DEFINITION OF THE COMPLAINT
Cough is one of the most common presenting complaints to pediatricians. Importantly, a cough is not a disease by itself, but rather a manifestation of an underlying pathology. A cough is a protective action, and can be initiated both voluntarily and via stimulation of cough receptors located throughout the respiratory tract (ear, sinuses, upper and lower airway to the level of the terminal bronchioles, pleura, pericardium, and diaphragm). A cough may serve to remove irritating substances, excessive/abnormal secretions, or may be secondary to intrinsic/extrinsic airway compression.
A cough is divided into four distinct phases: inspiratory, compressive, expiratory, and relaxation. These phases are characterized by deep inspiration, closure of the glottis, contraction of expiratory muscles with glottic opening, and relaxation of intercostal and abdominal muscles. Thus, one can see how selective patients with laryngeal or neuromuscular diseases may have ineffective coughs.
Classification should initially involve differentiating an acute from a chronic cough. A chronic cough is one which lasts longer than 3 weeks. Furthermore, the clinical description of the cough can often be helpful in suggesting an etiology: staccato (pertussis, chlamydia), barking (croup), grunting (asthma), or honking (psychogenic). Timing of the cough, relationship to daily activities, and age of the patient are important factors in further defining the etiology (Table 4-1).
COMPLAINT BY CAUSE AND FREQUENCY
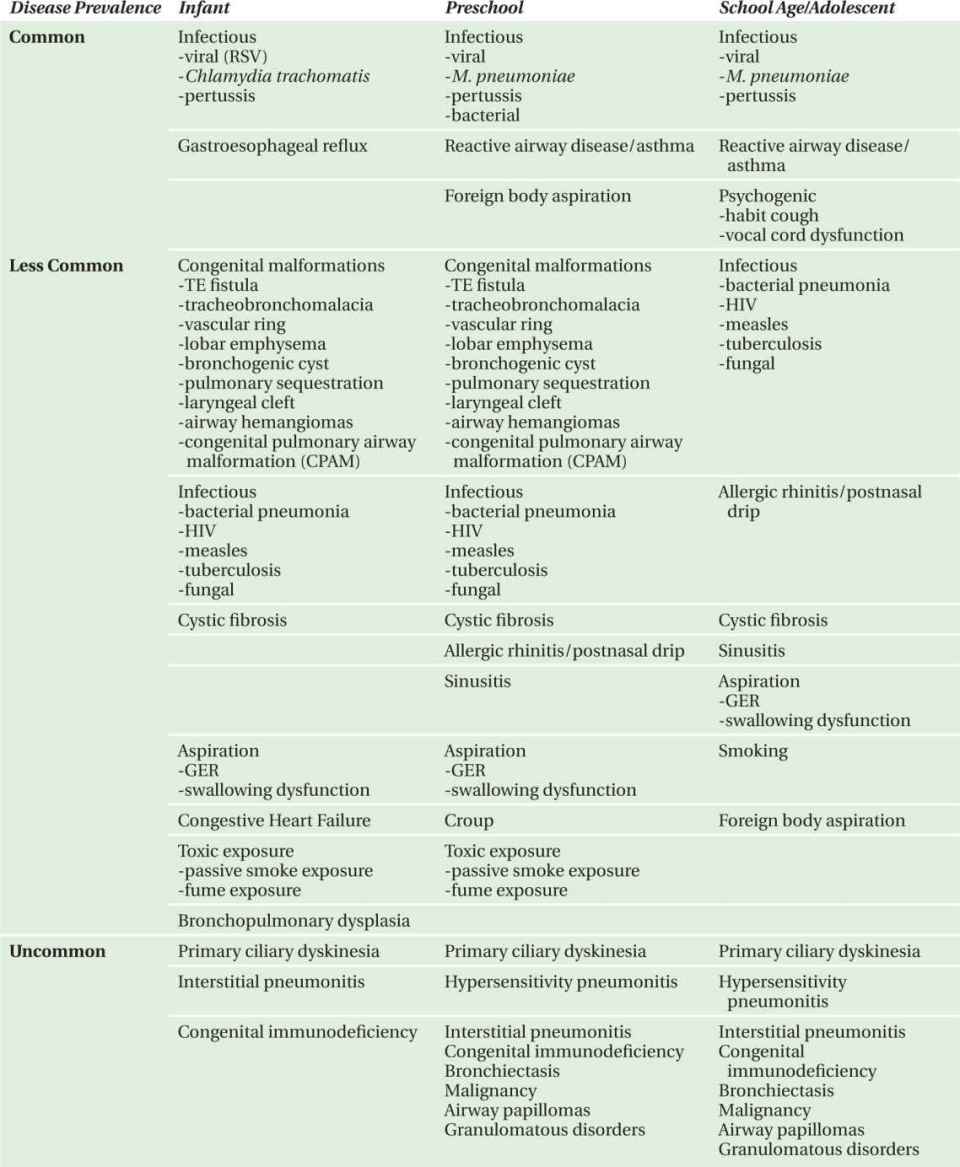
Overall, some of the most common causes of chronic cough include viral upper respiratory tract infections and asthma (Table 4-2). Beyond these etiologies, age is very important in creating a differential diagnosis for the patient with a chronic cough (Table 4-3). Causes of cough may also be divided by diagnostic category including infectious, allergic/inflammatory, congenital malformations, irritants, aspiration, psychogenic, and other categories (Table 4-4).
TABLE 4-2. Most common causes of cough in children.
CLARIFYING QUESTIONS
In most cases of a child who presents with a cough, the diagnosis is obtained with a thorough history and physical examination. The following questions may help to define the diagnosis:
• Did the cough begin with an upper respiratory tract infection?
—The most common cause for a cough is a viral upper respiratory tract infection. This can occur with or without reactive airways disease/asthma. Without other significant generalized signs of illness or respiratory distress, often no significant initial evaluation or therapy is necessary. Many young children will have frequent (6–8) viral infections per year accompanied by a cough, giving the appearance of chronic cough.
• Are there systemic signs or symptoms that may suggest a particular etiology?
—Fevers, sinus tenderness, and headaches can be present with sinusitis. Weight loss and night sweats may indicate tuberculosis or malignancy. Dysphagia suggests an esophageal foreign body, whereas dysphonia indicates laryngeal or glottic pathology.
• In young infants, is there any history of conjunctivitis in association with a cough and tachypnea?
—Conjunctivitis and pneumonitis in a young infant may suggest infection due to Chlamydia trachomatis.
• Are there environmental stimuli that may irritate the airway?
—Passive smoke in infants and young children and active smoking in adolescents can trigger a chronic cough. Solvent fumes as well as recreational drug use can exacerbate a chronic cough.
• How is the cough related to time of day and to daily activities?
—A cough which is most prominent during or after eating is suggestive of aspiration or gastroesophageal reflux. If exposure to cold air and exercise precipitates the cough, reactive airway disease should be considered. Seasonal coughing suggests an allergic component. Similarly, a nighttime cough may indicate postnasal drip secondary to either allergies or sinusitis.
• Does the cough resolve with sleep?
—Coughs which disappear when the patient is asleep or appear only when an adult is present may suggest a psychogenic cough.
• Is there a history of a choking episode?
—Often, a significant choking episode occurs at the time of foreign body ingestion. For this reason, a thorough history is essential. Foreign body aspiration is most common in toddlers; however, older siblings can often place inappropriate objects in the mouth of infants. If foreign body aspiration is suspected, one should obtain either lateral decubitus or inspiratory and expiratory chest roentgenograms.
• Is there a history of recurrent pneumonias or other infections?
—Recurrent infections should cause one to consider immune dysfunction such as HIV and congenital immunodeficiencies. Recurrent pneumonias associated with sinusitis, multiple otitis medias, bronchiectasis, and situs inversus, suggest primary ciliary dyskinesia.
• How is the patient’s growth?
—Failure to thrive, steatorrhea, and recurrent pneumonias can occur with cystic fibrosis. Other features suggestive of cystic fibrosis are nasal polyps, recurrent sinusitis, and rectal prolapse.
• Is there any history of hemoptysis?
—Hemoptysis can be seen with a viral or bacterial pneumonia. However, it is also present in many other conditions, including fungal disease, autoimmune diseases, granulomatous disorders, cystic fibrosis, congenital heart disease, tuberculosis, and pulmonary hemosiderosis.
GENERAL DIAGNOSTIC APPROACH
Commonly, cough is self-limited and rarely requires intervention. However, chronic cough (i.e., cough lasting longer than 3 weeks) suggests an underlying disease process requiring close investigation. A detailed history and physical examination will often lead to a diagnosis. However, adjunctive diagnostic tests, such as chest radiography and spirometry, may further aide this process. As discussed above, the patient’s history will elucidate important information, such as age, circumstances present at onset of cough, nature of the cough, timing, triggers, and response to previous therapies. Medical, family, and medication histories will also provide important information regarding associated signs and symptoms. Physical examination will further validate a suspected diagnosis. Specifically, examination should pay particular attention to the presence and absence of the following: failure to thrive, signs of increased work of breathing (tachypnea, retractions, accessory muscle use, chest wall appearance, and airway sounds), allergic signs (shiners, boggy nasal turbinates, nasal polyps, halitosis, and pharyngeal cobblestoning), abnormal heart sounds, hepato- and/or splenomegaly, digital clubbing, cyanosis, and hypotonia. Chest radiography rarely provides a definitive diagnosis (except possibly in the case of foreign body aspiration) but will provide important information if subsequent diagnostic testing is required. Lastly, pulmonary function testing (when available), such as spirometry and lung volumes, will distinguish obstructive from restrictive processes. The differential diagnosis of chronic cough is broad, but with the appropriate approach, can be successfully diagnosed and treated.
SUGGESTED READINGS
1. Pasterkamp, H. The history and physical examination. In: Chernick V, Boat TF, eds. Kendig’s Disorders of the Respiratory Tract in Children. Philadelphia, PA: W.B. Saunders Company; 1998:98-101.
2. Durbin WA. Cough. In: Hoekelman RA, Friedman SB, Nelson NM, Seidel HM, Weitzman ML, eds. Primary Pediatric Care. St. Louis, MO: Mosby; 1997:895-897.
3. Tunnessen WW. Cough. In: Tunnessen WW, ed. Signs and Symptoms in Pediatrics. Philadelphia, PA: Lippincott Williams & Wilkins; 1999:375-382.
4. Bachur R, Cough. In: Fleisher GR, Ludwig S, eds. Text-book of Pediatric Emergency Medicine. Philadelphia, PA: Lippincott Williams & Wilkins; 2000:183-186.
5. Morgan WJ, Taussig LM. The child with persistent cough. Pediar Rev. 1987;8:249-253.
6. Kamie RK. Chronic cough in children. Pediatr Clin N Am. 1991;38:593-605.
7. Chang AB. Cough. Pediatr Clin N Am. 2009;56:19-31, ix.
8. Goldsobel AB, Chipps BE. Cough in the pediatric population. J Pediatr. 2010;156:352-358.
The following cases represent less common causes of cough in childhood.
CASE 4-1
Sixteen-Year-Old Girl
HISTORY OF PRESENT ILLNESS
The patient is a 16-year-old black female with a 2-week history of a dry cough. She described shortness of breath, which worsened while lying down. She had been sleeping on three pillows since her cough began. Her cough and shortness of breath were waking her at night and symptoms were somewhat relieved by sitting up. She had chest tightness with deep inspiration as well as pain when lying on her left side. She had intermittent fevers (38.0°C) during the last few weeks as well as drenching night sweats.
She was initially treated with albuterol for her cough without significant relief. One week into her illness she was seen by an allergist who noted decreased breath sounds on the left side. A chest roentgenogram was obtained and the patient admitted for further evaluation.
Review of systems revealed decreased appetite with early satiety. She had some weight loss during the last 12 months. She denied headaches, sore throat, or changes in her voice. She reported an episode of diffuse pruritis 2 months prior to admission.
MEDICAL HISTORY
The patient’s history was remarkable for no prior significant illness and no hospitalizations. She had a fibroadenoma excised from her right breast 3 months prior to this admission. Her family history was significant for a maternal grandmother with uterine cancer, an aunt with breast cancer, and a maternal great-grandmother with thyroid cancer.
PHYSICAL EXAMINATION
T 38.1°C; P 127 bpm; RR 20/min; BP 139/73 mmHg
Weight 50th percentile; Height 95th percentile
Initial examination revealed an alert young woman, sitting forward, with noticeable shortness of breath and cough. Physical examination was remarkable for decreased breath sounds on the left, most significantly at the base. She had two small subcutaneous nodules on her left superior chest wall. The remainder of her physical examination was normal.
DIAGNOSTIC STUDIES
Laboratory analysis revealed a peripheral blood count with 21 200 WBC/mm3 with 94% segmented neutrophils, 3% lymphocytes, and 3% monocytes. Hemoglobin was 8.4 g/dL with an MCV of 63 fL. Platelets were 386 000/mm3. Electrolytes, blood urea nitrogen, creatinine, and liver function tests were normal. Erythrocyte sedimentation rate (ESR) was elevated at 60 mm/h.
COURSE OF ILLNESS
A chest computed tomography (CT) scan was obtained to further assess abnormalities on chest radiograph (Figure 4-1). The patient was admitted to the intensive care unit with concerns of impending respiratory failure.

FIGURE 4-1. Chest computed tomogram.
DISCUSSION CASE 4-1
DIFFERENTIAL DIAGNOSIS
The most common causes of cough in adolescents include infections and asthma. While viral infections are certainly the most common, other agents may include Mycoplasma pneumoniae, Bordetella pertussis, and bacterial infections. Less common infectious causes include HIV and related infections, measles, tuberculosis, and fungal infections. Patients with asthma would be most likely to have had a prior history of mild wheezing and possibly atopy. Other diagnoses that should be considered in an adolescent with a chronic cough include but are not limited to allergic rhinitis, sinusitis, gastroesophageal reflux, and smoking. Rare causes would include primary ciliary dyskinesia, interstitial pneumonitis, granulomatous disorders, and malignancies.
The concerning features of this case involve the patient’s significant shortness of breath and orthopnea. These are not common complaints with simple viral infections or asthma exacerbations. Her persistent fevers and night sweats suggest a systemic disorder, such as tuberculosis or oncologic disease, and definitely warrant further investigation.
DIAGNOSIS
A chest roentgenogram revealed a large anterior mediastinal mass with rightward tracheal deviation. Chest CT revealed a large, bulky infiltrating anterior mediastinal mass that extended from above the level of the clavicle to near the level of the diaphragm (Figure 4-1).
It also extended posteriorly to involve the middle mediastinum. There was also bilateral axillary lymphadenopathy and a left pleural effusion. Chest tube placement yielded 1200 cc of pleural fluid with 3900 red blood cells/mm3, 772 white blood cells/mm3 (22% segmented neutrophils and 78% lymphocytes), pH 7.44, glucose 100 mg/dL, LDH 386 U/L, and amylase <30 U/L. A lymph node biopsy performed in the operating room revealed nodular sclerosing Hodgkin lymphoma. Bone marrow biopsy did not reveal hematologic involvement. With a negative abdominal CT scan, her lymphoma was considered Stage IIB and she commenced a course of chemotherapy.
INCIDENCE AND EPIDEMIOLOGY
Hodgkin disease is a malignancy of mature B cells. It has a bimodal age distribution in industrialized countries, with an early peak in 15-35 years old and a second peak after 50 years of age. There seem to be three separate forms of the disease: childhood (<14 years of age), young adults (15-34 years), and older adults (55-74 years). It is the most commonly diagnosed cancer for adolescents (15-19 years of age) and is rare among children less than 5 years of age. The incidence is slightly higher in males.
Hodgkin disease develops when a single transformed B cell is clonally expanded.
The malignant cells consist of Reed-Sternberg cells, lymphocytic and histiocytic cells. Interestingly, only 1% of the tumor consists of malignant cells. The greatest proportions of cells are inflammatory cells resulting from a significant cytokine release. Based on histology, there are four subtypes of Hodgkin disease: lymphocytic predominance, mixed cellularity, lymphocytic depletion, and nodular sclerosing. Nodular sclerosing is the most common type in adolescents, whereas mixed cellularity is more common in children with Hodgkin Lymphoma. Epstein-Barr virus (EBV) has been associated with some cases of Hodgkin disease with virus reactivation as a possible causative factor. This appears to be more common in younger children. Current 10-year survival rates are greater than 90% in patients diagnosed prior to the age of 45 years.
CLINICAL PRESENTATION
Often, a patient will present with firm but painless lymphadenopathy. Approximately 60% of patients may have mediastinal involvement as part of their initial presentation. It is rare to have primary disease present in subdiaphragmatic locations. Constitutional symptoms are common and include fatigue, weight loss, anorexia, fevers, and night sweats. Interestingly, as seen in this patient, pruritis is often a complaint of patients with Hodgkin disease.
Abnormal laboratory findings may include leukocytosis, lymphopenia, eosinophilia, monocytosis, anemia, and thrombocytopenia. In contrast to many adults, most children have normal lymphocyte counts at diagnosis. Autoimmune disorders that can accompany Hodgkindisease include nephrotic syndrome and autoimmune hemolytic anemia, neutropenia, and thrombocytopenia. Nonspecific markers of inflammation such as the erythrocyte sedimentation rate and ferritin levels may be elevated.
DIAGNOSTIC APPROACH
A large pleural effusion and anterior mediastinal mass are concerning findings in this adolescent patient. Initial evaluation included a chest roent-genogram as well as aspiration of the pleural fluid.
Chest roentgenogram. Discovery of a large pleural effusion requires further diagnostic studies. Initially, lateral decubitus chest roentgenograms may be used to determine whether the fluid is free-flowing or loculated. Loculated pleural fluid suggests infection while free-flowing pleural fluid may be seen in many conditions. As in this patient, mediastinal masses, hilar lymphadenopathy, and pleural effusions may be noted in cases of malignancy.
Ultrasound. Chest ultrasound can facilitate management decisions by rapidly determining whether a pleural effusion is loculated or free-flowing.
Chest CT with intravenous contrast. CT of the chest may also be useful to analyze a pleural effusion for loculations. Chest CT is very useful to view the lung parenchyma in depth, revealing subtle lung disease not apparent on chest roentgenogram. Furthermore, a chest CT is able to further delineate masses, including mediastinal lesions and lymphadenopathy that may have been suggested on chest roentgenogram. With Hodgkin disease specifically, the most common extranodal disease sites include the lung parenchyma, chest wall, pleura, and pericardium.
Aspiration of pleural fluid. Ultimately, with a significant pleural effusion the pleural fluid should be aspirated. This is important from a diagnostic, and in many cases, therapeutic, standpoint. Pleural fluid should be sent for pH, culture, gram stain, cell count with differential, glucose, protein, and LDH. These studies can help to divide pleural effusions into exudates and transudates (Table 4-5). Exudates are most common in parapneumonic effusions, neoplasms, and connective tissue disease, whereas transudates are more common in congestive heart failure, nephrotic syndrome, and cirrhosis. If the pleural fluid pH is greater than 7.4, it is unlikely to be an exudative process. Malignant pleural effusions, such as those from Hodgkin lymphoma, are exudative in nature and most often have quite reduced glucose levels. On occasions, malignant cells themselves are noted in the pleural fluid.
TABLE 4-5. Pleural fluid analysis.

Tissue biopsy. The key to diagnosis of Hodgkin disease is pathologic confirmation of disease. This can be obtained from mass lesions as well as affected lymph nodes.
CT of neck/chest/abdomen and pelvis. Once the diagnosis of Hodgkin disease is established, these studies are performed in addition to a chest CT to stage the degree of disease.
MRI. In selected cases, MRI may be helpful in further delineating lymph node involvement and thus with staging of the disease.
Bone marrow biopsy. Useful for staging.
18-Fluorodeoxyglucose positron emission tomography (FDG-PET). PET scans are used to supplement initial staging and to monitor response to therapy. FGD-PET seems to be more sensitive than gallium scans in the detection of Hodgkin lymphoma.
TREATMENT
Determination of staging is essential in choosing the course of therapy for Hodgkin disease. Stages are based on the Ann Arbor staging classification and include:
• Stage I—Involvement of a single lymph node region or of a single extralymphatic organ or site.
• Stage II—Involvement of two or more lymph node regions on the same side of the diaphragm or localized involvement of an extralymphatic organ or site and one of more lymph node regions on the same side of the diaphragm.
• Stage III—Involvement of lymph node regions on both sides of the diaphragm.
• Stage IV—Diffuse or disseminated involvement of one or more extralymphatic organs or tissues with or without associated lymph node involvement.
Patients are stratified into low, intermediate, and high-risk categories on the basis of distribution of disease, bulk, and presence of B symptoms. Standard therapy for Hodgkin disease includes risk-adapted chemotherapy and involved-field radiation. Patients with favorable presentations (localized nodal involvement with no constitutional symptoms and without bulky disease) have been treated with fewer cycles of chemotherapy and lower radiation doses. In contrast, those patients with unfavorable presentations (constitutional symptoms, bulky mediastinal, or peripheral lymphadenopathy, and advanced Stage IIIB/IV disease) receive much more intense protocols.
SUGGESTED READINGS
1. Hudson MM, Onciu M, Donaldson SS. Hodgkin lymphoma. In: Pizzo PA, Poplack DG, eds. Principles and Practice of Pediatric Oncology. 5th ed. Philadelphia: Lippincott Williams & Wilkins; 2006:695-721.
2. Montgomery M. Air and liquid in the pleural space. In: Chernick V, Boat TF, eds. Kendig’s Disorders of the Respiratory Tract in Children. 6th ed. Philadelphia: W.B. Saunders Company; 1998:389-411.
3. Punnet A, Tsang RW, Hodgson DC. Hodgkin lymphoma across the age spectrum: epidemiology, therapy, and late effects. Semin Radiat Oncol. 2010;20:30-44.
Seven-Week-Old Boy
HISTORY OF PRESENT ILLNESS
The patient is a 7-week-old boy who was in good health until 3 weeks prior to presentation. At that time, he developed rhinorrhea, congestion, and cough. He had no history of fever. He was evaluated by his pediatrician. A chest roentgenogram demonstrated a left lower lobe infiltrate. He was then started on a 10-day course of erythromycin for treatment of pneumonia. On completion of this antibiotic course, his mother felt that his respiratory status had improved to some extent, but his work of breathing was still increased from his baseline. Furthermore, his cough was persistent in nature. One week later, a repeat chest roentgenogram revealed persistence of the left lower lobe infiltrate and he was referred for further evaluation. His review of symptoms revealed good oral intake and normal urine output.
MEDICAL HISTORY
He was born at 41 weeks gestation with a birth weight of 3000 g. There were no pregnancy or birth-related complications. He had no history of cyanosis or feeding difficulties. He was feeding on formula, taking two ounces every 2 hours. He has two older siblings who are both healthy.
PHYSICAL EXAMINATION
T 37.3°C; P 153 bpm; RR 54/min; BP RUE 93/59 mmHg, LUE 87/62 mmHg, RLE 94/63 mmHg; SpO2 95% in room air
Weight 4.5 kg
Initial examination revealed a well-developed infant in moderate respiratory distress.
Physical examination was remarkable for nasal flaring, intercostal retractions, and intermittent grunting. He had good aeration and scattered rales at both lung bases. Cardiac examination revealed a normal S1 with a prominent P2. A II-III/VI systolic murmur was appreciated at the left sternal border. The liver edge was palpated 4 cm below the right costal margin. The remainder of the physical examination was normal.
DIAGNOSTIC STUDIES
Laboratory analysis revealed a peripheral blood count of 8400 white blood cells/mm3 with 35% segmented neutrophils, 60% lymphocytes, and 5% eosinophils. The hemoglobin was 11.4 g/dL and there were 203 000 platelets/mm3. Electrolytes, blood urea nitrogen, and creatinine were all within normal limits. Antigens of respiratory viruses were not detected by immunofluorescence of nasopharyngeal washings.
COURSE OF ILLNESS
An electrocardiogram was performed that suggested a diagnosis (Figure 4-2).
FIGURE 4-2. Left image: Two-dimensional echocardiogram. Right image: Superimposed color Doppler flows, demonstrating systolic blood flow from left to right through the ventricular septal defect (in red-orange; normal aortic outflow shown in blue). (From: Roger Breitbart, MD, Department of Cardiology, Children’s Hospital Boston, with permission.)
DISCUSSION CASE 4-2
DIFFERENTIAL DIAGNOSIS
The causes of persistent cough in infants are diverse, but the most common etiology are infections. Viral infections, including respiratory syncytial virus, parainfluenza, and influenza, are the most common. Normal children may develop up to eight viral respiratory infections per year, and if each infection lasts 7-14 days, recurrent infections can appear to cause a chronic cough. Bacterial infections may also cause prolonged cough. In infants with a history of conjunctivitis or maternal cervical infection, Chlamydia trachomatis should be considered. The staccato-like cough is the classic description associated with this infection. Bordetella pertussis can occur in infants and presents with a chronic cough, along with apneic pauses, gagging, cyanosis, and bradycardia. Most often, infants are unable to generate the force necessary for the classic “whoop.” Certainly, other bacterial pneumonias should be considered with the lobar infiltrate noted on chest roentgenogram in the case above.
There are several noninfectious causes of persistent cough in infants. Asthma should always be considered, especially if there is associated wheezing and clinical response to bronchodilators. One should consider gastroesophageal reflux as a possible etiology for cough in infancy. Other less common causes of cough in this age group would include congenital malformations, including trachoesophageal fistulas, tracheobronchomalacia, vascular rings, lobar emphysema, bronchogenic cyst, pulmonary sequestration, laryngeal cleft, and airway hemangiomas. Cystic fibrosis should be on the differential diagnosis, particularly if the patient has a history of meconium ileus, steatorrhea, failure to thrive, or a positive family history for this disease.
Congestive heart failure should always be considered, particularly if there are also feeding difficulties, poor growth, tachycardia, tachypnea, nasal flaring, intercostal retractions, grunting, murmur, and/or hepatomegaly. Etiologies in infancy may include volume overload (patent ductus arteriosus, truncus arteriosus, ventricular septal defect, common atrioventricular canal, total anomalous pulmonary venous return), myocardial dysfunction (myocarditis, anomalous left coronary artery), arrhythmias (supraventricular tachycardia), pressure overload (coarctation of the aorta, aortic stenosis), and secondary causes (hypertension, sepsis).
The features of this case that prompted additional evaluation were the presence of a heart murmur, a loud P2, bilateral rales, and hepatomegaly on the physical examination, as well as biventricular hypertrophy seen on the electrocardiogram. Additional suspicious features would have been the recognition of cardiomegaly and increased vascular markings on the chest roentgenogram.
DIAGNOSIS
Electrocardiogram revealed a ventricular rate of 150 bpm and biventricular hypertrophy. An echocardiogram revealed a large perimembranous ventricular septal defect (VSD) with left to right shunting. It also demonstrated moderately depressed biventricular function with a shortening fraction of 29%. The diagnosis is a perimembranous VSD.
INCIDENCE AND EPIDEMIOLOGY
Ventricular septal defects account for up to 40% of cardiac anomalies, making them the most common cardiac malformation seen in children. Studies have shown the incidence of VSD in newborns to be 5-50 per 1000 children with a slight female predominance. VSDs are the most common form of congenital heart disease associated with chromosomal disorders, although most patients with VSD do not have a chromosomal abnormality. Other types of genetic disorders that have recently been shown to be associated with VSD include single gene defects, such as mutations in TBX5 and GATA4. Single gene defects account for only 3% of patients with congenital heart disease. VSD has also been associated with environmental factors such as maternal infection or teratogens.
VSDs may be classified into four types: perimembranous, also known as membranous or infracristal (most common, 80%); outlet, also known as subpulmonary, supracristal, conal, infundibular, or doubly committed subarterial (5%-7%); inlet (5%-8%); and muscular (5%-20%). A diagram of various types of VSD is depicted in Figure 4-3. Muscular defects have the greatest likelihood to undergo spontaneous closure. Approximately 75%-80% of small VSDs will close spontaneously, most often by 2 years of age.

FIGURE 4-3. Anatomy of perimembranous ventricular septal defect. (From the Multimedia Library of Congenital Heart Disease, Children’s Hospital, Boston, MA, editor Robert Geggel, MD, www.childrenshospital.org/mml/cvp, with permission.)
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree