24 Atopic and Rheumatic Disorders
 Pathophysiology and Defense Mechanisms
Pathophysiology and Defense Mechanisms
Atopic or Allergic Disorders
The development of an atopic disorder or allergic response involves a susceptible individual who is both exposed to an offending antigen and has a predisposition to selective synthesis of IgE when in contact with common environmental antigens. If these conditions are in place and contact with an offending antigen occurs, the following biochemical chain of cascading events unfolds:
• There is a brisk proliferation of T-helper type 2 (Th2) cells that secrete cytokines: interleukin (IL)-3, IL-4, IL-5, IL-9, and IL-13.
• Cytokines are involved in IgE synthesis and activation of eosinophils.
• IgE binds to receptors on mast cells, basophils, and Langerhans cells.
• Chemical mediators that cause biochemical reactions and allergic-related injury to target organs (skin and respiratory tract) are released. Examples of chemical mediators include but are not limited to:
Rheumatic Disorders
Systemic Lupus Erythematosus
Children with SLE exhibit a marked increase in the production of autoantibodies that attack the body’s deoxyribonucleic acid (DNA). This leads to immune complex formation and tissue damage from either direct bonding in tissues, immune complex deposition, or a combination of both (Klein-Gitelman, 2010). Various immune phenomena are associated with SLE, including altered immunologic reactions in the T- and B-lymphocyte function. There is a loss of T-lymphocyte control and hyperactivity of B lymphocytes resulting in nonspecific and specific antibody and autoantibody production. There is a strong link between a faulty immune mechanism and SLE because this disease is characterized by inflammatory damage to target organs brought on by autoantibodies attacking self-antigens. The exact etiology of SLE is unknown, but many factors including genetics, hormones, and environment are linked to the immune dysregulation that characterizes this rheumatic disease. Environmental factors that are thought to play a role in its pathogenesis are oral contraceptive use, pregnancy, microbials (viral agents mostly), temperate climates, exposure to ultraviolet light, and certain drugs (e.g., hydralazine and procainamide).
 Assessment
Assessment
History
Key factors to consider in the history of a child who has an atopic disorder include the following:
• A family history or personal history of allergies, asthma, AD (eczema), or AR is frequently found; similar allergic diseases tend to be found in families.
• Pruritus is a significant finding in AD and AR.
• The rash of AD is characteristically found in certain locations of the body.
• Coughing or shortness of breath with exercise or exertion and nighttime coughing and wheezing are characteristic of asthma.
• Signs and symptoms of AR and asthma may be associated with certain allergens or key triggering agents and may be seasonal.
Key factors to consider in the history of a child who has a rheumatic disease include the following:
• History of a characteristic rash or joint involvement (arthritis), or both (common findings). May have symptoms of serositis, enthesitis, myositis, and vasculitis, which are other inflammatory markers
• Other systemic manifestations of disease such as fever and hematologic, renal, and/or pulmonary abnormalities
Physical Examination
A detailed physical examination of the cardiovascular, respiratory, integumentary, and musculoskeletal systems may reveal characteristic signs (see Chapters 30, 31, 36, and 37).
 Management
Management
General Measures
The following general measures should be part of the management of atopic and rheumatoid disorders:
• Encourage self-care and learning about one’s disease.
• Address issues of burdens associated with living with a chronic disease, such as:
• Nutrition and the avoidance of obesity, if activity is limited, or foods if they are triggers
Medications
 Immunomodulators—to inhibit the inflammatory response. They include topical preparations and subcutaneous agents.
Immunomodulators—to inhibit the inflammatory response. They include topical preparations and subcutaneous agents.• Omalizumab is a recombinant DNA-derived, humanized IgG monoclonal antibody that binds to human IgE on the surface of mast cells and basophils. This anti-IgE monoclonal antibody is used as a second-line treatment for children older than 12 years who have moderate to severe allergy-related asthma and react to perennial allergens. It is used when symptoms are not controlled by inhaled corticosteroids (ICSs).
 Analgesics (salicylates or nonsteroidal antiinflammatory drugs [NSAIDs])—to relieve arthritis or joint pain; to relieve pain in general
Analgesics (salicylates or nonsteroidal antiinflammatory drugs [NSAIDs])—to relieve arthritis or joint pain; to relieve pain in generalParent and Patient Education
Patients and parents need to be instructed about the following:
• Signs and symptoms necessitating immediate reevaluation.
• Medications—clear instructions are needed on how to administer, how much and when to give, monitoring side effects, and how long medication should be taken. A written plan is highly recommended based on either symptoms or, in the example of asthma, peak expiratory flow rate (PEFR).
• Correct administration of inhaled medications—for example, when two puffs or sprays are ordered, the child should activate one puff or spray and then inhale, followed in 1 to 2 minutes by a second puff or spray and second inhalation. A parent or child may think incorrectly that being told to take two puffs or two sprays means to activate two puffs or sprays and then inhale.
• Any other measures relevant to the treatment plan (e.g., bathing instructions, monitoring peak flow rate, avoiding allergens, and environmental control)
• Parent support groups and professional organizations and resource groups
 Specific Immunologic Problems of Children: Common Atopic Disorders
Specific Immunologic Problems of Children: Common Atopic Disorders
Asthma
Description
• Immunohistopathologic responses that produce:
 Inflammatory infiltration by eosinophils, lymphocytes (Th2-like cells), and neutrophils (especially in fatal asthma)
Inflammatory infiltration by eosinophils, lymphocytes (Th2-like cells), and neutrophils (especially in fatal asthma)• Airway inflammation contributes to airflow limitations, including:
• Airflow obstruction is often reversible, either spontaneously or with treatment.
• Persistent inflammation can result in airway wall remodeling and irreversible changes.
• Airway inflammation also triggers hyperresponsiveness (to any of a variety of stimuli, such as physical, chemical, or pharmacologic agents; allergens; exercise; and cold air) and is a factor in disease chronicity. Inflammation causes bronchospasms with resultant characteristic symptomatology of wheezing, breathlessness, chest tightness, or cough typically worse at night or after exercise (Sharma and Gupta, 2010).
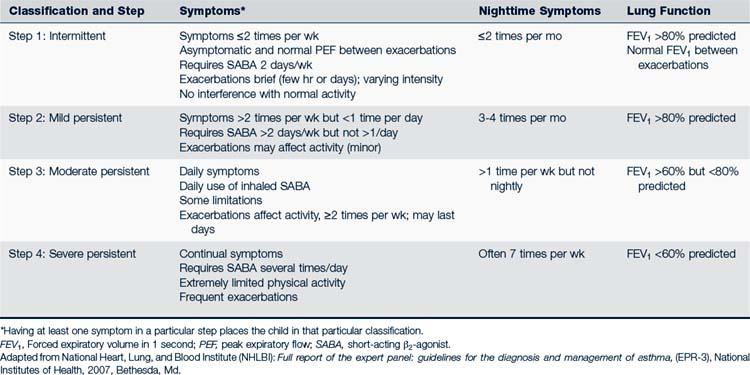
Asthma in children is classified as intermittent, mild persistent, moderate persistent, or severe persistent depending on symptoms, recurrences, need for specific medications, and pulmonary function measurements (Table 24-1). Children classified at any level of asthma can have episodes involving mild, moderate, or severe exacerbations. Exacerbations involve progressive worsening of shortness of breath, cough, wheezing, chest tightness, or any combination of these symptoms. The degree of airway hyperresponsiveness is usually related to the severity of asthma. Children younger than 5 years of age experience greater airway hyperresponsiveness than older children. Airway remodeling can result from chronic inflammation caused by asthma. Irreversible structural changes take place and result in decreased pulmonary function (Lasley, 2006). A child’s classification can change over time.
Many children experience early- and late-phase responses to their asthma episode. The early asthmatic response (EAR) phase is characterized by activation of mast cells and their mediators, with bronchoconstriction being the key feature. EAR starts within 15 to 30 minutes of mast cell activation and resolves within approximately 1 hour if the individual is removed from the offending allergen. The late-phase asthmatic response is a prolonged inflammatory state that usually follows the EAR within 4 to 12 hours after exposure to the allergen, is often associated with airway hyperresponsiveness more severe than the EAR presentation, and can last from hours to several weeks (Liu et al, 2004).
Epidemiology
• Atopic individual response to allergens—inhaled, topical, ingested
• Exposure to known irritants (paint fumes, smoke, air pollutants) and occupational chemicals
• Exposure to tobacco smoke (for infants, especially smoking by mother)
• Environmental changes—rapid changes in barometric pressure, temperature, especially cold air
• Psychological factors or emotional stresses (e.g., crying, laughter, anxiety attack, or panic disorder)
• Drugs (e.g., acetaminophen, aspirin, beta-blockers)
Clinical Findings
History
In order to assess the degree of asthma severity and impairment of function, a standardized instrument can be used. Tests for this purpose are available in the guidelines summary (NHLBI, 2007, p 17). Other instruments such as the Asthma Control Questionnaire, Asthma Therapy Assessment Questionnaire, Asthma Control Test, and the Asthma Control Score are available via the Internet. The advantages of a standardized questionnaire are that it allows the health care provider to assess changes in the patient’s asthma, then alter the management plan as needed. The history of a patient being seen for asthma can include the following:
• Family history of asthma or other related allergic disorders (e.g., eczema or AR)
• Conditions associated with asthma (e.g., chronic sinusitis, nasal polyposis, gastroesophageal reflux, and chronic otitis media)
• Complaints of chest tightness or dyspnea
• Cough, particularly at night and in the early morning
• Cough or shortness of breath with exercise or exertion
• Seasonal, continuous, or episodic pattern of symptoms
• Episodes of recurrent “bronchitis” or pneumonia
• Precipitation of symptoms by known aggravating factors (upper respiratory infections [URIs], acetaminophen, aspirin)
Physical Examination
The following may be seen on physical examination:
• Wheezing (may be absent if severe obstruction) or coughing
• Prolonged expiratory phase, high-pitched rhonchi
• Signs of respiratory distress, including tachypnea, retractions, nasal flaring, use of accessory muscles, increasing restlessness, apprehension, agitation, drowsiness to coma
• Tachycardia, hypertension or hypotension, pulsus paradoxus
• Cyanosis of lips and nailbeds if underlying hypoxemia
• Other possible associated findings include sinusitis, atopic dermatitis, and rhinitis.
Diagnostic Studies
• Oxygen saturation by pulse oximetry to assess severity of acute exacerbation. This should be a routine part of every assessment on a patient with asthma. Pulse oximetry measures the oxygen saturation (Sao2) of hemoglobin—the percent of total hemoglobin that is oxygenated—as follows:
• A complete blood count (CBC) if secondary infection or anemia is suspected (also check for elevated numbers of eosinophils)
• Chest radiograph for the first episode, and then only if secondary respiratory infection or other pulmonary disorders are suspected or if child is less than 1 year old with persistent wheezing
• Sinusitis should be considered as a cause of an asthma exacerbation. However, imaging of the sinus is not routinely needed to make the diagnosis of sinusitis. (See Chapter 31 for a discussion on sinusitis and the criteria needed for ordering computed tomography [CT] scans, which are more sensitive and specific than sinus radiographs.)
• Allergic workup, including skin testing, IgE, or ImmunoCAP (refer child to pediatric allergist)
• Sweat test if cystic fibrosis is a possibility
 Formal spirometry testing is the gold standard in children older than 4 years for diagnosing asthma.
Formal spirometry testing is the gold standard in children older than 4 years for diagnosing asthma. Start with PEF assessment in children 4 to 5 years or older to monitor the effectiveness of β-agonist treatment (measurements before and after treatments) and assess the severity of airflow obstruction.
Start with PEF assessment in children 4 to 5 years or older to monitor the effectiveness of β-agonist treatment (measurements before and after treatments) and assess the severity of airflow obstruction.Pulmonary monitoring and typical findings include the following:
• PEF can be used in some children as young as 4 to 5 years. Noteworthy is the fact that values are instrument specific. Use child’s personal best value as a guideline to help detect possible changes in airway obstruction; can use predicted range for height and age if personal best rate is not available (Table 24-2).
• Interpretation of PEF reading (Box 24-1 describes use of peak flowmeter and interpretation of results)—if PEF is in the:
• Chest radiograph findings: Typically normal and may show hyperinflation of the lungs with flattening of the diaphragm and peribronchial thickening on radiograph with or without atelectasis (Liu et al, 2007)
TABLE 24-2 Predicted Average Peak Expiratory Flow for Normal Children and Adolescents
| Height (Inches) | Males and Females (L/Minute) |
|---|---|
| 43 | 147 |
| 44 | 160 |
| 45 | 173 |
| 46 | 187 |
| 47 | 200 |
| 48 | 214 |
| 49 | 227 |
| 50 | 240 |
| 51 | 254 |
| 52 | 267 |
| 53 | 280 |
| 54 | 293 |
| 55 | 307 |
| 56 | 320 |
| 57 | 334 |
| 58 | 347 |
| 59 | 360 |
| 60 | 373 |
| 61 | 387 |
| 62 | 400 |
| 63 | 413 |
| 64 | 427 |
| 65 | 440 |
| 66 | 454 |
| 67 | 467 |
note: It is recommended that peak expiratory flow (PEF) rate objectives for therapy be based on each individual’s “personal best,” which is established after a period of PEF rate monitoring while the individual is under effective treatment.
From National Heart, Lung, and Blood Institute (NHLBI): Executive summary: guidelines for the diagnosis and management of asthma, NIH Pub No 94-3042A, National Institutes of Health, 1994, Bethesda, Md; adapted from Polger G, Promedhar V: Pulmonary function testing in children: techniques and standards, Philadelphia, 1971, Saunders.
BOX 24-1 Use of the Peak Flowmeter and Its Interpretation
Steps to follow in using a peak flow meter:
2. Make sure that indicator is at the base of the numbered scale.
3. Ask child to take a deep breath.
4. Have the child place the peak flow meter in the mouth with the lips sealing the mouthpiece. Tell the child not to put his or her tongue in the hole of the mouthpiece.
5. Tell the child to blow out as hard and fast as possible.
6. Record the rate, but if the child coughs, do not write down that number.
7. Repeat steps 2 through 6, two more times.
Peak Expiratory Flow Rate
The maximum flow rate that is produced during forced expiration with fully inflated lungs
Differential Diagnosis
• Acute bronchiolitis, laryngotracheobronchitis, bronchopneumonia
• Bronchial foreign body aspiration
• Congenital malformations of the respiratory, cardiovascular, or gastrointestinal (GI) systems
• Tracheal or foreign body compression (e.g., vascular aortic ring, enlarged lymph nodes or tumors)
• Chronic lower respiratory tract infections caused by immunodeficiency disorders
• Gastroesophageal reflux disease
Management
The Full Report of the Expert Panel: Guidelines for the Diagnosis and Management of Asthma (ERP-3) (NHLBI, 2007) provides the most recent standards for the treatment of asthma in children. Management strategies are based on whether the child has intermittent, mild persistent, moderate persistent, or severe persistent asthma (see Table 24-1). A stepwise approach is recommended. If control of symptoms is not maintained at a particular step of classification and management, the health care provider first should reevaluate for compliance and administration factors. If these factors do not appear to be responsible for the lack of symptom control, the health care provider should go to the next step. Likewise, gradual step-downs in pharmacologic therapy may be considered when the child is well controlled for 3 months. ICS may be reduced about 25% to 50% every 3 months to the lowest possible dose needed to control the child’s asthma (NHLBI, 2007).
Chronic Asthma
• Avoid exposure to known allergens or irritants.
• Avoid use of acetaminophen in children at risk for asthma (McBride, 2011).
• Administer yearly influenza vaccine.
• Control environment to eliminate or reduce offending allergen.
• Provide allergen immunotherapy.
• Treat rhinitis, sinusitis, or gastroesophageal reflux.
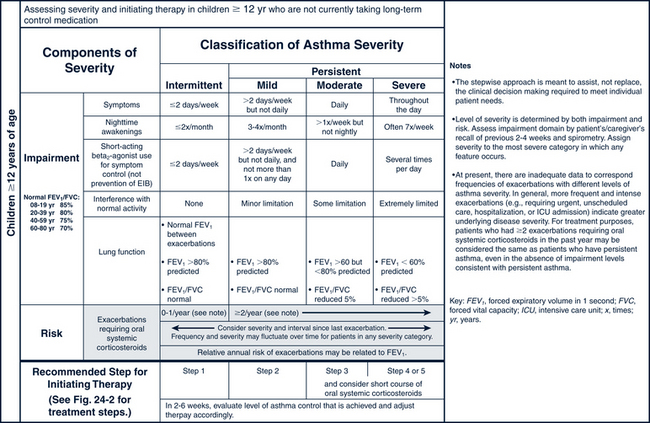
The pharmacologic management of asthma in children is based on the severity of asthma and the child’s age. The stepwise approach to treatment (Figs. 24-1 and 24-2) is based on severity of symptoms and the use of pharmacotherapy to control chronic symptoms, maintain normal activity, prevent recurrent exacerbations, and minimize adverse side effects, and nearly “normal” pulmonary function. Within any classification, a child may experience mild, moderate, or severe exacerbations. NHLBI guidelines for assessing asthma control and initiating and adjusting asthma therapy for the various pediatric age groups are found in Figs. 24-3 and 24-4.
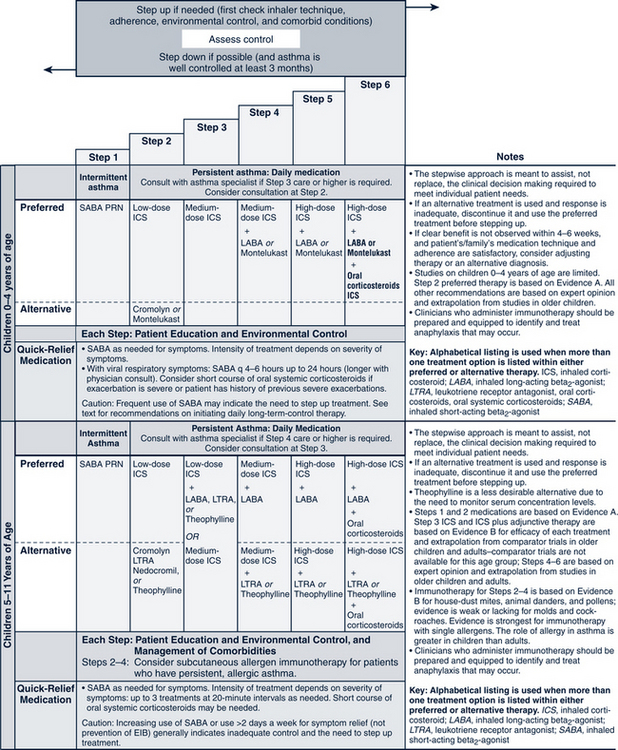
FIGURE 24-1 Stepwise approach for managing asthma in children 0 to 4 years of age and 5 to 11 years of age.
(From National Heart, Lung, and Blood Institute, National Asthma and Prevention Program: Expert panel report 3: guidelines for the diagnosis and management of asthma. Summary report 2007, NIH Pub No. 08-5846, 2007, Bethesda, MD, U.S. Department of Health and Human Services, p 42, www.nhlbi.nih.gov/guidelines/asthma/asthsumm.pdf.)
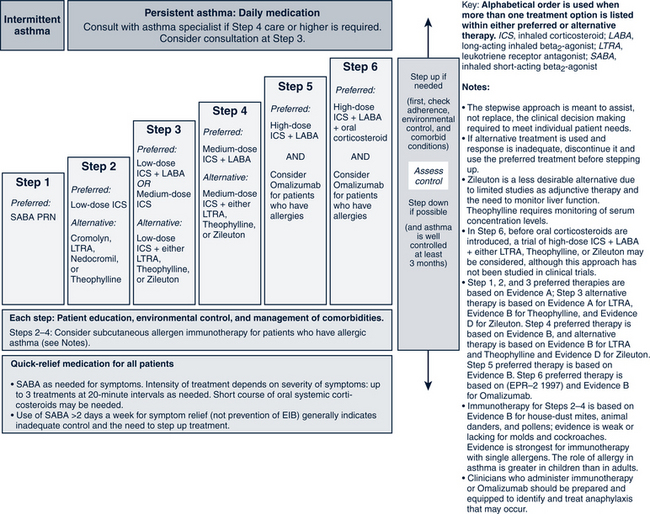
FIGURE 24-2 Stepwise approach for managing asthma in youths more than or equal to 12 years of age and adults.
(From National Heart, Lung, and Blood Institute, National Asthma and Prevention Program: Expert panel report 3: guidelines for the diagnosis and management of asthma. Summary report 2007, NIH Pub No. 08-5846, 2007, Bethesda, MD, U.S. Department of Health and Human Services, p 45, www.nhlbi.nih.gov/guidelines/asthma/asthsumm.pdf.)
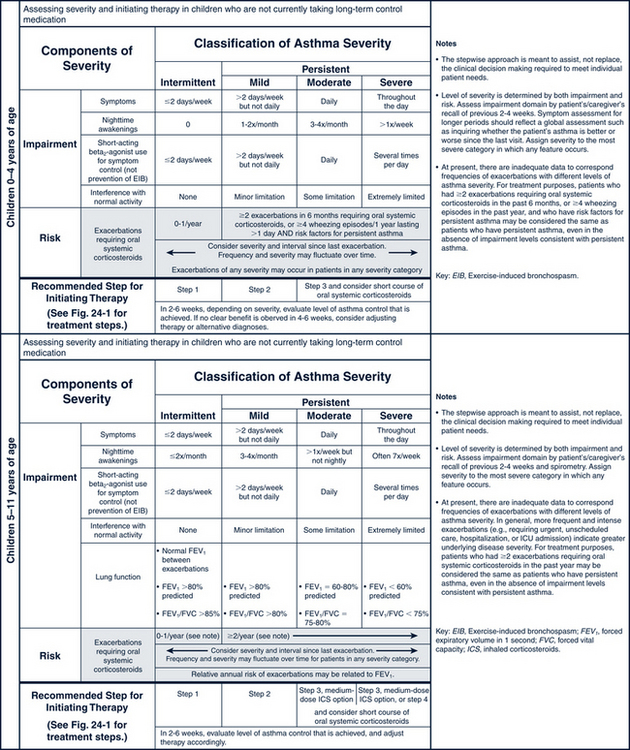
FIGURE 24-3 Classifying asthma severity and initiating treatment in children 0 to 4 years of age and 5 to 11 years of age. (From National Heart, Lung, Blood Institute, National Asthma Prevention Program: Expert panel report 3: guidelines for the diagnosis and management of asthma. Summary report 2007, NIH Pub No. 08-6846, Bethesda, MD, 2007, U.S. Department of Health and Human Services, pp 40-41. www.nhlbi.nih.gov/guidelines/asthma/asthsumm.pdf.)
Important considerations to note in the pharmacologic treatment of asthma include the following:
• Control of asthma should be gained as quickly as possible by starting at the classification step most appropriate to the initial severity of the child’s symptoms or at a higher level (e.g., a course of systemic corticosteroids or higher dose of inhaled corticosteroid). After control of symptoms, decrease treatment to the least amount of medication needed to maintain control.
• Systemic corticosteroids may be needed at any time and step if there is a major flare-up of symptoms.
• Children with intermittent asthma may have long periods in which they are symptom-free; they can also have life-threatening exacerbations, often provoked by respiratory infection. In these situations a short course of systemic corticosteroids should be used (NHLBI, 2007).
• Variations in asthma necessitate individualized treatment plans.
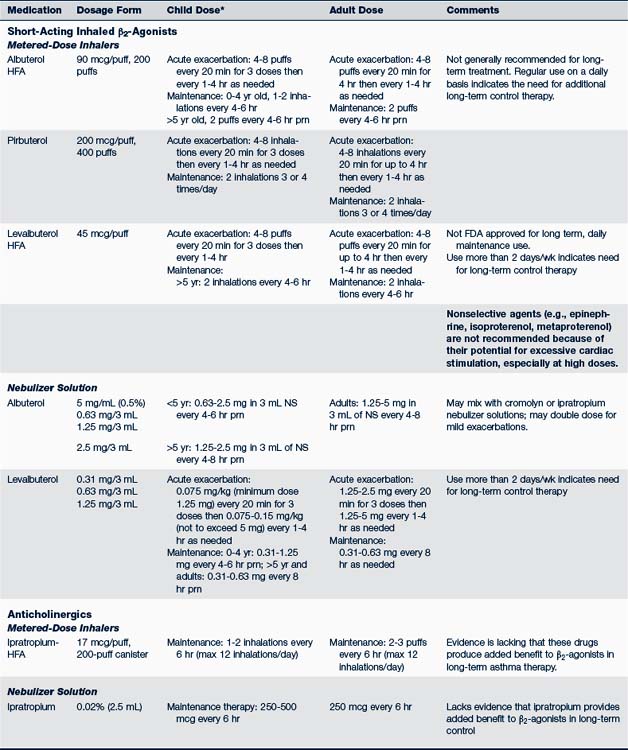
• The β2-agonist can be given by nebulization with a compressor. Nebulization can be a more effective route than metered-dose inhaler (MDI) therapy for young infants (2 years old or younger) or children who progress to moderate or severe airway obstruction.
• A spacer or holding chamber with an attached mask enhances the delivery of MDI medications to the lower airways of a child. Spacers eliminate the need to synchronize inhalation with activation of MDI. Older children can use the spacer without the mask.
• Dry powder inhalers (DPIs) do not need spacers or shaking before use. Instruct children to rinse their mouth with water and spit after inhalation. DPIs should not be used in children younger than 4 years old.
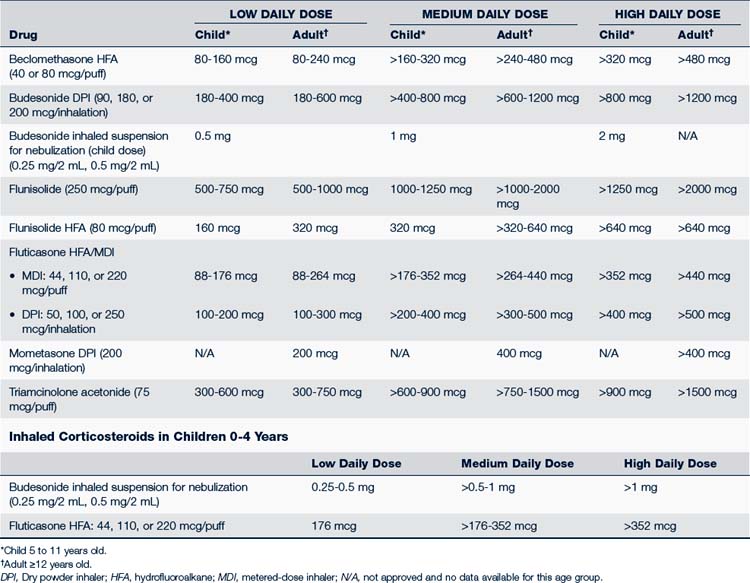
• Different inhaled corticosteroids are not equal in potency to each other on a per puff or microgram basis. Table 24-3 compares the daily low, medium, and high doses of the various inhaled corticosteroids used for children. Combination inhaled corticosteroid and long-acting β2-agonist can be used in children from 4 years of age (NHLBI, 2007; Taketomo et al, 2010).
• For treatment of exercise-induced bronchospasm:
 Use either an inhaled short-acting β2-agonist or a mast cell stabilizer (cromolyn or nedocromil) or both. Combination of both types of drugs is the more effective therapy. A long-acting β2-agonist can be used in older children.
Use either an inhaled short-acting β2-agonist or a mast cell stabilizer (cromolyn or nedocromil) or both. Combination of both types of drugs is the more effective therapy. A long-acting β2-agonist can be used in older children. Use two puffs of a β2-agonist and/or cromolyn MDI 15 to 30 minutes before exercise (Kelly and Oppenheimer, 2010). Tolerance may develop if a β2-agonist is used more than a few times a week; it should not be used as a controller monotherapy. Those who exercise regularly should use controller medication, preferably an inhaled corticosteroid. Oral montelukast can also be used on a daily schedule at least 2 hours prior to exercise (Storms, 2010).
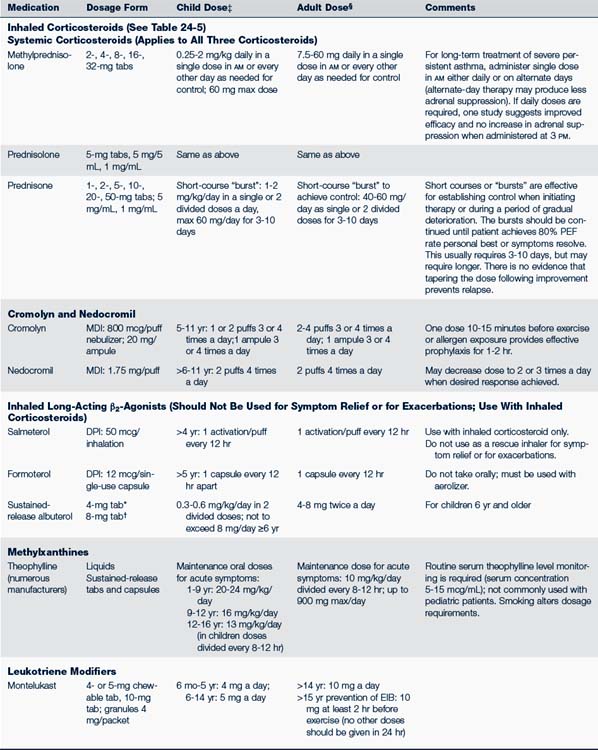
Use two puffs of a β2-agonist and/or cromolyn MDI 15 to 30 minutes before exercise (Kelly and Oppenheimer, 2010). Tolerance may develop if a β2-agonist is used more than a few times a week; it should not be used as a controller monotherapy. Those who exercise regularly should use controller medication, preferably an inhaled corticosteroid. Oral montelukast can also be used on a daily schedule at least 2 hours prior to exercise (Storms, 2010).Table 24-4 identifies the usual dosages for long-term control medications (exclusive of inhaled corticosteroids) used to treat asthma in children. Quick-relief medications are listed in Table 24-5.
Acute Exacerbations of Asthma
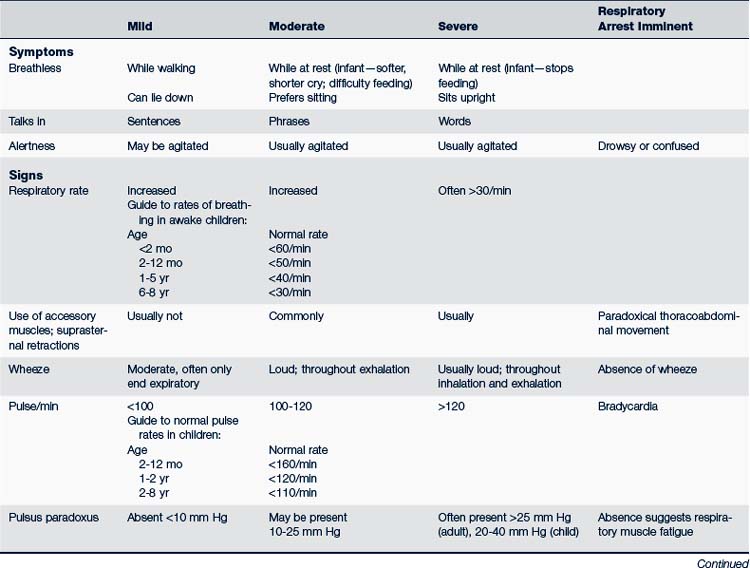
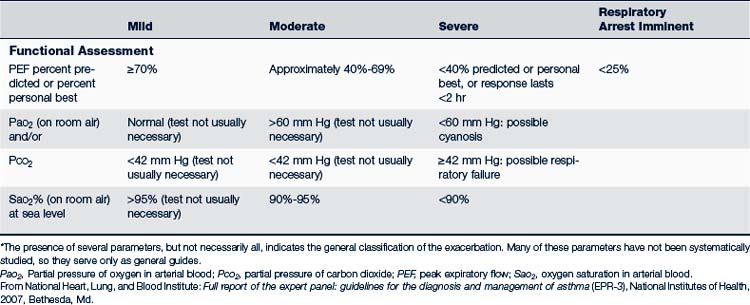
The treatment of acute episodes of asthma is also based on classification of the severity of the episode. Acute episodes are classified as mild, moderate, and severe. Signs and symptoms are summarized in Table 24-6. Early recognition of warning signs and treatment should be stressed in both patient or parent education, or both.
Characteristics of a mild acute episode are:
• Wheezing, usually at the end of expiration
• No signs of respiratory distress, cyanosis, or activity restriction
• PEF or forced expiratory volume (FEV1) greater than 70% of expected value
• Ability to speak in normal sentences between breaths
Children with a moderate acute episode of asthma manifest the following:
• Increase in respiratory rate
• Unable to walk or utter more than three to five words between breaths
• Use of accessory muscles plus lower rib and suprasternal retractions; nasal flaring
• Agitation and the ability to say only single words between breaths
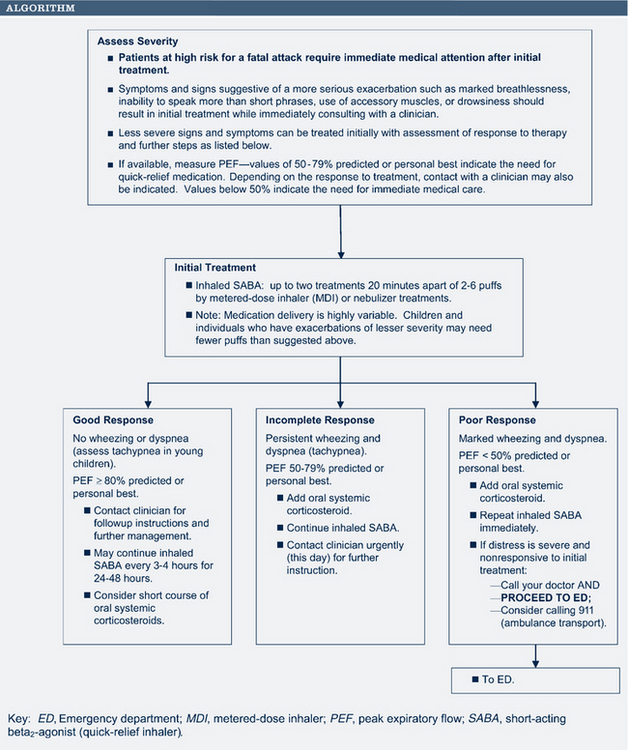
The initial pharmacologic treatment for acute asthma exacerbations is shown in Fig. 24-5. It consists of inhaled short-acting β2-agonists, two to six puffs every 20 minutes for three treatments by way of MDI with or without a spacer, or a single nebulizer treatment (0.15 mg/kg; minimum 1.25 to 2.5 mg of 0.5% solution of albuterol in 2 to 3 mL of normal saline).
If the initial treatment results in a good response (PEF/FEV1 greater than 70% of the patient’s best), the inhaled short-acting β2-agonists can be continued every 3 to 4 hours for 24 to 48 hours. Consider a 7- to 10-day burst of oral corticosteroids.
Patient and Parent Education and Prevention
• Factors responsible for asthma symptoms (i.e., inflammation, airway hyperresponsiveness, and obstruction)
• Environmental control of allergens or triggers, such as smoking and dust
• Medication use (when to take, how often to take, side effects)
• How to use inhalers, spacer devices, or aerosol equipment (Box 24-2)
• Proper cleaning of aerosol equipment
• What to do if symptoms worsen (what medications to add or increase; how frequently to use inhaled medication; specific indications about when to seek additional medical treatment for worsening of symptoms); development of a written action/treatment plan with the child or parent to cover these issues (Fig. 24-6 shows a sample home treatment plan)
• Need to have an adequate supply of all medications (including oral corticosteroids) at home and medications readily accessible to the child at school or other settings where the child frequents
• Management of the child at school, camp, or other places away from home
• The need for regular follow-up every 1 to 6 months and as needed with exacerbations
BOX 24-2 How to Use a Metered-Dose Inhaler
Adapted from National Asthma Education and Prevention Program: Facts about controlling asthma, NIH Pub No 97-2339. National Heart, Lung, and Blood Institute (NHLBI). Bethesda, Md. A reproducible handout.
Using an inhaler seems simple, but most patients do not use it correctly.
Steps for Using an Inhaler for Children Younger Than 5 Years of Age
1. The use of a mask chamber with a metered-dose inhaler (MDI) allows the delivery of inhaled medications even in an uncooperative child.
2. The child should be placed in the parent’s lap, and the mask placed around the child’s mouth.
3. Press down on the MDI while firmly holding the mask around the child’s mouth. The child will eventually take a deep breath and inhale the medication.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree