregnancy is associated with a ruptured appendix, one study did not confirm this finding (75).
Infertility
The incidence of ectopic pregnancy increases with age and parity, and there is a significant increase in nulliparous women undergoing infertility treatment (27,32,49). For nulliparous women, conceptions after at least 1 year of unprotected intercourse are 2.6 times more likely to be tubal (78). Additional risks for infertile women are associated with specific treatments, including reversal of sterilization, tuboplasty, ovulation induction, and in vitro fertilization (IVF). Various studies examining risk factors for ectopic pregnancy found that infertility increased the odds of tubal pregnancy at least 2.5 times and perhaps as much as 21 times (32).
Hormonal alterations characteristic of clomiphene citrate and gonadotropin ovulation-induction cycles may predispose to tubal implantation. About 1.1% to 4.6% of conceptions associated with ovulation induction are ectopic pregnancies (78–80). In many of these patients, the results of hysterosalpingography are normal, and there is no evidence of intraoperative tubal pathology. Hyperstimulation, with high estrogen levels, may play a role in tubal pregnancy; however, not all studies showed this relationship (81–83).
When the first pregnancy obtained with IVF was a tubal pregnancy; about 2% to 8% of the conceptions are tubal (84). Tubal factor infertility is associated with a further increased risk of 17% (85–88). Predisposing factors are unclear but may include placement of the embryo high in the uterine cavity, fluid reflux into the tube, and a predisposing tubal factor that prevents the refluxed embryo from returning to the uterine cavity.
Smoking
Cigarette smoking is associated with an increased risk for tubal pregnancy in a dose-dependent fashion. A case-control study showed a dose relationship: smokers of more than 20 cigarettes a day had a relative risk of 3.5 compared with nonsmokers, whereas smokers of up to 10 cigarettes had a risk of 2.3 (89). A case-control study in France found similar relative risk estimates (90). Alterations of tubal motility, ciliary activity, and blastocyst implantation are associated with nicotine intake.
Abortion
Multiple studies suggest an association between ectopic pregnancy and spontaneous abortion (79,91). With recurrent spontaneous abortion the risk may be increased up to four times (92). This may reflect a shared risk factor, such as with luteal phase defect. Substantial evidence found no increased risk with elective abortion; one study did find a slightly increased risk, particularly with multiple abortions (93–96).
Salpingitis Isthmica Nodosa
Salpingitis isthmica nodosa (SIN) is a noninflammatory pathologic condition of the tube in which tubal epithelium extends into the myosalpinx and forms a true diverticulum. The incidence in healthy controls is 6% to 11%, but this condition is found more often in the tubes of women with an ectopic pregnancy than in nonpregnant women (97). In one study, 46% of women with isthmic tubal pregnancy were found to have SIN (98). Myometrial electrical activity over the diverticula is abnormal. Whether tubal pregnancy is caused by SIN or whether the association is coincidental is unknown.
Endometriosis or Leiomyomas
Endometriosis or leiomyomas can cause tubal obstruction. Neither is commonly associated with ectopic pregnancy.
Histologic Characteristics
Chorionic villi, usually found in the lumen, are pathognomic findings of tubal pregnancy (99). Gross or microscopic evidence of an embryo is seen in two-thirds of cases. An unruptured tubal pregnancy is characterized by irregular dilation of the tube, with a blue discoloration caused by hematosalpinx. The ectopic pregnancy may not be readily apparent. Bleeding associated with tubal pregnancies is mainly extraluminal but may be luminal (hematosalpinx) and may extrude from the fimbriated end. A hematoma is frequently seen surrounding the distal segment of the tube. Patients who have tubal pregnancies that spontaneously resolved and those treated with methotrexate frequently have an enlargement of the ectopic mass associated with blood clots and extrusion of tissue from the fimbriated end. Hemoperitoneum is nearly always present but is confined to the cul-de-sac unless tubal rupture occurred. The natural progression of tubal pregnancy is either expulsion from the fimbriated end (tubal abortion), involution of the conceptus, or rupture, usually around the eighth gestational week. Some tubal pregnancies form a chronic inflammatory mass that is associated with involution and reestablishment of menses and thus is difficult to diagnose. Extensive histologic sampling may be required to disclose a few ghost villi.
Histologic findings associated with tubal gestation include evidence of chronic salpingitis and SIN. Inflammation associated with salpingitis causes adhesions as a result of fibrin deposition. Healing and cellular organization lead to permanent scarring between folds of tissue. This scarring may allow transport of sperm but not the passage of the larger blastocyst. About 45% of patients with tubal pregnancies have pathologic evidence of prior salpingitis (56).
The cause of SIN is unknown but is speculated to be an adenomyosis-like process or, less likely, inflammation (100). This condition is rare before puberty, indicating a noncongenital origin. Tubal diverticula are identified in about one-half of patients who have ectopic pregnancies, as opposed to 5% of women who do not have ectopic pregnancies (101).
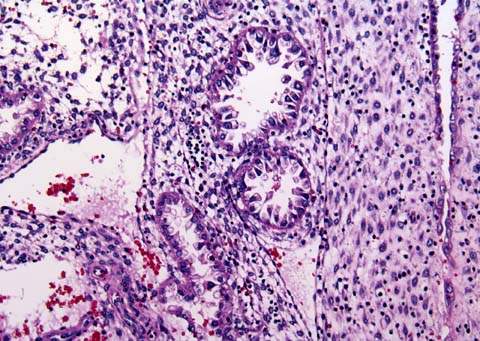
Histologic findings include the Arias-Sella reaction, which is characterized by localized hyperplasia of endometrial glands that are hypersecretory. The cells have enlarged nuclei that are hyperchromatic and irregular (102). The Arias-Sella reaction is a nonspecific finding that can be seen in patients with intrauterine pregnancies (Fig. 20.3).
Figure 20.3 The Arias-Stella reaction of the endometrium. The glands are closely packed and hypersecretory with large, hyperchromatic nuclei suggesting malignancy.
Diagnosis
The diagnosis of ectopic pregnancy is complicated by the wide spectrum of clinical presentations, from asymptomatic cases to acute abdomen and hemodynamic shock. The diagnosis and management of a ruptured ectopic pregnancy is straightforward; the primary goal is achieving hemostasis. If an ectopic pregnancy can be identified before rupture or irreparable tubal damage occurs, consideration may be given to optimizing future fertility. With patients presenting earlier in the disease process, the number of those without symptoms or with minimal symptoms increased. There must be a high degree of suspicion of ectopic pregnancy, especially in areas of high prevalence. History and physical examination identify patients at risk, improving the probability of detection of ectopic pregnancy before rupture occurs.
History
Patients who have an ectopic pregnancy have an abnormal menstrual pattern or the perception of a spontaneous pregnancy loss. Pertinent points in the history include the menstrual history, previous pregnancy, infertility history, current contraceptive status, risk factor assessment, and current symptoms.
The classic symptom triad of ectopic pregnancy is pain, amenorrhea, and vaginal bleeding. This symptom group is present in about 50% of patients and is typical in patients with a ruptured ectopic pregnancy. Abdominal pain is the most common presenting symptom, but the severity and nature of the pain vary widely. There is no pathognomonic pain that is diagnostic of ectopic pregnancy. Pain may be unilateral or bilateral and may occur in the upper or lower abdomen. The pain may be dull, sharp, or crampy and either continuous or intermittent. With rupture, the patient may experience transient relief of the pain, as stretching of the tubal serosa ceases. Shoulder and back pain, thought to result from hemoperitoneal irritation of the diaphragm, may indicate intra-abdominal hemorrhage.
Physical Examination
The physical examination should include measurements of vital signs and examination of the abdomen and pelvis. Frequently, the findings before rupture and hemorrhage are nonspecific, and vital signs are normal. The abdomen may be nontender or mildly tender, with or without rebound. The uterus may be slightly enlarged, with findings similar to a normal pregnancy (103,104). Cervical motion tenderness may or may not be present. An adnexal mass may be palpable in up to 50% of cases, but the mass varies markedly in size, consistency, and tenderness. A palpable mass may be the corpus luteum and not the ectopic pregnancy. With rupture and intra-abdominal hemorrhage, the patient develops tachycardia followed by hypotension. Bowel sounds are decreased or absent. The abdomen is distended, with marked tenderness and rebound tenderness. Cervical motion tenderness is present. Frequently, the findings of the pelvic examination are inadequate because of pain and guarding.
History and physical examination may or may not provide useful diagnostic information. The accuracy of the initial clinical evaluation is less than 50% (105). Additional tests are frequently required to differentiate early viable intrauterine pregnancy or suspected ectopic or abnormal intrauterine pregnancy.
Laboratory Assessment
Quantitative β-human chorionic gonadotropin (β-hCG) measurements are the diagnostic cornerstone for ectopic pregnancy. The hCG enzyme immunoassay, with a sensitivity of 25 mIU/mL, is an accurate screening test for detection of ectopic pregnancy. The assay is positive in virtually all documented ectopic pregnancies.
Reference Standards
There are three reference standards for β-hCG measurement. The World Health Organization introduced the First International Standard (1st IS) in the 1930s. Testing for hCG and its subunits improved over the years. The Second International Standard (2nd IS), introduced in 1964, has varying amounts of β-hCG and β subunits. A purified preparation of β-hCG is now available. Originally referred to as the First International Reference Preparation (1st IRP), the test standard is now referred to as the Third International Standard (3rd IS). Although each standard has its own scale, the 2nd IS is about one-half of the 3rd IS. For example, if a level is reported as 500 mIU/mL (2nd IS), it is equivalent to a level of 1,000 mIU/mL (3rd IS). The assay standard used must be known to interpret hCG results correctly (106). In several recent articles, attention was drawn to a problem known as phantom hCG, in which the presence of heterophile antibodies or proteolytic enzymes causes a false-positive hCG result. Because the antibodies are large glycoproteins, significant quantities of the antibody are not excreted in the urine. In the patient with hCG levels less than 1,000 mIU/mL, a urine pregnancy test should be performed and confirmatory positive results obtained before instituting treatment (107,108).
Doubling Time
The hCG level correlates somewhat with the gestational age (109). During the first 6 weeks of amenorrhea, serum hCG levels increase exponentially. During this period, the doubling time of hCG is relatively constant, regardless of the initial level. After the sixth week of gestation, when hCG levels are higher than 6,000 to 10,000 mIU/mL, the hCG rise is slower and not constant (110).
The hCG doubling time can help to differentiate an ectopic pregnancy from an intrauterine pregnancy—a 66% rise in the hCG level over 48 hours (85% confidence level) represents the lower limit of normal values for viable intrauterine pregnancies (111). About 15% of patients with viable intrauterine pregnancies have less than a 66% rise in hCG level over 48 hours, and a similar percentage with an ectopic pregnancy have more than a 66% rise. If the sampling interval is reduced to 24 hours, the overlap between normal and abnormal pregnancies is even greater. Patients with normal intrauterine pregnancies usually have more than a 50% rise in their hCG levels over 48 hours when the starting level is less than 2,000 mIU/mL. The hCG pattern that is most predictive of an ectopic pregnancy is one that has reached a plateau (a doubling time of more than 7 days). For falling levels, a half-life of less than 1.4 days is rarely associated with an ectopic pregnancy, whereas a half-life of more than 7 days is most predictive of ectopic pregnancy.
Serial hCG levels are usually required when the results of the initial ultrasonography examination are indeterminate (i.e., when there is no evidence of an intrauterine gestation or extrauterine cardiac activity consistent with an ectopic pregnancy). When the hCG level is less than 2,000, doubling time helps to predict viable intrauterine gestation (normal rise) versus nonviability (subnormal rise). With normally rising levels, a second ultrasonography examination is performed when the level is expected (by extrapolation) to reach 2,000 mIU/mL. Abnormally rising levels (less than 2,000 mIU/mL and less than 50% rise over 48 hours) indicate a nonviable pregnancy. The location (i.e., intrauterine versus. extrauterine) must be determined surgically, either by laparoscopy or dilation and curettage. Indeterminate ultrasonography results and an hCG level of less than 2,000 mIU/mL is diagnostic of nonviable gestation, either ectopic pregnancy or a complete abortion. Rapidly falling hCG levels (50% over 48 hours) occur with a completed abortion, whereas with an ectopic pregnancy levels rise or plateau.
Single Human Chorionic Gonadotropin Level
A single hCG measurement has limited usefulness because there is considerable overlap of values between normal and abnormal pregnancies at a given gestational age. The ectopic pregnancy site and hCG level do not correlate (112). Many patients in whom the diagnosis of ectopic pregnancy is being considered are uncertain about their menstrual dates. A single hCG level may be useful when measured by sensitive enzyme immunoassays that, if negative, exclude a diagnosis of ectopic pregnancy. Measurement of a single level may be helpful in predicting pregnancy outcome after timed conceptions using advanced reproductive technology. If the hCG level is more than 300 mIU/mL on day 16 to 18 after artificial insemination, there is an 88% chance of a live birth (113). If the hCG level is less than 300 mIU/mL, the chance of a live birth is only 22%. A single hCG level may facilitate the interpretation of ultrasonography when an intrauterine gestation is not visualized. An hCG level greater than the ultrasound discriminatory zone indicates a possible extrauterine pregnancy. Determination of serial hCG levels may be needed to differentiate an ectopic pregnancy from a completed abortion. Further tests are required for patients in whom ultrasonography examination results are inconclusive and hCG levels are below the discriminatory zone.
Serum Progesterone
The mean serum progesterone level in patients with ectopic pregnancies is lower than that in patients with normal intrauterine pregnancies (114,115). However, in studies of more than 5,000 patients with first-trimester pregnancies, a spectrum of progesterone levels was found in patients with both normal and abnormal pregnancies (35,116,117). About 70% of patients with a viable intrauterine pregnancy have serum progesterone levels higher than 25 ng/mL, whereas only 1.5% of patients with ectopic pregnancies have serum progesterone levels higher than 25 ng/mL, and most of these pregnancies exhibit cardiac activity (35,116,117).
A serum progesterone level can be used as an ectopic pregnancy screening test for both normal and abnormal pregnancy, particularly in settings in which hCG levels and ultrasonography are not readily available. A serum progesterone level of less than 5 ng/mL is highly suggestive of an abnormal pregnancy, but it is not 100% predictive. The risk of a normal pregnancy with a serum progesterone level of less than 5 ng/mL is about 1 in 1,500 (118). Serum progesterone measurements alone cannot be used to predict pregnancy nonviability.
Other Endocrinologic Markers
In an effort to improve early detection of ectopic pregnancy, various endocrinologic and protein markers were studied. Estradiol levels increase slowly from conception until 6 weeks of gestation and then rise rapidly as placental production of estradiol increases (119). Estradiol levels are significantly lower in ectopic pregnancies when compared with viable pregnancies. However, there is considerable overlap between normal and abnormal pregnancies and between intrauterine and extrauterine pregnancies (120,121).
Maternal serum creatinine kinase was studied as a marker for ectopic pregnancy diagnosis (122). Maternal serum creatine kinase levels were significantly higher in all patients with tubal pregnancy when compared with those in patients who had missed abortions or normal intrauterine pregnancies. No correlation was found between the creatine kinase level and the clinical presentation of the patient, and there was no correlation with the hCG levels. Schwangerschafts protein 1 (SP1), also known as pregnancy-associated plasma protein C (PAPP-C) or pregnancy-specific β-glycoprotein (PSBS), is produced by the syncytiotrophoblast (120). The main advantage of SP1 level assessment may be in the diagnosis of conception after recent hCG administration. A level of 2 ng/L might be used for the diagnosis of pregnancy; however, it is doubtful that a diagnosis can be established before delay of menses. Although the level of SP1 increases late in all patients with a nonviable pregnancy, a single SP1 level assessment does not have prognostic value (123).
Relaxin is a protein hormone produced solely by the corpus luteum of pregnancy. It appears in the maternal serum at 4 to 5 weeks of gestation, peaks at about 10 weeks of gestation, and decreases until term (124). Relaxin levels are significantly lower in ectopic pregnancies and spontaneous abortions than in normal intrauterine pregnancies. Prorenin and active renin levels are significantly higher in viable intrauterine pregnancies than in either ectopic pregnancies or spontaneous abortions, with a single level of more than 33 pg/mL excluding the diagnosis of ectopic pregnancy (125). The clinical utility of relaxin, prorenin, and renin levels in diagnosing ectopic pregnancy is not yet determined.
CA125 is a glycoprotein, the origin of which is uncertain during pregnancy. Levels of CA125 rise during the first trimester and return to a nonpregnancy range during the second and third trimesters. After delivery, maternal serum concentrations increase (126,127). Levels of CA125 were studied in an effort to predict spontaneous abortion. Although a positive correlation was found between elevated CA125 levels 18 to 22 days after conception and spontaneous abortion, repeat measurements at 6 weeks of gestation did not correlate with outcome (128). Conflicting results were reported—one study showed a higher serum CA125 level in normal pregnancies than in ectopic pregnancies 2 to 4 weeks after a missed menses, whereas another study found higher CA125 levels for ectopic pregnancies compared with normal pregnancies (129,130).
Maternal serum α-fetoprotein (AFP) levels are elevated in ectopic pregnancies; however, the use of AFP measurements as a screening technique for ectopic pregnancy was not studied (131,132). A combination of AFP with three other markers—β-hCG, progesterone, and estradiol—has 98.5% specificity and 94.5% accuracy for the prediction of ectopic pregnancy (133,134). Serum placental growth factor may prove to be a diagnostic biomarker for ectopic pregnancy because it was shown to be undetectable in ectopic and nonviable pregnancies.
C-reactive protein is an acute-phase reactant that increases with trauma or infection. Levels of this protein are lower in patients with ectopic pregnancy than in patients with an acute infectious process. When an infectious process is part of the differential diagnosis, measurement of C-reactive protein may be beneficial (135).
Ultrasonography
Improvements in ultrasonography resulted in the earlier diagnosis of intrauterine and ectopic gestations (136). The sensitivity of the β-hCG assay allows the diagnosis of pregnancy before direct visualization by ultrasonography.
The complete examination should include both transvaginal and transabdominal ultrasonography. Transvaginal ultrasonography is superior to transabdominal ultrasonography in evaluating intrapelvic structures. The closeness of the vaginal probe to the pelvic organs allows use of higher frequencies (5–7 mHz), which improves resolution. Intrauterine pregnancy can be diagnosed 1 week earlier with transvaginal than with transabdominal ultrasonography. Evidence of an empty uterus, detection of adnexal masses and free peritoneal fluid, and direct signs of ectopic pregnancy are more reliably established with a transvaginal procedure (137–142). Transabdominal ultrasonography permits visualization of both the pelvis and abdominal cavity and should be included as part of the complete ectopic pregnancy evaluation to detect adnexal masses and hemoperitoneum.
The earliest ultrasonographic finding of an intrauterine pregnancy is a small fluid space and the gestational sac, surrounded by a thick echogenic ring, located eccentrically within the endometrial cavity. The earliest normal gestational sac is seen at 5 weeks of gestation with transabdominal ultrasonography and at 4 weeks of gestation with transvaginal ultrasonography (143,144). As the gestational sac grows, a yolk sac is seen within it, followed by an embryo with cardiac activity.
The appearance of a normal gestational sac may be simulated by intrauterine fluid collection, the pseudogestational sac, which occurs in 8% to 29% of patients with ectopic pregnancy (145–147). This ultrasonographic lucency, centrally located, probably represents bleeding into the endometrial cavity by the decidual cast. Clots within this lucency may mimic a fetal pole.
Morphologically, identification of the double decidual sac sign (DDSS) is the best method of ultrasonographically differentiating true sacs from pseudosacs (148). The double sac, believed to be the decidua capsularis and parietalis, is seen as two concentric echogenic rings separated by a hypoechogenic space. Although useful, this approach has some limitations in sensitivity and specificity—the DDSS sensitivity ranges from 64% to 95% (147). Pseudosacs may occasionally appear as the DDSS; intrauterine sacs of failed pregnancies may appear as pseudosacs.
The appearance of a yolk sac within the gestational sac is superior to the DDSS in confirming intrauterine pregnancy (149). The yolk sac is consistently visible on transabdominal ultrasonography with a gestational sac size of 2 cm and on transvaginal ultrasonography with a gestational sac size of 0.6 to 0.8 cm (150,151). Intrauterine sacs smaller than 1 cm on transabdominal ultrasonography and smaller than 0.6 cm on transvaginal ultrasonography are considered indeterminate. Larger sacs without DDSS or yolk sac represent either a failed intrauterine or ectopic pregnancy.
The presence of cardiac activity within the uterine cavity is definitive evidence of an intrauterine pregnancy. This finding essentially eliminates the diagnosis of ectopic pregnancy because the incidence of combined intrauterine and extrauterine pregnancy is 1 in 30,000.
The presence of an adnexal gestational sac with a fetal pole and cardiac activity is the most specific but least sensitive sign of ectopic pregnancy, occurring in only 10% to 17% of cases (135,152,153). The recognition of other characteristics of ectopic pregnancy improves ultrasonographic sensitivity. Adnexal rings (fluid sacs with thick echogenic rings) that have a yolk sac or nonliving embryo are accepted as specific ultrasonographic signs of ectopic pregnancy (154). Adnexal rings are visualized in 22% of ectopic pregnancies using transabdominal ultrasonography and in 38% using transvaginal ultrasonography (137). Other studies identified adnexal rings in 33% to 50% of ectopic pregnancies (135,153). The adnexal ring may not always be apparent because bleeding around the sac results in the appearance of a nonspecific adnexal mass.
Complex or solid adnexal masses are frequently associated with ectopic pregnancy; however, the mass may represent a corpus luteum, endometrioma, hydrosalpinx, ovarian neoplasm (e.g., dermoid cyst), or pedunculated fibroid (4,155–157). The presence of free cul-de-sac fluid is frequently associated with ectopic pregnancy and is no longer considered evidence of rupture. The presence of intra-abdominal free fluid should raise concern about tubal rupture (158,159).
Accurate interpretation of ultrasonography findings requires correlation with the hCG level (discriminatory zone) (146,151,154,160). All viable intrauterine pregnancies can be visualized by transabdominal ultrasonography for serum hCG levels higher than 6,500 mIU/mL; none can be seen at 6,000 mIU/mL. The inability to detect an intrauterine gestation with serum hCG levels higher than 6,500 mIU/mL indicates the presence of an abnormal (failed intrauterine or ectopic) pregnancy. Intrauterine sacs seen at hCG levels below the discriminatory zone are abnormal and represent either failed intrauterine pregnancies or the pseudogestational sacs of ectopic pregnancy. If there is no definite sign of an intrauterine gestation (the empty uterus sign) and the hCG level is below the discriminatory zone, the differential diagnosis includes the following considerations:
The discriminatory zone is lowered progressively with improvements in ultrasonography resolution. Discriminatory zones for transvaginal ultrasonography are reported at levels from 1,000 to 2,000 mIU/mL (146,151,154,160). Discriminatory zones vary according to the expertise of the examiner and capability of the equipment.
Although the discriminatory zone for intrauterine pregnancy is well established, there is no such zone for ectopic pregnancy. Levels of hCG do not correlate with the size of ectopic pregnancy. Regardless of how high the hCG level may be, nonvisualization does not exclude ectopic pregnancy. An ectopic pregnancy may be present anywhere in the abdominal cavity, making ultrasonographic visualization difficult.
Doppler Ultrasonography
A Doppler shift occurs whenever the source of an ultrasound beam is moving. The usual sources of Doppler-shifted frequencies are red blood cells. The presence of intravascular blood flow, flow direction, and flow velocity can be determined (161). Pulsed Doppler provides ultrasonographic control over which vessels are sampled. The vascular information is provided both by the shape of the time-velocity waveform (high- or low-resistance flow) and by its systolic, diastolic, and mean velocities (or Doppler frequency shifts) (162). Color flow Doppler ultrasonography analyzes very low amplitude signals from an entire ultrasound tomogram; the Doppler shift is then modulated into color. This information is used to gauge generalized tissue vascularity and to guide pulsed Doppler vascular sampling of specific vessels.
The waveform in the uterine arteries in the nongravid state and in the first trimester of pregnancy shows a high-resistance (little or no diastolic flow), low-velocity pattern. Conversely, a high-velocity, low-resistance signal is localized to the area of developing placentation (163–165). This pattern, seen near the endometrium, is associated with normal and abnormal intrauterine pregnancies and is termed peritrophoblastic flow. Whereas transvaginal ultrasonography requires a well-developed double decidual sac (or possibly cardiac activity) to localize an intrauterine gestation, the use of Doppler techniques allows detection of an intrauterine pregnancy at an earlier date. The combined use of Doppler and two-dimensional imaging allows the differentiation of pseudogestational sacs and true intrauterine gestational sacs and the differentiation of the empty uterus sign as either the presence of an intrauterine pregnancy (normal and abnormal) or absence of an intrauterine pregnancy (with an increased risk for ectopic pregnancy) (158,166).
A similar high-velocity, low-impedance flow characterizes ectopic pregnancies. The addition of Doppler to the ultrasonographic evaluation of suspected ectopic pregnancy improves diagnostic sensitivity for individual diagnoses: from 71% to 87% for ectopic pregnancy, from 24% to 59% for failed intrauterine pregnancy, and from 90% to 99% for normal intrauterine pregnancy (154,158,166). Transvaginal color Doppler ultrasonography did not increase overall detection rates (167). Magnetic resonance imaging (MRI) was studied for its possible uses in the diagnosis of ectopic pregnancy. The role of MRI in the detection of ectopic pregnancy is inconclusive. It was 96% accurate in detecting fresh hematoma associated with ectopic pregnancy. Further studies need to be done to assess its predictive value (168).
Dilation and Curettage
Uterine curettage is performed when the pregnancy is confirmed to be nonviable and the location of the pregnancy cannot be determined by ultrasonography. The decision to evacuate the uterus in the presence of a positive pregnancy test must be made with caution to avoid the unintentional disruption of a viable intrauterine pregnancy. Although suction curettage traditionally was performed in the operating room, it can be accomplished under local anesthesia on an outpatient basis. Endometrial sampling methods (e.g., a Novak curettage or Pipelle endometrial sampling device) are accurate in diagnosing abnormal uterine bleeding, but their reliability for intrauterine pregnancy evacuation was not studied. These devices might miss intrauterine villi and falsely suggest the diagnosis of ectopic pregnancy.
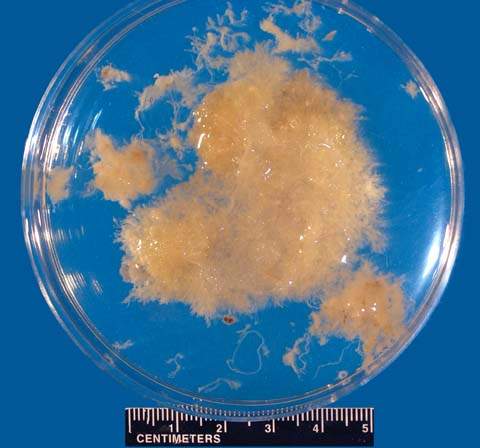
It is essential to confirm the presence of trophoblastic tissue as rapidly as possible so that therapy may be instituted. After tissue is obtained by curettage, it can be added to saline, in which it will float (Fig. 20.4). Decidual tissue does not float. Chorionic villi are usually identified by their characteristic lacy frond appearance. The sensitivity and specificity of this technique are 95% when the tissue is examined with the aid of a dissecting microscope. Because flotation of curettage sample tissue is not 100% accurate in differentiating an intrauterine from extrauterine gestation, histologic confirmation or serial β-hCG level measurement is required. The presence of chorionic villi may be assessed rapidly with frozen section analysis, which avoids the waiting period of at least 48 hours for permanent histologic evaluation. Immunocytochemical staining techniques are used to identify intermediate trophoblasts that are not normally identified by light microscopy (169).
Figure 20.4 When floated in saline, chorionic villi are often readily distinguishable as lacy fronds of tissue. (From Stovall TG, Ling FW. Extrauterine pregnancy: clinical diagnosis and management. New York: McGraw-Hill, 1993:186, with permission.)