11.4 Congenital and perinatal infections
Introduction
Modes of acquisition of infection
Many organisms can cause infection in the fetus and newborn. Box 11.4.1 lists some of the more common or clinically significant.
Risk assessment
The risk of fetal damage can be estimated, based on:
• the likelihood of maternal exposure and infection
• the likelihood of transmission to the fetus
• the stage in gestation at which infection occurs – this influences the risk of vertical transmission and/or the fetal or perinatal consequences.
Congenital infections
• Some common viral infections (e.g. cytomegalovirus, parvovirus, rubella, varicella-zoster virus) can cause fetal infection with severe consequences if acquired during pregnancy by a non-immune woman.
• Ensuring that a woman has antenatal testing and is immunized with all recommended vaccines is an important part of care during pregnancy.
• Management of a pregnant woman exposed to relevant viral infections depends on a careful risk assessment, which includes knowledge of the woman’s immune status, history of the exposure, stage in the pregnancy, and clinical presentation in the woman.
Organisms associated with perinatal infection
Cytomegalovirus
Primary infection
• 50% of young women are seronegative (susceptible). In developing countries and lower socioeconomic groups, primary infection occurs at a younger age and fewer women are susceptible.
• 1% of women seroconvert during pregnancy.
• 30% of fetuses of women with primary infection are infected.
• The risk of congenital CMV infection after primary maternal CMV infection remains increased for up to 4 years after seroconversion, with the highest risk being in the first 2 years.
• Infection is transplacental; severe fetal damage is more likely early in gestation.
• 10% of infants infected during primary maternal infection are symptomatic at birth: of these, 90% have significant long-term handicap.
• 90% of infants infected during primary maternal infection are asymptomatic at birth: of these, 10% go on to develop deafness or intellectual handicap.
• The overall incidence of congenital infection due to primary maternal infection is 1 in 1000.
Reactivation
• 20–30% of seropositive women reactivate latent infection during pregnancy.
• 2–5% of their infants are infected in utero but significant CMV disease is rare; mild sequelae (unilateral deafness) occur infrequently (< 10%).
• The overall incidence of congenital infection is 1–2% – the majority are unaffected.
Clinical features
The clinical features of severe intrauterine CMV infection include:
• intrauterine growth restriction
• hepatosplenomegaly, hepatitis
• microcephaly, encephalitis, cerebral calcification and chorioretinitis
• SNHL in congenital CMV affects 50% of symptomatic infants and 10% of asymptomatic infants. It is the most common long-term consequence. It ranges from mild unilateral to profound bilateral hearing loss. In asymptomatic infants it may be underdiagnosed because the SNHL is detected too late to prove congenital infection
• cerebral palsy, intellectual disability, epilepsy and visual impairment.
Parvovirus B19
Parvovirus B19 can also cause:
• mild respiratory tract illness without rash
• chronic bone marrow failure in immunodeficient patients
• transient aplastic crisis in patients with haemolytic anaemia (e.g. sickle cell disease).
Fetal risks following maternal infection
• fetal loss in the first 20 weeks of pregnancy (15% compared with 5% in controls, i.e. 10% excess fetal loss)
• congenital abnormalities: < 1% (anecdotal reports only)
• hydrops fetalis: following maternal infections at 9–20 weeks’ gestation (incidence is approximately 3%); may result in:
Table 11.4.1 Risk assessment – parvovirus B19
| Any pregnant woman exposed to parvovirus (%) | Pregnant woman with proven recent infection (%) | |
|---|---|---|
| Excess fetal loss in first 20 weeks | 0.4–1 (1 in 100–250) | 5 (1 in 20) |
| Death from hydrops or its treatment | 0.05–0.1 (1 in 850–2000) | 0.6 (1 in 170) |
There are no specific congenital abnormalities associated with maternal parvovirus infection.
Rubella
Clinical features
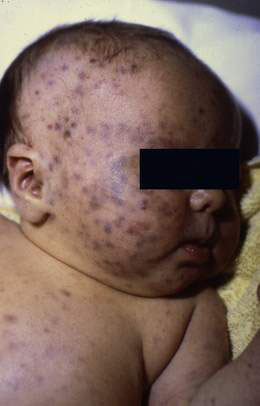
The risk of fetal infection and damage is greatest during the first 8 weeks of pregnancy and damage is rare after 16 weeks. Congenital rubella syndrome (Fig. 11.4.1) may include a number of clinical features, some of which may not present until adolescence or adulthood:
• intrauterine growth restriction
• neonatal purpuric rash and hepatosplenomegaly
• microcephaly and developmental delay
• cardiac: pulmonary artery hypoplasia, patent ductus arteriosus
• eye: cataract, retinopathy; microphthalmia
Diagnosis
Congenital rubella is confirmed by:
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree