Chapter 16 Common Medical and Surgical Conditions Complicating Pregnancy
The more common medical, infectious, and surgical disorders that may complicate pregnancy are covered in this chapter. The pharmacologic agents recommended for these disorders have been classified by the Food and Drug Administration (FDA) for fetal risk (see Box 7-1 on page 73). Up to date information on these drugs can be found at www.FDA.gov/ by selecting “Drugs” from the menu and searching for a specific agent.
 Endocrine Disorders
Endocrine Disorders
DIABETES MELLITUS
Incidence and Classification
GDM is defined as glucose intolerance with onset or first recognition during pregnancy. Pregnancy is associated with progressive insulin resistance. Human placental lactogen, progesterone, prolactin, cortisol, and tumor necrosis factor are associated with increased insulin resistance during pregnancy. Studies suggest that women who develop GDM have chronic insulin resistance and that GDM is a “stress test” for the development of diabetes in later life. Most obstetricians use White’s classification of diabetes during pregnancy. This classification is helpful is assessing disease severity and the likelihood of complications (Table 16-1).
TABLE 16-1 WHITE’S CLASSIFICATION OF DIABETES IN PREGNANCY
| Class | Description | Therapy |
|---|---|---|
| A1 | Gestational diabetes; glucose intolerance developing during pregnancy; fasting blood glucose and postprandial plasma glucose normal | Diet alone |
| A2 | Gestational diabetes with fasting plasma glucose >105 mg/dL; or 2-hr postprandial plasma glucose >120 mg/dL, or 1-hr postprandial plasma glucose >140 mg/dL | Diet and insulin |
| B | Overt diabetes developing after age 20 yr and duration < 10 yr | Diet and insulin |
| C | Overt diabetes developing between ages 10 and 19 yr or duration 10-19 yr | Diet and insulin |
| D | Overt diabetes developing before age 10 yr or duration 20 yr or more or background retinopathy | Diet and insulin |
| F | Overt diabetes at any age or duration with nephropathy | Diet and insulin |
| R | Overt diabetes at any age or duration with proliferative retinopathy | Diet and insulin |
| H | Overt diabetes at any age or duration with arteriosclerotic heart disease | Diet and insulin |
Complications
Maternal and fetal complications of diabetes are listed in Table 16-2. Diabetes often coexists with the metabolic syndrome. Most fetal and neonatal effects are attributed to the consequences of maternal hyperglycemia, or, in the more advanced classes, to maternal vascular disease. Glucose crosses the placenta easily by facilitated diffusion, causing fetal hyperglycemia, which stimulates pancreatic β cells and results in fetal hyperinsulinism. Fetal hyperglycemia during the period of embryogenesis is teratogenic. There is a direct correlation between birth defects in diabetic pregnancies and increasing glycosylated hemoglobin levels (HbA1C) in the first trimester. Fetal hyperglycemia and hyperinsulinemia cause fetal overgrowth and macrosomia, which predisposes to birth trauma, including shoulder dystocia and Erb’s’ palsy. Fetal demise is most likely due to acidosis, hypotension from osmotic dieresis, or hypoxia from increased metabolism coupled with inadequate placental oxygen transfer.
TABLE 16-2 MATERNAL AND FETAL COMPLICATIONS OF DIABETES MELLITUS
| Entity | Monitoring |
|---|---|
| MATERNAL COMPLICATIONS | |
| OBSTETRIC COMPLICATIONS | |
| Polyhydramnios | Close prenatal surveillance; blood glucose monitoring, ultrasonography |
| Preeclampsia | Evaluation for signs and symptoms |
| Infections, e.g., urinary tract infection and candidiasis | Urine culture, wet mount, appropriate therapy |
| Cesarean delivery | Blood glucose monitoring, insulin and dietary adjustment to prevent fetal overgrowth |
| Genital trauma | Ultrasonography to detect macrosomia, cesarean delivery for macrosomia |
| DIABETIC EMERGENCIES | |
| Hypoglycemia | Teach signs and symptoms; blood glucose monitoring; insulin and dietary adjustment; check for ketones, blood gases, and electrolytes if glucose > 300 mg/dL |
| Diabetic coma | |
| Ketoacidosis | |
| VASCULAR AND END-ORGAN INVOLVEMENT OR DETERIORATION (IN PATIENTS WITH PREGESTATIONAL DIABETES MELLITUS) | |
| Cardiac | Electrocardiogram first visit and as needed |
| Renal | Renal function studies, first visit and as needed |
| Ophthalmic | Funduscopic evaluation, first visit and as needed |
| Peripheral vascular | Check for ulcers, foot sores; noninvasive Doppler studies as needed |
| NEUROLOGIC | |
| Peripheral neuropathy | Neurologic and gastrointestinal consultations as needed |
| Gastrointestinal disturbance | |
| Long-Term Outcome | |
| Type 2 diabetes | Postpartum glucose testing, lifestyle changes (diet and exercise) |
| Metabolic syndrome | Lifestyle changes (diet and exercise) |
| Obesity | Lifestyle changes (diet and exercise) |
| Cardiovascular disease | Annual checkup by physician, lifestyle changes (diet and exercise) |
| Fetal and Neonatal Complications | |
| Maintenance of maternal euglycemia will decrease most of these complications. | |
| Macrosomia with traumatic delivery (shoulder dystocia, Erb’s palsy) | Ultrasonography for estimated fetal weight before delivery; consider cesarean delivery if estimated fetal weight > 4250-4500 g |
| DELAYED ORGAN MATURITY | |
| Pulmonary, hepatic, neurologic, pituitary-thyroid axis; with respiratory distress syndrome, hypocalcemia | Avoid delivery before 39 weeks in the absence of maternal or fetal indications unless amniocentesis indicates lung maturity. Maintain euglycemia intrapartum. |
| CONGENITAL DEFECTS | |
| Cardiovascular anomalies | Preconception counseling and glucose control, HbAlc in the first trimester |
| Neural tube defects | Maternal serum alpha-fetoprotein screening; fetal ultrasonography and fetal echocardiogram; amniocentesis and genetic counseling |
| Caudal regression syndrome | |
| Other defects, e.g., renal | |
| FETAL COMPROMISE | |
| Intrauterine growth restriction | Serial ultrasonography for fetal growth and estimated fetal weight, serial fetal surveillance with nonstress test, amniotic fluid index, and fetal Doppler. Avoid postdates pregnancy. |
| Intrauterine fetal death | |
| Abnormal fetal heart rate patterns | |
Diagnosis
An abnormal screening GCT is followed with a diagnostic 3-hour 100-g oral glucose tolerance test. This involves checking the fasting blood glucose after an overnight fast, drinking a 100-g glucose drink, and checking glucose levels hourly for 3 hours. If there are two or more abnormal values on the 3-hour GTT, the patient is diagnosed with GDM (Table 16-3). If the 1-hour screening (50-g oral glucose) plasma glucose exceeds 200 mg/dL, a glucose tolerance test is not required and may dangerously elevate blood glucose values.
TABLE 16-3 THREE-HOUR ORAL GLUCOSE TOLERANCE TEST
| Test | Maximal Normal Blood Glucose (mg/dL) |
|---|---|
| Fasting | 95 |
| 1 hr | 180 |
| 2 hr | 155 |
| 3 hr | 140 |
From Carpenter and Coustan.
Management
THERAPY.
Insulin use is the gold standard to maintain euglycemia in pregnancy. The peak action of lispro insulin is at 30 to 90 minutes, of regular insulin at 2 to 3 hours, and of NPH insulin at 6 to 10 hours. A combination of rapid-acting or short-acting (lispro or regular) and intermediate-acting (NPH) insulin is usually given in split morning and evening doses or more frequently to achieve euglycemia. A method for calculating insulin dosage is shown in Box 16-1.
THYROID DISEASES
Normal Thyroid Physiology during Pregnancy
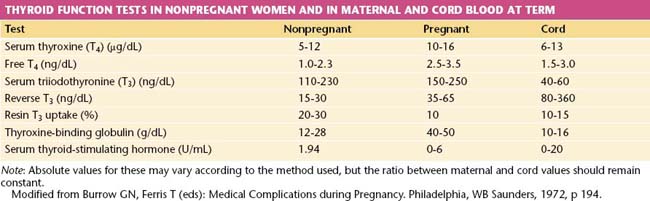
THYROID FUNCTION TESTS.
The free thyroxine (free T4) concentration is the only accurate method of estimating thyroid function that compensates for changes in thyroxine-binding globulin (TBG) capacity because serum levels of bound triiodothyronine (total T3) and total T4 are increased during pregnancy. Values of thyroid function tests during pregnancy are shown in Table 16-4.
 Heart Disease
Heart Disease
MANAGEMENT OF CARDIAC DISEASE DURING PREGNANCY
The New York Heart Association’s functional classification of heart disease is of value in assessing the risk for pregnancy in a patient with acquired cardiac disease and in determining the optimal management during pregnancy, labor, and delivery (Table 16-5). In general, the maternal and fetal risks for patients with class I and II disease are small, whereas risks are greatly increased with class III and IV disease or if there is cyanosis. However, the type of defect is important as well. Mitral stenosis and aortic stenosis carry a higher risk for decompensation than do regurgitant lesions. Other patients at high risk include those with significant pulmonary hypertension, a left ventricular ejection fraction less than 40%, Marfan syndrome, a mechanical valve, or a previous history of a cardiac event or arrhythmia.
TABLE 16-5 NEW YORK HEART ASSOCIATION’S FUNCTIONAL CLASSIFICATION OF HEART DISEASE
| Class I | No signs or symptoms of cardiac decompensation |
| Class II | No symptoms at rest, but minor limitation of physical activity |
| Class III | No symptoms at rest, but marked limitation of physical activity |
| Class IV | Symptoms present at rest, discomfort increased with any kind of physical activity |
 Autoimmune Disease in Pregnancy
Autoimmune Disease in Pregnancy
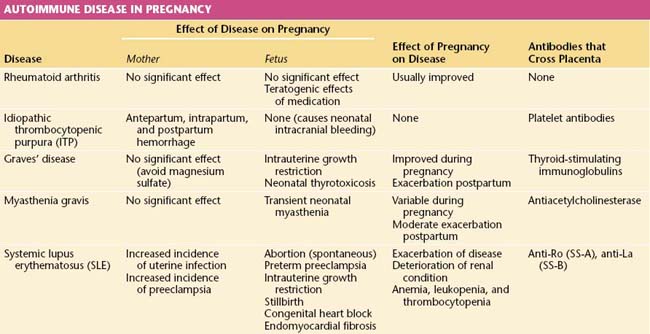
An autoimmune disease is one in which antibodies are developed against the host’s own tissues. A summary of the interactions of primary immunologic disorders and pregnancy is shown in Table 16-6.
SYSTEMIC LUPUS ERYTHEMATOSUS
Lupus occurs mainly in women. Associated antibodies include antinuclear, anti-RNP and anti-SM antibodies; anti-dsDNA is associated with nephritis and lupus activity; anti-Ro (SS-A) and anti-La (SS-B) are present in Sjögren’s syndrome and neonatal lupus with heart block; while antihistone antibody is common in drug-induced lupus. The diagnosis of systemic lupus is made if 4 or more of the 11 revised criteria of the American Rheumatism Association are present, serially or simultaneously (Table 16-7).
TABLE 16-7 AMERICAN RHEUMATISM ASSOCIATION 1997 REVISED CRITERIA FOR SYSTEMIC LUPUS ERYTHEMATOSUS
| Criteria∗ | Comments |
|---|---|
| Malar rash | Malar erythema |
| Discoid rash | Erythematous patches, scaling, follicular plugging |
| Photosensitivity | |
| Oral ulcers | Usually painless |
| Arthritis | Nonerosive involving two or more peripheral joints |
| Serositis | Pleuritis or pericarditis |
| Renal disorder | Proteinuria > 0.5 g/day or > 3+ dipstick, or cellular casts |
| Neurologic disorders | Seizures or psychosis without other cause |
| Hematologic disorders | Hemolytic anemia, leukopenia, lymphopenia, or thrombocytopenia |
| Immunologic disorders | Anti-dsDNA or anti-Sm antibodies, or false-positive VDRL, immunoglobulin M or G anticardiolipin antibodies, or lupus anticoagulant |
| Antinuclear antibodies | Abnormal titer of antinuclear antibodies |
VDRL, Venereal Disease Research Laboratory.
∗ If four criteria are present at any time during course of disease, systemic lupus can be diagnosed with 98% specificity and 97% sensitivity.
From Hochberg MC: Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 40(9):1725, 1997. Copyright 1997 American College of Rheumatology. Reprinted with permission of John Wiley & Sons, Inc.

 Renal Disorders
Renal Disorders

